Introduction
Necrotic enteritis (NE) is a multi-factorial disease, resulting in an important negative impact on the poultry industry due to decreased weight gain, increased mortality, worse feed conversion ratio, greater medication costs and increased risk of contamination of poultry products for human consumption (Timbermont et al., 2011; Paiva and McElroy, 2014).
Clostridium perfringens (C. perfringens) is considered to be the main causative agent of NE in birds (Maier et al., 2000). However, development of NE is a complex process requiring one or several predisposing factors rather than just the presence of pathogenic C. perfringens (Van Immerseel et al., 2009). Any factor that induces stress in animals could be a predisposing factor for increased risk of developing NE, because it could alter the intestinal environment, damage the enterocytes and allow the colonization of the C. perfringens (Park et al., 2008). These predisposing factors mainly include the type of diet, the co-infection with other pathogens particularly coccidia, as well as environmental, nutritional and management factors (Jackson et al., 2003).
Inflammation is an important consequence of NE. Microscopic evaluation of early stages of necrotic enteritis reveals strong inflammatory reactions to C. perfringens. In case of inflammation, the intestinal mucosa is damaged, which decreases nutrient digestion and absorption, reduces growth rate, and increases feed conversion ratio (Lee et al., 2011). However, the impact of inflammation on performance reduction due to NE, may have been underestimated. Restriction on the use of in-feed “growth promoting” antibiotics has led to elevated incidence of NE in poultry production systems (Shojadoost et al., 2012).
Therefore, the development of effective antibiotic alternatives to control NE has become an important & necessary challenge. Antibacterial additives (against C. perfringens) are a logical alternative however, prevention of predisposing intestinal damage is also an important factor for such a multi-factorial disease knowing that proliferation of C. perfringens benefits from epithelial barrier damage and inflammation. Therefore, a combination of different active ingredients acting synergistically with direct effects on pathogenic bacteria, intestinal epithelial layer and inflammation may hold the most promising approach to prevent NE. In our previous in-vitro and in-vivo studies (Khadem et al., PSA 2018), we demonstrated the synergistic anti-inflammatory effects of Lumance® (obtained from Innovad® SA/NV Belgium), a complex blend, combining target-release butyrate, fatty acids, plant extracts and essential oils.
This study a) evaluated the in-vitro antibacterial activity of Lumance® and b) correlated the findings with a large-scale in-vivo trial that evaluated the ability of Lumance® to replace three commercial non-antibiotic feed additives used to control NE and maintain performance in the absence of antibiotic growth promoters.
Materials and methods
Evaluation of the in-vitro anti-bacterial activity of Lumance® against C. perfringens
The antibacterial activities of Lumance® against C. perfringens isolated from the intestine of broilers were studied in-vitro. Lumance® was added at different concentrations (ranging from 400 to 2000 mg/kg) to a medium inoculated with C. perfringens. The antimicrobial susceptibility testing was performed using the broth microdilution assay based on the ISO 10932/IDF 223 standard protocol. Bacterial growth was monitored by measuring the optical Density (OD) at 580 nm.
Evaluation of the in-vivo growth promotion and control of necrotic enteritis
This in-vivo large-scale trial (7.3 million broilers) was carried out from February 2018 to April 2018 in an antibiotic free broilers program in North America. The USA broiler complex had been using a combination of three feed additives. Namely, a calcium butyrate (Kemin®), a probiotic (Novus®) and a natural anticoccidial (Phibro®) product to prevent necrotic enteritis and improve performance. These three-feed additives were replaced with a single approach, Lumance® (Innovad® SA/NV Belgium).
Results and discussion
Evaluation of the in-vitro anti-bacterial activity of Lumance®
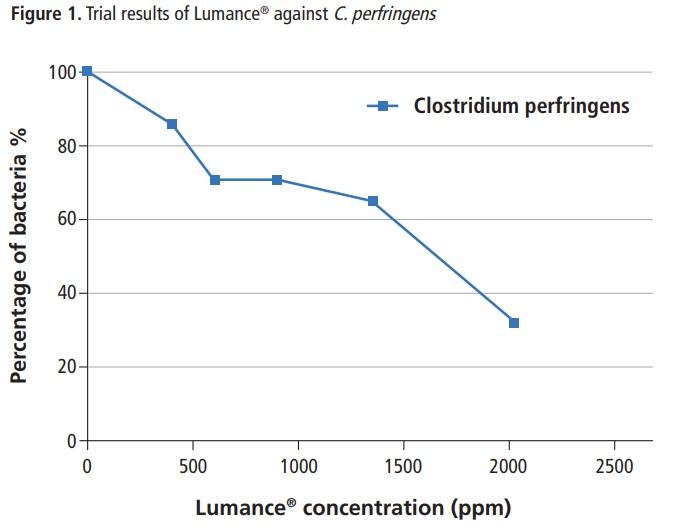
Lumance® effectively inhibited the in-vitro growth of C. perfringens in a dose-response manner (Figure 1). The mechanism of this beneficial effect was attributed to the ability of the active ingredients (target-release butyrate, fatty acids) of the product to passively pass across the cell membrane of the cells, dissociate in the more alkaline interior, acidify the cell cytoplasm, dissipate the proton motive force and cause cell lysis. The results showed that use of the product at 2000 ppm reduced pathogenic C. perfringens by 70%. The data suggest that such dose may have a beneficial impact on reduction of pathogenic C. perfringens in field conditions. In conclusion, the in-vitro susceptibility data obtained in this study provides support that Lumance® could be an effective treatment for prevention of NE in animals.
Evaluation of in-vivo growth promotion and necrotic enteritis control
No signs of necrotic enteritis were found in the Lumance® group, whereas in other group necrotic enteritis was seen.
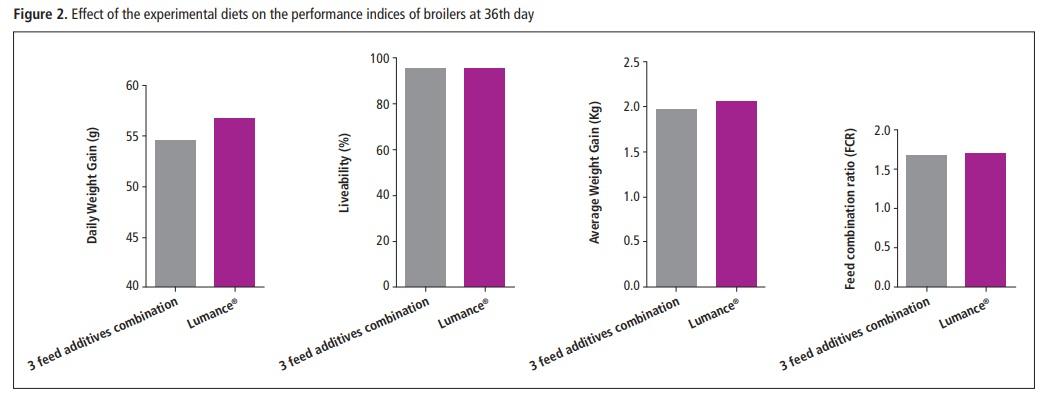
Lumance® exhibited a trend of improving Daily Weight Gain against the combination of the three feed additives although not statistically significant (DWG 54.5 vs. 56.7 respectively). No differences were found between the combination of the three feed additives and the Lumance® groups in relation to average weight gain (AWG), feed conversion ratio (FCR), and liveability % (Liv) (AWG:1.96 vs. 2.06; FCR: 1.68 vs.1.69; Liv: 95.81 vs. 95.93; respectively).
Lumance® successfully controlled necrotic enteritis and replaced effectively all other additives, without the use of medication and growth promoters with a substantial cost saving potential.
Lumance® demonstrated a clear ability as for use in starter, grower and finisher diets by ensuring top performance, beneficial feed conversion and controlled necrotic enteritis in an economical way. However, to further minimize necrotic enteritis (NE) issues, maintaining moisture levels of the poultry house litter within the recommended range is necessary. To achieve this, poultry producers must ensure that high quality feed is provided to the flock, disease pathogens are not permitted to enter the premises, and adequate ventilation systems with quality bedding material are used.
Conclusion
In conclusion, this research shows that Lumance® can improve the health status of the bird hindgut and have a beneficial impact on the prevention of NE in broilers by a) reducing subclinical infections via reduction of C. perfringens levels, and b) by reducing the risk of wet litter situations (diarrhea).
This combined with previous research on the antiinflammatory action of Lumance® (Khadem et al., PSA 2018) demonstrates the potential of Lumance® to regulate important factors related to disease (not only those contributing to NE) and consequently, improve the growth performance of broilers, in an antibiotic-free and economical way
Lumance® successfully controlled necrotic enteritis and replacedeffectively all other additives, without the use of medication andgrowth promoters with a substantial cost saving potential.















.jpg&w=3840&q=75)