IBV Variants in the Middle East
Published: September 2, 2016
By: Dr. Husam Bakri.
/ Technical and Pharmacovigilance, Manager, MSD Middle East - Dubai.
Introduction
Infectious bronchitis (IB) virus, first described in the 1930s (Schalk & Hawn, 1931), continues to be a major cause of disease in chickens of all ages and types in all parts of the world (Anon, 1988, 1991). Good quality vaccines have been available to control IB infections since the 1950s. However, despite their careful use, IB continues to be a major problem. One reason for this is the large number of antigenic types of IB that are known to exist worldwide. The first IB serotype to be described was Massachusetts (Schalk & Hawn, 1931). This was followed in the mid-1950s by the Connecticut serotype (Jungherr et al., 1956). Since that time, new IB serotypes continued to be reported from the US (Gelb et al., 1991), Europe (Davelaar et al., 1984; Cook & Huggins, 1986) and many other parts of the world. (Cubillos et al., 1991); Middle Eastern countries are also suffering from this problem.
With the help of molecular studies, it is now known that it is the S1 part of the IB virus that is responsible for the determination of its serotype. Furthermore, a new IB virus serotype(s) can arise as a result of only a very few changes in the amino acid.
The Middle East area has an estimated poultry population of 3 billion broilers, 137 million commercial layers, 30 million breeders, 2 million layer breeders and 0.5 million GP broilers. In the last few years it was noticed that breeder and layer flocks in the Middle East area were facing different problems such as false layers, penguin position, drop in egg production and no peak in production; while broilers were suffering from respiratory signs, nephritis and high mortality up to 25% with an increase when it is combined with AI-H9N2 or ND.
Although several vaccines were available in the market for controlling IB infection, new variant strains have appeared and cannot be controlled by them. Thus, it has been believed valuable to find out what type of IB variant strains are playing a role in the Middle East area and to check using live-attenuated IB vaccines belonging to different serotypes (IB Ma5 + IB 4/91) to help control different IB variant strains.
Materials and methods
Samples from infected flocks were taken from Kingdom Saudi Arabia (KSA), Jordan, Syria, Lebanon, United Arab Emirates (UAE), Kuwait, Algeria, Bahrain and Yemen and analyzed using an HI test at MSDAH- Regional Service Lab / Jordan and PCR techniques at X-Ovo lab and Intervet International R&D lab (Netherlands).
Results
The results showed that different IBV variant strains played a role in the Middle East. QX and 4/91 showed high titer (graph 1), while PCR results showed the presence of more than one IB variant (QX, 4/91,Var 2, Iranian, Sul/01/09, D274 and M41 in the same farm even in the same flocks [graph 2]).
Graph 1: % of positive resultsa of HI test. Graph 2: % of positive PCR results
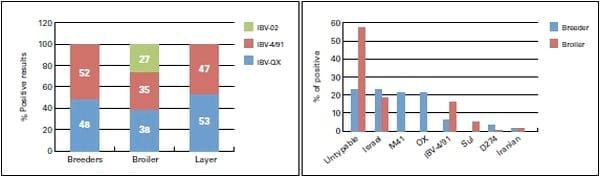
a: positive results for breeders and layers HI >6 and HI >4 for broilers.
For better IB control the ProtectotypeTM concept (Ma5 and the variant strain 4/91) was applied in breeders, layers and broilers. Involved flocks were vaccinated and monitored according to:
Broiler ProtectotypeTM program:
@ day 1: Nobilis IB Ma5
@ 14 days: Nobilis IB 4/91
@ 14 days: Nobilis IB 4/91
The following parameters were our monitoring parameters:
For broilers: the reduction in mortality, improvement in body weight, feed conversion, and for breeders and layers, production and egg quality were monitored.
The ProtectotypeTM program was implemented in two broiler farms–A & B–and according to the graph 3, 4, and 5 showed a reduction in % of mortality, increase of body weight and improvement in production index.
Graph 3: Mortality before and after the new IB vaccine protocol was implemented on Farm A. | Graph 4: Average weight before and after implementation of the new IB vaccine protocol on Farm A. | |
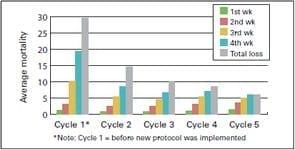 | 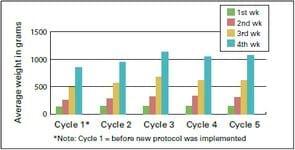 |
Table 1: Performance results on Farm B before and after implementation of the IB ProtectotypeTM protocol
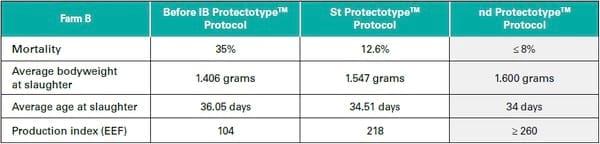
On the other hand, the following ProtectotypeTM program was applied for breeders/ layers:
@ day 1: Nobilis IB Ma5 +Nobilis IB 4/91
@ 14 days: Nobilis IB Ma5
@ 8 weeks: Nobilis IB 4/91
@ 14 days: Nobilis IB Ma5
@ 8 weeks: Nobilis IB 4/91
The following parameters were our monitoring parameters:
Egg production and egg quality
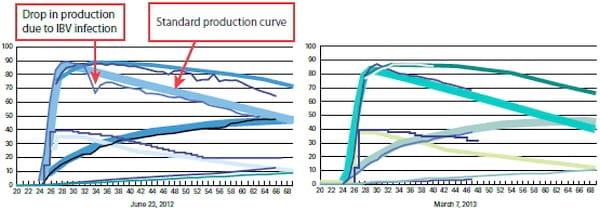
As shown in graph 5 it was noticed that there is a drop in egg production during IBV infection, starting from 32 weeks, compared to standard curve. After implementation of ProtectotypeTM program the drop disappeared and production curve returned back to the standard level as seen in graph 6.
Graph 5: Egg production.
A. During IBV infection before implementation of ProtectotypeTM. - B. After ProtectotypeTM implementation.
Additionally, many field visits to flocks to check the clinical signs, in Kingdom Saudi Arabia (KSA), Jordan, Syria, Lebanon, United Arab Emirates (UAE), Kuwait, Algeria and Yemen.
Graph 6: Clinical signs in breeders and broilers? Penguin position, oviduct fluidity and egg quality.
A. Breeders B. Broilers

Conclusion
Researchers, however, have shown that some serotypes of the IB virus can induce cross-protection against other serotypes and that a new live vaccine is not required to control each new serotype that emerges. Existing IB serotypes that can provide cross-protection are referred to as ProtectotypesTM. Recent experience on two broiler farms in the Middle East has demonstrated that after utilizing IB vaccine ProtectotypeTM protocols featuring Nobilis IB Ma5, Nobilis IB Ma5 + Clone 30 and Nobilis IB 4/91, body weight increased, mortality decreased and feed conversion improved. Additionally, in breeders, it was noticed that drop in production disappeared and production curve returned back to its standard level. Our results were consistent with the previous one that confirms the ProtectotypeTM concept (Cook et al,1999).
References
1. Anon. (1988). In E.F. Kaleta & U. Heffels-Redmann (Eds), Proceedings of the First International Symposium on Infectious Bronchitis, Rauischholzhausen, Germany.
2. Anon. (1991). In E.F. Kaleta & U. Heffels-Redmann (Eds), Proceedings of the Second International Symposium on Infectious Bronchitis, Rauischholzhausen, Germany.
3. Cook, JKA, Orbell, SJ, Woods, MA & Huggins, MB (1996). A survey of the presence of a new infectious bronchitis virus designated 4/91 (793B). Veterinary Record, 138, 178±180.
4. Cook, JKA, Orbell ,SJ, Woods MA& Huggins , MB (1999). Breadth of protection of the respiratory tract provided by different live-attenuated infectious bronchitis vaccines against challenge with infectious bronchitis viruses of heterologous serotypes. Avian Pathology, 28, 477-485.
5. Cubillos, A, Ulloa, J, Cubillos, V & Cook, JKA (1991). Characterisation of strains of infectious bronchitis virus isolated in Chile. Avian Pathology, 20, 85±99.
6. Davelaar, FG, Kouwenhoven, B & Burger, AG (1984). Occurrence and signi® cance of infectious bronchitis virus variant strains in egg and broiler production in the Netherlands. The Veterinary Quarterly, 6, 114±120.
7. Gelb, J, Wolff, JB & Moran, CA (1991). Variant serotypes of infectious bronchitis virus isolated from commercial layer and broiler chickens. Avian Diseases, 35, 82±87.
8. Jungherr , EL, Chomiak, TW & Luginbuhl, RE (1956). Immunologic differences in strains of infectious bronchitis virus. Proceedings of 60th Annual Meeting of the United States Livestock Sanitary Association, Chicago, IL (pp. 203±209).
9. Schalk, AF & Hawn, MC (1931). An apparently new respiratory disease of baby chicks. Journal of the American Veterinary Medical Association, 78, 413 ± 422.
Related topics:
Authors:
Vaxxinova
Recommend
Comment
Share

9 de septiembre de 2019
Can hussam bakri help us to identify new strains of IB.
we have issues like sudden production drops suggestive of IB symptomatically inspire of using killed IB , MA5 AND 4/91. what ever samples required can be sent where ever advised.
No mortality. no respiratory symptoms. production drops while reaching peak from 80% to 40%. Fertility also drops. confirmed not A.E. brown layer flocks 26 weeks age.
Recommend
Reply

23 de febrero de 2019
You did all your vaccines correctly.
But found a viral disease.
There is no known medicament. Reason not known. maybe vaccine did not work. maybe the flock got it before the immunity is established?
Any spray will further distress the respiratory system. Any attempt to inject will spread the problem through the needle. The spread is slow and worrying daily with deaths and drop in production.
So what is left in doctor's hand. you spread it fast and it will end soon. Better you use an attenuated spread (vaccine) rather than the disease virus spreading slowly in vaccinated birds.
Recommend
Reply
Vaxxinova
2 de abril de 2017
Dear Sawsan ,
First thank you for your message . It has mentioned on my article, IB 4/91 , QX , Vart 2 , selumaneh , Iranian strains .
All the best
Bakri
Recommend
Reply
Vaxxinova
18 de octubre de 2016
Dear Dr. Bahram ,
First thank you very much for your comment and question.
1- Using IB 4/9 + Clone30 at day one 100% is ok . in the middle east we are using it since we started to have early IB variant challenge as you know ND also is challenging at early age that why they need to cover IB and ND from the first day.
2- As you know vaccines before be in the market , safety study is one from the major point has to be clear to avoid the point you mentioned and I am sure most of the good companies do that .
Hope I could cover your points.
All the best
Bakri
Recommend
Reply
.jpg&w=3840&q=75)

Candidate Genes Associated with Survival Following Highly Pathogenic Avian Influenza Infection in Chickens
Suggested link
Vaxxinova
21 de septiembre de 2016
Dear Dr. Mohamed ,
Thank you for comments . Great work
All the best
Bakri
Recommend
Reply
Vaxxinova
19 de septiembre de 2016
Salam alekum ,
For sure I will just send me your email ASAP you will get them .
Have a nice day
Recommend
Reply
Vaxxinova
18 de septiembre de 2016
Salam Dr.
We have done more than 30 cross protraction study with 30 variants strain are playing a role over the world incloding Middle East . Results showed very good protection level for these strains . We strongly believe that program can give very good results . The most important is to control other diseases are playing a role .
All the best
Bakri
Recommend
Reply
Vaxxinova
18 de septiembre de 2016
Salam,
We are advising the following vaccination program to cover ND , IB and AI sure gumboro is included but the time of Gumboro vaccination deepened on MDA:
- Day one Ma5+Clone30 Spray course
ND+AI killed at day one.
14-16 days Clone30+IB 4/91 spray.
The above vaccination program gave excellent results.
We do have emergency vaccination program against IB specially when we get variant IB at early age we advise the following:
MA5+4/91 at day one
Ma5+Clone30 at day 14 .
Hope this will help you according to your area.
All the best
Bakri
Recommend
Reply
Vaxxinova
17 de septiembre de 2016
Dear Dr. Som ,
Thank you for your feedback it is very useful for all of us and it is highly appreciated .
I fully agree with all your comments , that why due to may variant strains recently playing a role and not easy to produce vaccines for them in short term. Now a days most of the countries using vaccines available in the market can give good cross protection against the new variant strains. MSD did cross protection studies in in SPF chicken for more than 30 variant strains has been detected over the world by using Ma5 and 4/91 results were very good from this combination. in 2006 we have faced QX challenge in our area we have control it by using Ma5+4/91 at day old with excellent results.
All the best
Bakri
Recommend
Reply


Evonik Animal Nutrition
EVONIK ANIMAL NUTRITION is committed to improve sustainability, health and efficiency along the food chain to feed growing population with animal protein.
Suggested link
Vaxxinova
17 de septiembre de 2016
Dear Dr. Siddig ,
First happy eid to you and your family. Absolutely you are right these 2 diseases are challenging the poultry industry in our regain also in Asia regain . In all our advice we have never ignored AI OR ND to have high priority in control program other wise we will not have been able to control the problem ( like we are closing the door and we are the windo open ) we have discovered that IB results in more damages when with AI or ND compered when it is IB alone .
Therefore we are advising our customers to look to the problem as package and not to ignore any of them even when only one of them showing the problem .
All the best and have a nice day. Please feel free to contact me any time
Bakri
Recommend
Reply

Would you like to discuss another topic? Create a new post to engage with experts in the community.







