Gizzard erosion in broilers and the impact of avian adenoviruses
Published: July 13, 2021
By: Manuel Contreras,
DVM, MS, Diplomate ACPV,
Special Nutrients
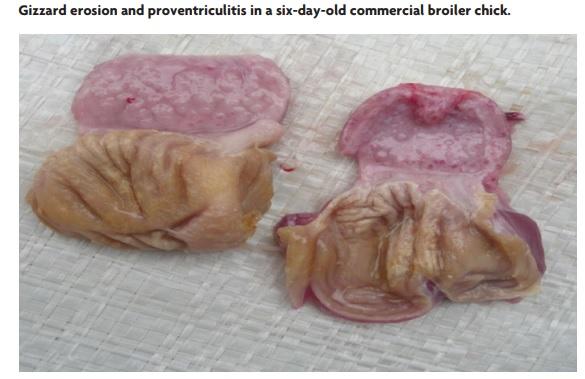
The identification of mild to moderate gizzard erosions (GE) in more than 20- 30% of the healthy commercial broilers examined in post mortem sessions, is commonly found in several countries in the Americas and Asia.
The gizzard, also called muscular stomach, is composed of a koilin (keratenoid) layer and an underlying mucosa. GE is characterised by damage in the form of erosions that can become ulcers in the koilin layer and in more severe cases in the mucosa.
Sometimes, erosions are detected as early as the first week of life or even in 18 dayold embryos in the hatchery.
Traditionally, the presence of GE in commercial broilers has been associated with the addition of some feed ingredients, the presence of mycotoxins in the ration and incorrect management practices in the hatchery and farms.
Other agents associated with this lesion include the administration of high levels of quaternary ammonium in the drinking water and nutritional deficiencies (pyridoxine, sulphur amino acids and vitamin E, among others).
Haemorrhages in the junction of the proventriculus and gizzard have been reported in flocks affected by very virulent strains of Infectious Bursal Disease Virus (vvIBDV). Velogenic strains of Newcastle disease can cause haemorrhage in the gizzard and other organs of the gastrointestinal tract. One of the relatively new culprits associated with GE are adenoviruses.
Mycotoxins and nutritional ingredients
The most important and common mycotoxins responsible for GE belong to the trichothecene group, which includes T-2 toxin and diacetoxyscirpenol (DAS). HT 2 toxin, a metabolite of T-2 toxin may also produce the lesion. Experimentally, vomitoxin (deoxynivalenol or DON), a trichothecene, has been reported as a cause of GE but only when extremely high concentrations are added to the ration (50-84 parts per million). Biogenic amines, by-products of bacterial degradation of some amino acids, are found mainly in ingredients of animal origin (meat, bone, blood, and feather meals) and are associated with poor performance in chickens.
Histamine, a biogenic amine, stimulates receptors located in the proventriculus glands and increases hydrochloric acid secretion and as a result GE. All these animal meals, as well as fish meal, have some potential for causing some degree of GE, even when they have been processed properly. Copper sulphate, a fungistatic and growth promoter, can produce GE because of overdosing, mixing errors and/or bad quality of the product.
Management in the hatchery and the farm
It is well established that keeping baby chicks in the hatchers for more than normal periods of time has a negative impact in the quality of the birds hatched and increases the incidence of GE.
For example, if eggs produced by young breeder flocks are hatched in the same hatcher as those laid by older hens, the latter will take longer to get dry. If the chicks hatched first are kept for additional hours waiting for their mates to dry up the feathers (down), a higher incidence of GE is usually reported.
As far as management in the farms, once the baby chicks are delivered from the hatchery, scientific reports have shown that gizzards from one-day-old chicks starved for the first three days of life and that were later fed normally, only showed minor ulcers after seven days’ post hatch.
After regular feeding was reestablished, GE mostly disappeared. Under commercial production, we usually visit farms where GE is reported the first or second week of life, but at the end of the production cycle no further presence is detected and performance is normal.
There are publications reporting that feed deprivation of broiler chickens at farm level from week one through week eight can be associated with the presence of GE.
Adenoviruses
In the 1990s, the first scientific reports indicating that fowl adenovirus (FAdV) was the aetiologic agents of GE came from Japan. In natural outbreaks and experimental studies, the presence of pancreatitis and lesions in the gizzard was observed.
One study from that country, investigated GE in 18 slaughterhouses and in 13 of these facilities, adenoviral GE was diagnosed using histopathology and immunohistochemistry.
Also, the virus was detected in nine of the 13 slaughterhouses evaluated. Close to a decade later, several reports came from different countries in Europe.
In 2013 a scientific paper reported several outbreaks of GE in 12 broiler flocks in Germany, caused by FAdV-1, where poor performance (low daily weight gain and mortality of up to 8%) was recorded.
Gross lesions in the gizzard included detachment of the koilin layer and ulcerative or necrotising damage of the mucosa.
Histopathologically, a necrotising ventriculitis with basophilic intranuclear inclusion bodies in epithelial cells was present.
Adenovirus-like particles were identified using immunohistochemistry, egg culture and electron microscopy. No other infectious agents were isolated.
According to most scientific reports from viruses identified in Asia and Europe, FAdV1, genotype A is the main causes of GE in commercial broilers. FAdV-4 and FAdV-8, both from the same genotype, have been reported as the aetiologic agents of a minor number of cases.
Development of immunity against FAdV-1
Published reports specify that maternal antibodies against FAdV-1 causing GE are not as important in protecting the progeny against challenge as in the case of maternal antibodies against IBH and haemorrhagic enteritis of turkeys.
On the other hand, vaccinating with a live virus from the same serotype (Chicken Embryo Lethal Orphan = CELO) has afforded protection against FAdV-1 challenge.
Some poultry companies in Asia facing field cases of GE, associated with FAdV1, have started to vaccinate with inactivated vaccines against IBH (FAdV-4 and FAdV-8), expecting cross protection against FAdV 1.
When making this decision, they do not take into consideration that most reports from Asia and Europe identify FAdV 1 as the main cause of GE.
How to differentiate if GE is caused by adenoviruses or other agents
Clinically, if mycotoxins are causing GE, the presence of some degree of gross (macroscopic) damage in other organs should be present. Usually, under field conditions, mycotoxins affect more than one organ.
That means that besides lesions in the gizzard we should see, some degree of bursal atrophy, enteritis, diarrhoea and/or lesions in the mouth.
Microscopically (histopathology), some typical changes in organs like the thymus should be present when T2 toxin or DAS are causing the damage in the gizzard.
If the lesions are caused by biogenic amines (meat/blood/bone/feather meals), we will not observe intranuclear inclusion caused by adenoviruses (histopathology).
In conclusion, there are many agents that must be taken into consideration to make an accurate diagnosis when observing GE in commercial broiler farms.
Since this lesion is multifactorial, not much emphasis is made on determining exactly what is the main cause of the problem.
Adenoviruses should be added to the list of probable aetiologic agents, depending on the clinical signs, age of the flocks and region of the world where the cases are observed.
Previously Posted on International Meat Topics • Volume 7, Number 6, 27. Positive Action.
Related topics:
Authors:
Special Nutrients
Influencers who recommended :
Tahir Naseem, Muhammad Zeeshan and 3 moreRecommend
Comment
Share
19 de enero de 2022
I have seen gizzard erosions consistently in Severe Newcastle Infections, Gumboro and Aflatoxicosis. Thank you for the broader inclusion of other causal diseases
Recommend
Reply
15 de julio de 2021
Very informative article.
I would like to add some other causes of Gizzard erosions i.e. ND, Avian Influenza, Marek's Disease,Chicken Infectious Anemia and Megalobactor infection.
Recommend
Reply
14 de julio de 2021
Excellent job, very informative article, clear n easy to understand, poultry industry will b benefited by this detailed information
Recommend
Reply
13 de noviembre de 2021
Excellent analysis off the topic
You can add the soya quality problems and the existence off nitrogen remains like’ ouria’. A substance used to increase the protein level off soya meal by false
Recommend
Reply
3 de octubre de 2021
Very nice informative topics i I would like to add some information that occasionally observed GE in caseAI infection. However vvnd could be considered as well
Recommend
Reply
Recommend
Reply
Naphco Pakistan Pvt LTD
19 de julio de 2021
Very informative thanks for share such nice information with us
Recommend
Reply

Would you like to discuss another topic? Create a new post to engage with experts in the community.













.jpg&w=3840&q=75)