I. INTRODUCTION
Human consumption of chicken meat products has risen exponentially over the past five decades and continues to grow on a global scale. Thus chicken meat production has seen unparalleled expansion, to meet the continually increasing consumer demand (Allievi et al., 2015). Such expansion has resulted in the chicken meat industry being at the forefront of animal production, where advances in animal nutrition and genetics are near optimal. Therefore, producers are continuously looking for new and innovative methods to enhance chicken meat performance, with the maternal environment providing an economically viable method to do so. The maternal environment can be described as the overall environment a female organism encounters at the time of reproduction. Several known factors can influence the maternal environment, including maternal stress, nutrition, geographical location and individual health, ultimately altering progeny development, with permanent phenotypic effects (Reynolds et al., 2010). Previous work has shown that exposure to chronic maternal stress can negatively influence progeny performance in production animals (Reynolds et al., 2010). This is a key finding for the chicken meat industry for two primary reasons. Firstly, the chicken meat breeder industry utilises feed restriction measures in their breeder flocks. Although implemented to enhance reproductive outputs in breeder hens, an increasing body of work suggests the use of feed restriction measures induces chronic stress in breeder hens, prompted by extended periods of prolonged hunger (Zulkifli et al., 2015). Secondly, commercial chicken meat birds now spend ~40% of their life within the in-ovo environment and recent findings have clearly exhibited the effects of the in-ovo environment on embryonic development in the chicken (Ho et al., 2011). Although there is strong evidence that suggests the maternal environment does impact offspring performance in chicken meat birds, considerable uncertainty still remains as to the precise effects environmental variations have on progeny performance. Therefore, the aim of this study was to utilise an in-ovo model to investigate how early-life exposure to stress may influence performance characteristics in subsequent offspring. Additionally, dietary Arg was supplemented as a means of alleviating the negative consequences of in-ovo exposure to CORT. Arg has been documented to enhance protein synthesis, whilst promoting the secretion of endocrine factors such as the thyroid hormones and growth hormone, both of which are involved in growth and metabolic pathways. Thus, supplementary Arg is hypothesised to alleviate, to some extent, the phenotypic consequences derived from in-ovo exposure to CORT.
II. METHODOLOGY
400 eggs collected from a commercial Cobb 500 broiler breeder flock were separated into two groups, with 200 eggs receiving 1 μg of corticosterone (CORT) dissolved in absolute ethanol and the remaining 200 eggs receiving a control (CON) solution. Solutions were injected into the chorioallontoic membrane at embryonic day 11. At hatch 112 CON and 100 CORT birds were weighed, then separated into four treatment groups, (1) CORT-Control, (2) CORT-Arg, (3) CON-Arg, (4) CON-Control. Birds were provided with ab libitum access to both feed and water. Birds fed an Arg supplemented diet received a standard chicken meat diet + 125% Arg:Lys ratio. Individual bird body weights were recorded weekly, along with pen total feed conversion ratio. A sub-sample of birds, three per treatment (n=12) were humanely killed at day 35 and subjected to a dual-energy x-ray absorptiometry scan (DEXA) to determine total body composition. Analysis of experimental data was performed by linear mixed model analysis following the procedures of IBM®, SPSS® Statistics 25 program (Armonk, NY, USA). The data were checked for normality by the Shapiro–Wilk test. Non-normalised data was analysed using nonparametric tests including Mann-Whitney U and Kruskal-Wallis. A probability level of less than 5% (P < 0.05) was deemed as statistically significant.
III. RESULTS
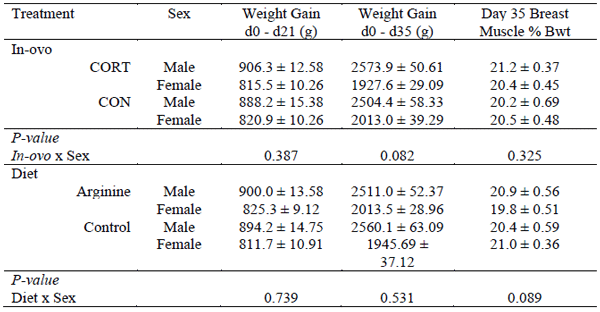
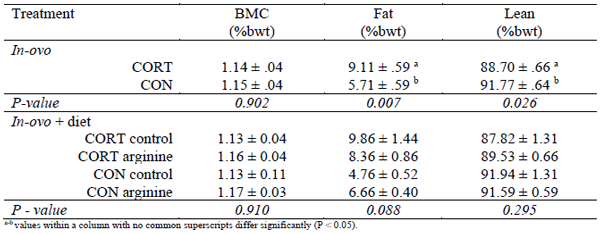
Weight gain between day 0 and 21 did not differ between CORT or CON treated birds, nor were there any sex dependent effects in relation to in-ovo treatment (P > 0.05) (Table 1). Total weight gain from day 0 to 35 did not differ between in-ovo treatments; however a potential sex dependent trend appeared (P > 0.05). CORT treated male birds recorded superior weight gain at day 35 compared to CON treated birds. Conversely, CORT treated females recorded lower total weight gain at day 35 than CON treated birds. Additionally, CORT treated males tended to exhibit enlarged breast muscle mass (%bwt), whilst no notable variation was identified in female birds relating to breast muscle mass. Furthermore, FCR was not influenced by in-ovo, nor dietary treatment independently, whilst no interaction was detected between in-ovo and dietary treatments. Female birds fed an Arg supplemented diet tended to exhibit reduced total breast muscle yield (%bwt), whilst no interaction was observed in male birds in relation to diet (P > 0.05). Dietary supplementation with Arg did not significantly influence weight gain at any age (P > 0.05). Female birds exposed to CORT in-ovo exhibited greater fat mass (%bwt) and reduced total lean mass (%bwt) at 35 days of age. Conversely, CON treated female birds displayed enhanced total lean mass (%bwt) and reduced total fat mass (% bwt) (Table 2). Supplementation of Arg into the diet did not influence total bird body composition at 35 days of age, although Arg supplementation tended to enhance lean mass and reduce fat mass in CORT treated birds.
Table 1 - Effects of in-ovo corticosterone exposure and dietary arginine supplementation on sex dependent performance characteristics in broiler chickens from 0 – 35 days post hatch. Values are average mean ± SEM.
Table 2 - Day 35 body composition of female chicken meat birds subjected to in-ovo CORT or CON treatment as well as birds fed an arginine supplemented diet or control diet. Values are average mean (%bwt) ± SEM.
IV. DISCUSSION
Although the body of work supporting feed restriction induced chronic stress in breeder hens is relatively strong, the transgenerational effects exposure to maternal stress has on subsequent performance characteristics in progeny remains unclear. The present study utilised an in-ovo model to investigate the potential phenotypic consequences invoked via in-ovo exposure to a stressor in chicken meat birds post-hatch. Previous work by Hynd et al. (2016) reported a reduction in male progeny weight at 42 days of age in birds produced from hens subjected to feed restriction measures. These findings corresponded with feed restricted hens exhibiting elevated plasma corticosterone concentrations and heterophil/lymphocyte ratios, indicating elevated levels of stress, which had been previously reported (Zulkifli et al., 2015).
Interestingly, the results from the current study suggests exposure to maternal stress may act in a sex dependent manner. However, it must be noted that significant variability exists within the literature regarding exposure to maternal stress and its ability to disrupt offspring development. Several avian species have been exposed to maternal stress under experimental conditions, with vastly different results reported between species, as well as within species (Ahmed et al., 2016, Hayward and Wingfield, 2004). Furthermore, supplementing diets with additional Arg did not influence weight gain. Arg has been documented to promote protein synthesis as well as influence growth and metabolic pathways associated with the thyroid hormones (Ebrahimi et al., 2014) and various other growth factors in birds. However, the chickens utilised in the current study were great grandparent birds, where performance variation is greater in birds positioned higher up the breeding pedigree. The use of such birds may have therefore unintentionally ‘masked’ any offspring phenotypic variation influenced by the dietary and in-ovo treatments administered. Thus, phenotypic variation may occur in separate strains further down the breeding pedigree, as has been reported within the literature.
The findings that female birds exposed to CORT exhibited enhanced total fat mass and reduced total lean mass compared to CON birds is somewhat novel. Such variation may eventuate as a consequence of alterations to the hormonal composition of the egg, disrupting physiological processes that influence the number of myofibres developed by the embryo, which is determined embryonically (Smith, 1963). Although previous studies reported that exposure to maternal stress could ‘influence’ weight gain in numerous avian species, whether such variation was attributed to muscle, fat or bone mass remained elusive. Additionally, supplementing CORT exposed birds with Arg tended to reduce the phenotypic consequences associated with early-life exposure to CORT. Although male total body composition was not measured due to insufficient numbers, these findings in female birds still suggest that the variations to the maternal environment may promote undesirable carcass characteristics in chicken meat birds. However, targeting the maternal environment provides a novel approach to improve total flock uniformity and subsequent carcass characteristics, albeit in a sex-dependent manner. Thus, future studies investigating the maternal environment and its ability to alter offspring development must incorporate total carcass composition along with weight gain and FCR, with specific interest in the sex-dependent variations that predominantly occur.
ACKNOWLEDGEMENTS: The authors would like to thank Feedworks Pty. Ltd., the University of Sydney and the South Australian Research and Development Institute for their contributions to this project. Additionally, many thanks are extended to Associate Professor Kapil Chousalkar & Dr. Chris Schultz for their efforts in supporting this project.
Presented at the 30th Annual Australian Poultry Science Symposium 2020. For information on the next edition, click here.










.jpg&w=3840&q=75)