Conclusions
- The dietary inclusion of Bacillus amyloliquefaciens CECT 5940 (Ecobiol®), antibiotic growth promoter (AGP), or competitor probiotics (Bacillus subtilis A, and B) significantly improved feed conversion, reduced footpad lesion and C. perfringens in ileum while providing better carcass yield compared to the challenge group without additives.
- Furthermore, uniformity of the body weight was significantly better in birds fed Ecobiol®, AGP or competitor B. subtilis A compared to the challenged group without additives.
- Additionally, dietary inclusion of Ecobiol® significantly provided lower wet litter score, fecal score, and overall enteritis score compared to the challenged group without additives.
Introduction
Coccidiosis is a common problem in broiler production, resulting from the development and reproduction of Eimeria species that can infect specific areas of the gut and cause tissue damage in the intestinal epithelial cells (Ritzi et al., 2014; Ritzi et al., 2016). In general, producers have been using anticoccidians to control Eimeria and avoid further development of enteric diseases such as necrotic enteritis arising from intestinal overgrowth of Clostridium perfringens (Awad 2017). In this context, antibiotic growth promoters (AGP) have been also used to control the growth of pathogenic bacteria while improving performance and gut health. However, concerns over antibiotic resistance led to regional bans in order to reduce AGP use in poultry feed. Consequently, the use of probiotics has received special attention as potential alternatives to replace AGPs since they may provide similar improvement in performance. Probiotics are living microorganisms provided in the feed, which are able to reduce pathogen load by competitive exclusion and can enhance health and nutrient absorption in the host. Ecobiol® is a probiotic product containing Bacillus amyloliquefaciens CECT 5940 strain, which is a naturally occurring strain with fast growing characteristics and high tolerance to gastric and bile secretions. Bacillus amyloliquefaciens CECT 5940 is able to improve feed conversion and reduce pathogenic bacteria in the intestine (Facts & Figures n°15160). Other commercially available probiotics are based mainly on Bacillus subtilis strains, which may have different mode of action compared to Bacillus amyloliquefaciens strains but, at the end, promote performance and improve gut health as well. Therefore, this study was conducted in order to evaluate the efficacy of Bacillus amyloliquefaciens CECT 5940 (Ecobiol®) in promoting both performance and gut health compared to AGP and other commercially available probiotics in broilers under challenge condition. This trial was conducted by Dr. Nilva Kazue Sakomura at “São Paulo State University (UNESP)” in Brazil.
Materials and Methods
One thousand eight-hundred thirty-six day-old male chicks (Cobb 500) with an average initial weight of 50.2 ± 0.1 g were randomly assigned to one of the six dietary treatments with nine pen replicates of 34 birds each. The basal diets composed by corn-soybean-meat and bone meal were formulated to meet Evonik amino acids recommendations (AMINOChick® 2.0) for starter(day 1-13), growerI (day 14-21), grower II (day 22-35) and finisher (day 36-42) phases with all diets provided in mash form. The treatments consisted of non-challenged group receiving a basal diet without feed additives; Challenged group receiving a basal diet without feed additive; Challenge + 0.05 g/kg BMD (Bacitracin methylene disalicylate); Challenge + 1g/kg Ecobiol® (1 x 106 CFU/g of Bacillus amyloliquefaciens CECT 5940); Challenge + 0.100 g/kg Competitor A (1 x 106 CFU/g of B.subtilis DSM17299) and Challenge + 0.05g/kg Competitor B (1 x 105 CFU/g of B. subtilis PB 6). The enteric pathogen challenge consisted of inoculation by oral gavage on day 17 with 1mL of Eimeriamaxima (5.0 X 104 CFU/g) and Clostridium Perfringens (2.5 X 106 CFU/g) on days 18, 19, and 20. Feed and water were provided ad libitum during the experimental period.
Body weight and feed intake were recorded on days 21, 35, and 42 of the experimental period for calculation of weight gain and feed conversion ratio. Cause, date andweight of dead birds were recorded daily. Score of litter moisture (0 = Dry friable; 1 = Dry with very fine texture; 2 = Sticky on compression or crumbles; 3 = Clod on compression; 4 = Wet; 5 = Drops of water come out on compression), fecal (0 = Solid with white cap; 1 = Bulckyand moist; 2 = Watery; 3 = No consistency, watery, mucus, undigested feed, sloughed mucosa, foamy) and cecal droppings (0 = Black to brown and firm, 1 = Lack consistenceand foamy; 2 = watery and foamy; 3 = Foamy like mousse, spread, liquid) were evaluated at day 28. Two birds from each pen were randomly selected and sacrificed on days 21 and 28 to evaluate general intestinal health and to collect digestasamples from ileum and cecum for further analysis. The score evaluation of the intestinal health considered the presence (1) or absence (0) of the following parameters: ballooning, Muscle tone, thin or fragile, abnormal content (excessive slime, water, gas, greasy aspect or mixture of these), swollen or red, sloughed mucosa or presence of undigested feed. The contents of the ileum and cecum were collected and stored at −20°C for Clostridium perfringens counts. Quantitative PCR targeting the 16S rRNA genes of C. perfringens was used in the quantification. Body weight uniformity, footpad lesions (0 = none; 1 = mild; 2 = moderate; 3 = Severe; 4 = extreme), hock burn (0 = none; 1 = mild; 2 = moderate; 3 = Severe; 4 = extreme), carcass and breast meat yields were evaluated on days 42. Treatments were statistically analyzed using one-way ANOVA procedure in SAS v9.4 software followed by mean comparison with SNK (Student-Newman-Keuls) test at 5% of probability and frequency of the scores were evaluated using a non-parametric Kruskal-Wallis test followed by Dunn-Bonferroni test at 5% probability.
Results and Discussion
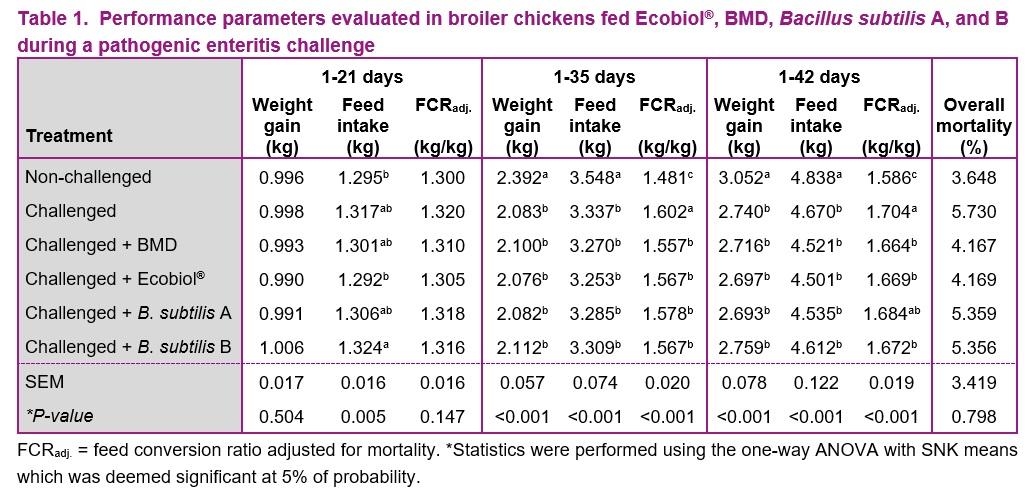
The results of the performance are presented in Tables 1. Feed intake was higher in the challenged group receiving B. subtilis B compared to the non-challenged group and the group receiving Ecobiol® (P < 0.05), but no significant differences were observed when compared to the challenged group without additives and the groups receiving BMD or B. subtilis A (P > 0.05) on day 21. Feed conversion ratio and weight gain were not significantly different between the treatments at 21 days (P > 0.05), which was the day right after the challenge. On the other hand, feed intake and weight gain were significantly lower in the challenged groups compared to the unchallenged group at 35 and 42 days (P < 0.05). The birds undergoing the enteric pathogen challenge had significantly better feed conversion ratio when fed any of the feed additives on day 35 compared to the challenge group without feed additives (P < 0.05). The challenged birds receiving Ecobiol®, BMD or B. subtilis B had significantly better feed conversion ratio compared to the challenge group without feed additives (P < 0.05) on day 42. The improvement with B. subtilis A was not enough to provide a significant difference on day 42 compared to the challenged group without additive (P > 0.05). Mortality was not significantly different between the treatments (P > 0.05).
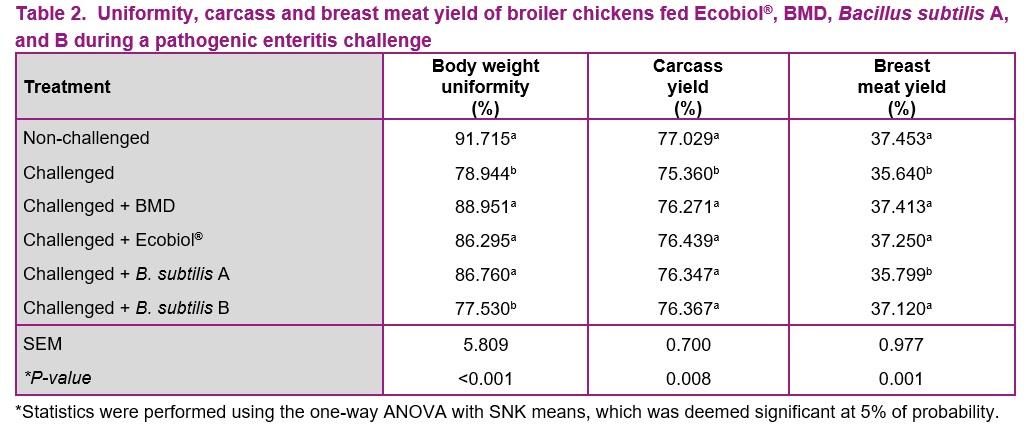
The results for uniformity, carcass and breast meat yields are presented in Table 2. Uniformity in the body weight, carcass and breast meat yields were improved in the challenged groups receiving either Ecobiol® or BMD compared to the challenged group without any feed additive (P < 0.05). The challenged group receiving B. subtilis B had significant improvement in breast meat yield (P < 0.05), but no improvement in uniformity (P > 0.05) compared to the challenged group without additives. On the other hand, the challenged group receiving B. subtilis A had significant improvement in uniformity (P < 0.05), but no improvement in breast meat yield (P > 0.05) compared to the challenged group without additives.
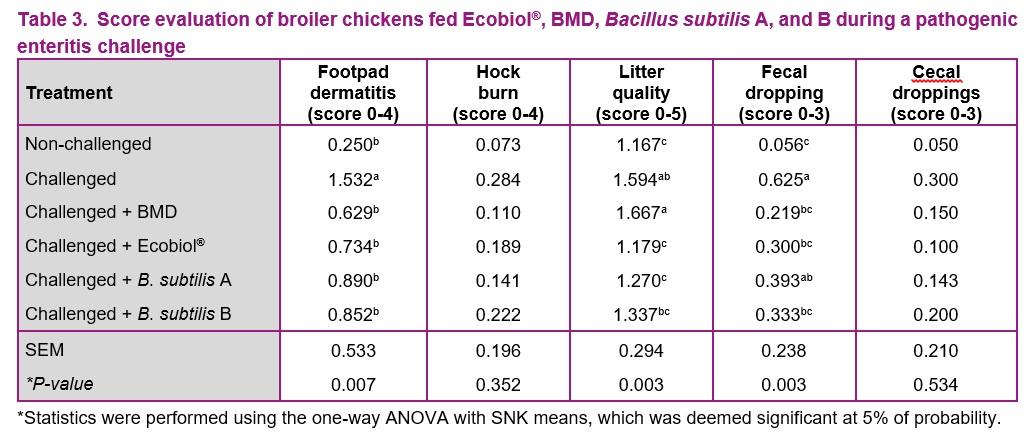
The results of the score evaluation are presented in Table 3. The average score for hock burn and cecal droppings were not significantly different between treatments (P > 0.05). There was an improvement in the average wet litter score of the challenged groups receiving Ecobiol® or B. subtilis A compared to the challenged group without feed additives (P < 0.05), but no significant improvement in groups receiving BMD or B. subtilis B (P > 0.05). The challenged groups receiving BMD, Ecobiol® or B. subtilis B presented lower fecal score compared to the challenged group without additives (P > 0.05), which was not significantly different from the challenged group receiving B. subtilis A (P > 0.05). The litter and dropping scores may have influenced in the observed footpad dermatitis score, being higher in the challenged group compared to the other treatments (P < 0.05).
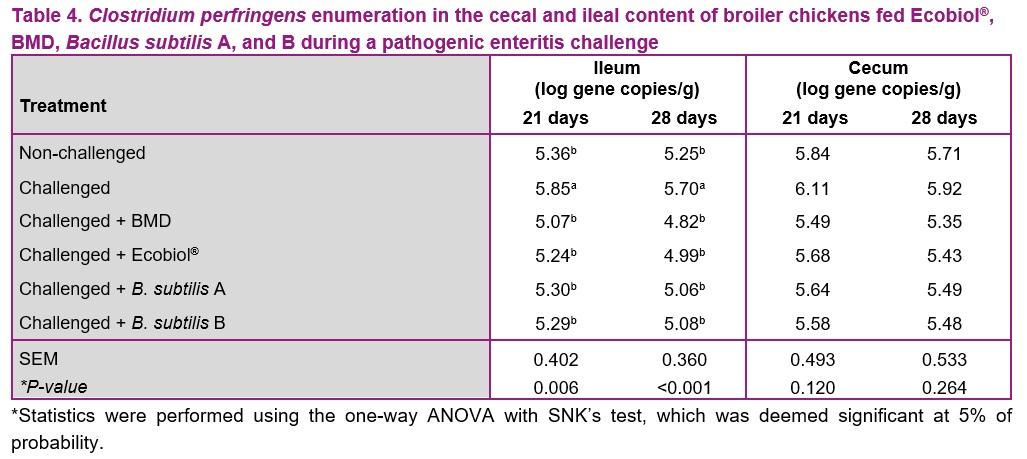
The results of the rt-qPCR analysis are presented in Table 4. The analysis indicated that BMD, Ecobiol®, Bacillus subtilis A, and B were able to reduce the numbers of clostridium perfringens in the ileum at 21 and 28 days. However, there was no significant differences between treatments for clostridium perfringens in cecum (P > 0.05) either at 21 or 28 days.
The observations of the intestinal health score are presented in Table 5. It was not observed the presence of undigested feed or weak muscle tone in the different treatments (equal to 0). The presence of abnormal content (excessive slime, water, gas, greasy aspect or mixture of these) was similar between the challenged groups at 21 days. There was no significant difference between treatments at 21 days for ballooning, thin mucosa, swollen intestine or sloughed mucosa (P > 0.05).
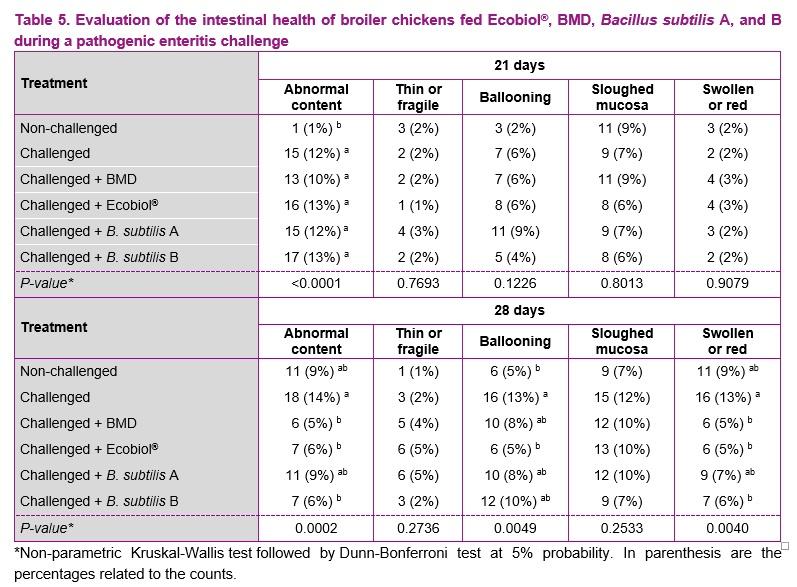
At 28 days of age, the challenged groups receiving Ecobiol®, BMD or B. subtilis B presented significantly lower frequency for abnormal content and swollen intestine compared to the challenged group without feed additives (P < 0.05), but not for B. subtilis A. A significant lower ballooning frequency was only observed in the challenged group receiving Ecobiol® and the unchallenged group, while the challenged groups receiving BMD, B. subtilis A, and B were not significantly different from the challenged group without feed additives (P > 0.05). There was no significant difference between treatments at 28 days for thin mucosa or sloughed mucosa (P > 0.05).
The main observations from necropsy as presented in Table 5 are common characteristics in dysbacteriosis and they are related to Infectious agents such as Clostridium perfringens, which may exacerbate Eimeria infections (Chapman et al., 2002; De Gussem, 2007). Therefore, the reduction of C. perfringens observed in the groups receiving Ecobiol®, BMD and B. subtilis B (Table 4) is one of the main factors resulting in the observed lower frequency of abnormal content and inflammation in the gut at 28 days of age. Although the reduction in C. Perfringens counts was evident, the treatments receiving BMD, B. subtilis A or B may still present some dysbacteriosis at 28 days and this may explain the presence of ballooning. In addition, dysbacteriosis is associated with an absolute reduction in available absorptive surface area (Teirlynck et al., 2011), which may explain why fecal or wet litter scores in these treatments were not significantly different from the challenged group without feed additives.
In conclusion, the results indicates that B. amyloliquefaciens CECT 5940 (Ecobiol®) and the other commercially available probiotics were effective as the AGP in promoting performance, however B. amyloliquefaciens CECT 5940 has potential to bring additional benefits such as improved uniformity, breast meat yield and improve enteric health.