Introduction
Milk is a liquid consisting of 86-87% water and this means that it is a bulky and heavy commodity. Milk is also produced on a daily basis. As a result, milk requires high cost transportation and there is a cost limit on the range over which it can be sold. It will only keep for a few days, which places a time limit on the period during which it can be used or processed and transformed into more shelf stable products. (Banda, 2010).
Milk and milk products play an important role in human nutrition throughout the world. Milk is also highly perishable and can easily be adulterated whilst the quality of the milk is highly dependent on farm management. Strict and comprehensive dairy regulations are therefore customary and necessary (Banda, 2010). The fluid or semi-fluid nature of milk and its chemical composition renders it one of the ideal culture media for microbial growth and multiplication (Mogessie and Fekadu, 1994).
The safety of dairy products with respect to food-borne diseases is a great concern around the world. This is especially true in developing countries where production of milk and various dairy products take place under rather unsanitary conditions and poor production practices (Mogessie, 1990; Zelalem and Faye, 2006). Moreover, the composition of milk makes it an optimum medium for the growth of microorganisms that may come from the interior of the udder, exterior surfaces of the animal, milk handling equipment and other miscellaneous sources such as the air of the milking environment (Richardson, 1985). The conventional procedure for measuring the sanitary quality of milk is to estimate its bacterial content. The number of bacteria in aseptically drawn milk varies from animal to animal, even from different quarters of the same animal. Aseptically drawn milk from healthy udder contains the average of 500 –1000 bacteria mL-1. The initial counts of 105 bacteria mL-1 in milk are evidence of poor production hygiene. Milk produced under hygienic conditions from healthy animals should not contain more than 5 × 105 bacteria per milliliter (mL) of milk (O’Connor, 1994).
The number and types of micro-organisms in milk immediately after milking are affected by factors such as lack of knowledge about clean milk production, use of unclean milking equipment and lack of potable water for cleaning purposes contributing to the poor hygienic quality of raw milk (Bekele and Bayileyegn, 2000).
The traditional milk handling and processing practices and raw milk quality in Eastern Wollega was reported by Alganesh (2002); traditional handling practices, preservation, utilization and consumption of dairy products and marketing systems in East Shoa Zone was reported by Lemma et al. (2005a, b) and production, handling, traditional processing practices and quality of milk in Bahir Dar milk shed area, was reported by (Asaminew, 2007). However, little is documented about milk composition and microbial quality in the Awi Zone in general and in Dangila District in particular. This study is thus aimed at filling the gap in this regards. The specific objective is, therefore:
- ·To determine the microbial quality and chemical composition of raw milk in the urban and peri urban areas of Dangila Town.
Materials and methods
Description of the Study Area
The study was conducted in Dangila district, which is found in Agew Awi zone, Amhara Regional State, in north-western Ethiopia, along the main road between the cities of Addis Ababa and Bahir Dar about 472 km from Addis Ababa, the capital of Ethiopia. From the regional capital, Bahir Dar, Dangila district is about 72 km. Astronomically, Dangila is located at 11° 18' N latitude and 36° 57' E longitude. Geographically, it is located on elevation of 2200 m above the sea level on area coverage of 9486.4 hectares with Woina Dega (temperature) climate. The annual average rainfall and temperature are 1576 mm and 17 0c, respectively. The total population of the district is 50,755, practicing three major religions. Orthodox 88.34%, Muslim 11.06% and Protestant 0.6%. The borders of the district are, on the south Faggeta Lekoma, on the southwest Guangua, on the northwest Jawi, and on the northeast West Gojjam Zone (Assaye, 2009).
Methods of Milk Sampling
The physical property (pH and temperature) and sensory quality of the samples of raw milk were observed and evaluated at sampling and at arrival of the laboratory and recorded accordingly to ensure it is normal milk. A total of thirty milk samples were taken (15 farm families or farmers from urban and 15 from peri urban milk production system among those surveyed). Samples of raw morning milk were taken from each household once every week over a period of two weeks for determination of the microbiological quality and chemical composition of raw milk from each production system following standard methods as described by Marth (1978).
The cows, which were selected for milk sample collection, were physically evaluated for health and hygienic condition prior to sampling. 300 ml milk samples were collected aseptically from individual cows milking from containers immediately after milking. Milk samples for chemical composition were collected depending on cows’ breed type while for microbial quality cows breed were not considered. Crossbred and local breed cows milk were collected in different week in different bottle for milk chemical composition analysis.
The samples collected were placed in a sterile container and kept in an ice box while transporting to Bahir Dar University Food and chemical analysis laboratory for analysis. All analyses were performed within 2 hours of sampling. Each farmer or farm households were visited twice for sample collection. In the First week, 15 samples from each urban kebeles were collected and analysed. In the second week, following the same procedure 15 samples from the peri urban areas were collected for laboratory analysis. In the third and fourth week samples from peri urban and urban areas were collected and analysed, respectively.
Milk Microbiological Tests
The microbial test of raw milk was analysed in Bahir Dar university food science laboratory within 2 hours of collection. Standard plate count, yeast and mould count and coliform count microbial tests were used for fresh raw milk. For determination of standard plate count, yeast and mould count and coliform count, peptone water was sterilized by autoclaving at 121ºC for 15 minutes. Similarly, the total plate count agar (Oxoid) used for determination of total viable organisms was sterilized by autoclaving at 121ºC for 15 minutes, while the violet red bile agar (VRBA: Oxoid) used for determination of Coliform Count was sterilized by boiling (Richardson, 1985).
Standard Plate Count (SPC)
For total plate count, appropriate serial dilutions were selected that would give the expected total number of colonies on a plate, i.e., between 30 and 300 colonies (Richardson, 1985). The standard plate count (SPC) agar was cooled to 450C after autoclaved and before pouring to petri-dish. One ml of milk sample was added into sterile test tube containing nine ml peptone water up to serial dilution of 10-4 and mixed thoroughly. Then one ml of the sample from appropriate decimal dilution was placed on a petri-dish and then molten agar medium (10- 15 ml) was poured onto the petri-dish and then it was incubated for 48 hours at 32°C. Finally, colony count was made using colony counter.
The estimated numbers of colonies were calculated by the formula given by FDA (2008).
N = ΣC(1*n1) + (0.1 * n2)x d
Where, N = Number of colonies per ml of milk sample
ΣC = Sum of all colonies on plates counted
n1 = Number of plates used in lowest dilution counted
n2 = Number of plates used in highest dilution counted
d = dilution factor of the lowest dilution used.
Coli Form Count (CC)
Serial decimal dilutions were prepared using 0.1% peptone water. Duplicate appropriate decimal dilutions were surface plated and incubated at 32°C for 24 hours on Violet Red Bile Agar and typical dark red colonies on plates were considered as coli forms and counted. This was followed by a confirmatory test by transferring and incubating four to five typical colonies from each plate into tubes containing 2% Brilliant Green Lactose Bile. Gas production within 48 hours of incubation at 35°C was considered as sufficient evidence for the presence of coli forms (Richardson, 1985).
Yeast and Mould Count (YMC)
Samples of milk were serially diluted in a peptone water and volumes of 0.1 millilitres of appropriate dilutions were plated in duplicate Petri-dishes by the pour plate techniques using chloromophenicol agar (the agar consisted of 5g yeast extract, 20g glucose, 0.1g chloramphenicol, 0.01g bromophenicol blue, and 15g agar per liter of distilled water) at a pH of 6.0 to 6.2.The dried plates were then incubated at 25oC for 3 to 5 days. Colonies with a blue green color were counted as yeasts and moulds (Yousef and Carlstrom, 2003).
Milk Chemical Composition Analysis
Milk fat: Gerber method was used to determine the milk fat content. Milk samples were kept at 370C for 30 minutes in a water bath to maintain the milk to normal body temperature of the cow. Ten milliliter of concentrated sulphuric acid was pipetted into a butyrometer. Then 11 ml of milk was added using milk pipette into a butyrometer having the sulphuric acid and then one milliliter of amyl alcohol was added. The butyrometer stoppers were put on and the samples were shaked and inverted several times until all the milk digested by the acid. Then the butyrometers were placed in a water bath at 650C for five minutes. The samples were placed in a Gerber centrifuge for four minutes at 1100 rpm (rotations per minute). Finally, the samples were placed into water bath for 5 minutes at 65 0 C and fat percentage was read from the butyrometer (Van den Berg, 1988). The average of duplicate readings were computed and recorded.
Total solids: To determine the total solids, five grams of milk sample were placed in a pre-weighed and dried with duplicate of crucibles. The samples were kept at 1020C in a hot air oven overnight. Then, the dried samples were taken out of the oven and placed in a desiccator. Then the dry samples were weighed (O’Connor, 1994).
Total solid=(crucible weight +Oven dry sample weight-crucible weight) x100Sample weight Solids- not -fat: The solids not fat (SNF %) was determined by subtracting the percent fat from total solids (O’Mahoney, 1988).
% SNF = (% TS-% Fat) x 100
Total Ash: The total ash was determined gravimetrically by igniting the dried milk samples in a muffle furnace in which the temperature was slowly raised to 5500C. The samples were ignited until carbon (black color) disappears or until the ash residue becomes white (Richardson, 1985).
Percent ash= (Weight of residue/Weight of sample) x100 Total protein: Formaldehyde titration method was used to determine the total protein content of milk (O’Connor, 1994). Ten ml of milk was added into a beaker. Then 0.4 ml of 0.4 percent potassium oxalate and 0.5 ml of 0.5 percent phenolphthalein indicator were added into the milk. It was allowed to stand for two minutes and then the mixture was titrated with N/9 sodium hydroxide solution until pink colour was obtained. At this stage, two ml of neutral 40 percent formalin (the formalin solution was made neutral by adding a few drops of phenolphthalein and then adding sodium hydroxide drop by drop until a faint pink colour was obtained) was added to discharge the pink colour. The titration was continued until a pink color of equal intensity was again obtained. Finally, the number of ml of the N/9 NaOH used after the addition of formalin multiplied by 1.74 gives the percentage protein in the milk (O’Connor, 1994).
Percent Protein = Burette reading x 1.74
Percent lactose =%Total solid-(%Protein+%Fat+%Ash)
Data Management and Statistical Analysis
Microbial count data was first transformed to logarithmic values (log10) before statistical analysis. Then, data on the chemical composition and the transformed microbial count values were analysed using the General Linear Model (GLM) procedure of Statistical Analysis System (SAS, 9.1). Mean separation was carried out using the simple t-test technique. Means and differences were considered significant at P<0.05.
- The following model was used for the analysis of the microbial quality of raw milk.
Yij = μ + βi + eij
Where Yij = individual observation for each test
μ = the overall mean
β = the ith urban and peri urban effect
eij = the error
- The following model was used for the analysis of the chemical composition of raw milk.
Yij = μ +β i+Li + eij
Where, Yij = individual observation for each test
μ = the overall mean
β = the ith urban and peri urban effect
L = the ith Cow breed effect
eij = the error
Results and discussion
Chemical Composition and Microbial Quality of Raw Milk
Chemical Composition of Milk
The mean (± SD) protein, fat, total solids (TS), solids-not-fat (SNF), and ash contents of local cows’ milk produced in urban and peri urban farms are indicated in Table 1.
Table 1: Average (± SD) chemical composition of crossbred cows' and local breed cows’ milk produced at urban and peri-urban farms in the study area.
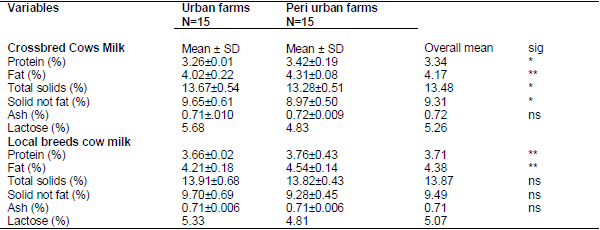
N = number of observations; SD = Standard deviation, ns = not significant (P> 0.05),* = significant (P< 0.05) ** = highly significant (P< 0.01), Sig. = Significance
The current study is comparable with the finding of Derese, (2008) in west shoa zone of Oromia region who reported the overall mean protein, fat, total solids, solid-not-fat (SNF) and ash contents of local cows' milk were 3.3, 5.14, 13.55, 8.41 and 0.74 percent. Similarly, Asaminew (2007) also reported the overall mean fat, protein, total solids, ash and solids-not-fat (SNF) contents of local cows’ milk produced in the study area were 4.71, 3.25, 13.47, 0.73 and 8.78 percent, respectively in Bahir Dar milkshed area.
The current study protein content mean of the area of local cows is higher than the finding of Derese (2008) but less than that of reported by Asaminew (2007) in West Shoa zone of Oromia region and Bahirdar milk shed area respectively. The current local cows’ fat content report is lower than the finding of Derese, (2008) and Asasminew, (2007).The total solid and solid not fat reported in the current study is higher than the finding of Derese (2008) and Asaminew(2007).while the fat content was lower than the report of both Asaminew (2007) and Derese (2008).
The current study result of protein, fat, SNF and ash content of crossbred cows milk produced in the urban farm is lower than the report of Derese (2008). While the total solid and ash content was higher than the report of Derese (2008) whose result was; the mean protein, fat, total solids, solid-not-fat (SNF) and ash contents of crossbred cows' milk produced in the study area were 3.67, 4.27, 13.07, 8.89 and 0.70 percent in urban farm, respectively. While these values were 3.7, 4.37, 13.18, 8.78 and 0.71 percent in peri-urban farm which were more similar with the result of current study. A similar report was done by Asaminew (2007), who reported the overall mean fat, protein, total solids, ash and SNF contents of crossbred cows’ milk were 4.14, 3.45, 13.15, 0.70 and 8.96 percent, respectively. All results of chemical composition of crossbred cows reported by Asaminew (2007) were more similar to the current study.
Microbiological Quality of Milk
The microorganisms present in milk can originate from interior of the udder, its exterior and/ or milking equipment. High initial microbial count in milk of >105 cfu/ml is evidence of serious faults in milk production hygiene, whereas production of milk having counts consistently below 105 cfu/ml reflects good hygiene practices (Ombui et al., 1995). A standard plate counts of 1x10 5 cfu/ml has been widely adopted for good quality raw milk intended for treatment before liquid consumption. However, some other countries have adopted different standards suited to local conditions. For example, the standard plate count for America is no more than 3x105 cfu/ml, while the standard for Kenya is no more than 2x106 cfu/ml (Ombui et al., 1995). In Sweden, the accepted limit for the total number of bacteria and somatic cell count in raw milk is 1x105cfu/ml and 4.99x103 somatic cells/ml, respectively (Maret, 1994). The standard plate count for pasteurized milk should be less than 30,000 cfu/ml (Saskatchewan, 1997).
The overall mean total bacterial count of cows’ milk produced in the study area was 6.14±0.72log10 cfu/ml, which is less than the finding of Asaminew (2007) who reported that total bacterial count of raw milk produced in Bahir Dar milk shed area was 7.6 log10cfu/ml. Fekadu (1994)reported that the minimum and maximum total bacterial count of raw cows’ milk produced in southern region to be 6 to 8.8 log10cfu/ml. Total bacterial count of cow milk produced in the urbn and peri urban were 5.92±0.73log10 cfu/ml and 6.35±0.67log10 cfu/ml, respectively.
Coliforms can rapidly build up in moist, milky residues in milking equipment, which then becomes the major source of contamination of milk produced. Coliform counts regularly in excess of 150 cfu/ml are considered generally as evidence of unsatisfactory production hygiene. However, relatively low coliform counts in milk don’t necessarily indicate effectively cleaned and disinfected equipment (Ombui et al., 1995).
colony-forming units; TBC= total bacterial count; CC = coliform count, YMC=yeast and mould count.
The overall mean coliform count of milk produced in the study area was 2.72±0.50log10 cfu/ml (Table 2). The coliform count obtained in the current study is less than that reported by Asaminew (2007) and Derese (2008) who reported 4.84 log10 cfu /ml and 4.49log10cfu/ml for cows' milk produced in Bahir Dar milk shed and west shoa zone of Oromia region respectively. The coliform count in the urban and peri urban milk production system are2.56±0.50log10cfu/ml and 2.88±0.46 log10 cfu/ml with an overall mean of 2.72±0.50log10 cfu/ml which shows that higher coliform count was observed in the peri urban milk production system. In general the relatively less number of coliform count found in the current study may be attributed to proper cleaning of the barn, cleaning of udder before milking and cleaning of dairy cows regularly and reduced fecal contamination of the samples either from the cows, the milkers, the containers or the milking environments.
The overall mean yeast and mould count of cows' milk produced in the study area was 0.68±0.41log10 cfu/ml (Table 2). Yeast and mould count in the urban and peri urban milk production systems of the study area were0.52±0.34log10 cfu/ml and 0.84±0.43log10 cfu/ml respectively. In the urban milk production system there was significantly (P<0.05) less number of yeast and mould count compared to the peri urban milk production system.
Generally, the microbial count of total bacterial count, coliform count and yeast and mould count of milk produced in the study district were high.
Table 2: The microbial counts of cows’ milk produced in the study area

SD= Standard deviation NS = not significant (P> 0.05) * = significant (P< 0.05), Sig. = Significance. cfu =
Summary and conclusions
Summary
The mean protein, fat, total solids (TS), solids-not-fat (SNF), ash and lactose contents of local cows’ milk were 3.71%, 4.38%, 13.87%, 9.49% 0.71%, and5.07 while these values were 3.34%, 4.17%, 13.48%, 9.31%, 0.72% and5.26 for crossbred cows milk, respectively.
The overall (± SD) total bacterial count, coliform count and yeast and mould count of milk produced in the urban area were 5.92±0.73log10cfu/ml, 2.56±0.50log10cfu/ml and 0.52±0.34log10cfu/ml while in the peri urban 6.35±0.67log10cfu/ml, 2.88±0.46 log10cfu/ml and 0.84±0.43log10cfu/ml respectively.
Conclusions and recommendations
Milk intended for human consumption must be free from pathogens and must, if conditions permit, contain no or few bacteria. Clean milk could only be obtained if effective sanitary measures are taken starting from the point of milk withdrawn from the cow until it reaches the consumers. High bacterial counts both in urban and peri-urban farms were observed as compared to the recommended international standards. Hence, adequate sanitary measures should be taken at all stages of production chain.
After analyzing all the microbiological data it can be concluded that the fluid milk microbial quality in both urban and peri urban farms of Dangila district was found to be poor compared to the recommended standard and it needs to be improved. Hence, adequate sanitary measures should be taken at all stages from production to consumption. These measures include proper handling of the cow, good personnel hygiene, hygienic milking and processing equipment, improving milk and milk handling environment.
The chemical compositions of milk in both locations were similar and met the acceptable standards. It is therefore recommended that adequate sanitary measures be observed at all stages of production to consumption of the milk from Urban and peri urban areas of Dangila district to protect the milk from spoilage.
High microbial counts both in urban and peri-urban farms were observed as compared to the recommended international standards therefore, further work is needed to improve microbial quality of milk and milk products in the study area.
Acknowledgements
This study was funded by the ministry of education (arba minch university).
References
Alganesh T (2002). Traditional milk & milk products handling practices & raw milk quality in Eastern Wollega. M.Sc. Thesis, Alemaya University. Dire Dawa, Ethiopia. 108p.
Asaminew T (2007). Production, handling, traditional processing practices and quality of milk in Bahir Dar milk shed area, Ethiopia. M. Sc. Thesis, Haramaya University, Ethiopia.
Assaye A (2009). Dangila town administration establishment and development level description bulletin, Dangila, Ethiopia. .
Banda PT (2010). Dairy processing module MAV 411.SADC-university of Zimbabwe regional postgraduate programme in dairy science and technology, Harare, Zimbabwe.Pp 1.
Bekele G and Bayileyegn M (2000). Bacteriological qaulity of raw cow’s milk from four dairy farms and a milk collection center in and around Addis Ababa. Berl. Munch Tierarzti. Wschr. 113:276-278.
Derese T (2008).Present situation of urban and peri-urban milk production and quality of raw milk produced in west Shoa zone, Oromia Region, Ethiopia. M.Sc. Thesis Haramaya University,Ethiopia.
Fayo D (2006). Assessment of Milk Production, Marketing, Feeds and Feeding System of Dairy Cows inand Around Dire Dawa Town. M.Sc.Thesis presented to the School of Graduate Studies of Alemaya University, Ethiopia.
FDA (2008). Code of Federal Regulations: 21 CFR 131.110 Milk and Cream. Available on: http://www.cfsan.fda.gov/~lrd/FCF131.html (accessed Sep 2008)
Lemma F, Fekadu B and PB Hegde (2005a). Rural smallholder milk and milk products production, utilization and marketing systems in three districts of East Shoa Zone of Oromia. In: Proc. Of the 12th Annual Conference of the Ethiopian Society of Animal Production (ESAP) Volume 2: Technical Papers held in Addis Ababa, Ethiopia, August 12-14, 2004. ESAP, Addis Ababa, Ethiopia. Pp.29-37
Lemma F, Fekadu B and PB Hegde (2005b). Traditional milk and milk products handling practices and preservation methods in three districts of East Shoa Zone of Oromia.. In: Proc. Of the 12th Annual Conference of the Ethiopian Society of Animal Production (ESAP) Volume 2: Technical Papers held in Addis Ababa, Ethiopia, August 12- 14, 2004. ESAP, Addis Ababa, Ethiopia. Pp.77-84
Marth EH (ed.) (1978). Standard Methods for the Examinations of Dairy Products. American Public Health Association, Washington, DC. 416p.Microbiology 68:173-186.
Marth EH. and Steele JL (2001). Applied Dairy Microbiology (2nd Edition). Marcel Dekker, Inc. New York, NY.
Mogessie A (1990).Microbiological quality of Ayib, a traditional Ethiopian cottage cheese. International Journal of Food Microbiology. 10 : 263-268.
O’Connor CB (1994). Rural Dairy Technology. ILRI training manual No.1. International Livestock Research Institute (ILRI), Addis Ababa, Ethiopia. 133p.
Ombui JN, Arimi SM, Mcdermott JJ, Mbugua SK, Githua AA and Muthoni J (1995). Quality of raw milk collected and marketed by dairy cooperative societies in Kiambu District, Kenya. Bull. Anim. Hlth. Prod. Afr. 43: 277-284.
Richardson GH,(1985). Standard Methods for the Examination of Dairy Products. 15 th ed. American Public Health Association. Washington, D. C. pp. 168-196.
Van den Berg JCT (1988). Dairy Technology in the Tropics and Subtropics.Pudoc, Wageningen, The Netherlands. 290p.
Zelalem Y and Faye B (2006). Handling and microbial load of cow?s milk and Irgo-fermented milk collected from different shops and producers in central highlands of Ethiopia. Eth. J. Anim. Prod. 6(2):7– 82.






.jpg&w=3840&q=75)





