Relationships between immunity and nutrition in birds
Published: January 4, 2018
By: K. C. Klasing
Department of Animal Science, University of California, Davis, CA, USA
The quantitative investment in immune defenses is thought to be under tight evolutionary control because they must be sufficient to thwart pathogens without excessively consuming resources (e.g. nutrients and energy) needed for other important processes or causing immunopathology (Ardia et al., 2010, Schmid-Hempel, 2011). In poultry production there is concern that the immune system competes for nutrients with tissue growth or egg production. The practical implications are twofold. First, unnecessary or overly robust immune responses may diminish the rate and efficiency of production. Second, that intensive genetic selection of poultry for efficient growth or egg production for many decades may have diminished the immune system and consequently reduced disease resistance.
Overview of the dynamics of an immune response
Individual components of the immune system respond to an infectious challenge at very different rates. Innate immune cells respond quickly to a challenge due to the presence of a common set of receptors on all phagocytic cells (e.g. macrophages and neutrophils) that recognize broad categories of pathogens (Medzhitov, 2001). Thus, a very large number of cells can recognize invading microbes and respond to them quickly. Conversely, the lymphocytes that mediate adaptive immunity have receptors that are narrowly tuned to a specific antigen and a diverse population of lymphocytes exists in order to identify a very large number of antigens (Bonilla and Oettgen, 2010). Because the initial population of lymphocytes that possess the appropriate receptor for a given pathogen is very small, this subset of lymphocytes must proliferate for several days to reach protective numbers. These responding lymphocytes transition from the least metabolically active cells in the body to some of the most active in order to support their rapid replication and copious secretion of effector molecules such as immunoglobulins.
The largest source of protective proteins during an immune response is hepatocytes. During the first day of an immune response to a pathogen challenge the liver transitions from maintaining homeostasis and supporting the nutritional demands of growth or reproduction to the production of proteins such as complement, mannan binding protein, and C-reactive protein that aid in the detection and neutralization of pathogens. During the acute phase response against a successful pathogen, the liver becomes the most important organ of the immune system when using nutritional demands as the metric. By five to seven days of a typical immune response the production of lymphocytes and immunoglobulin become quantitatively greater than the production of acute phase proteins. Overall the innate and adaptive immune systems work mutually to provide an immediate response to infection via innate processes, while slowly developing a specific response that is mediated by lymphocytes. This temporal division serves to spread the nutritional costs of a response over a longer period of time but a global accounting of the innate and adaptive responses is needed to determine if there are nutritionally important implications.
Size and nutrient content of the immune system.
Nutritionist have rigorously applied quantitative theory and modeling to nutrient needs for growth and reproduction as influenced by dietary and environmental factors. However, nutritionists have generally been remiss in applying robust quantitative tools to tradeoffs between performance and immunity. We have endeavored to make quantitative estimates of the size of these tradeoffs as well as each of the underlying processes that siphon nutrients away from growth and reproduction. To do this we have assessed the amount of nutrients needed for mounting an immune response using both direct and indirect estimates.
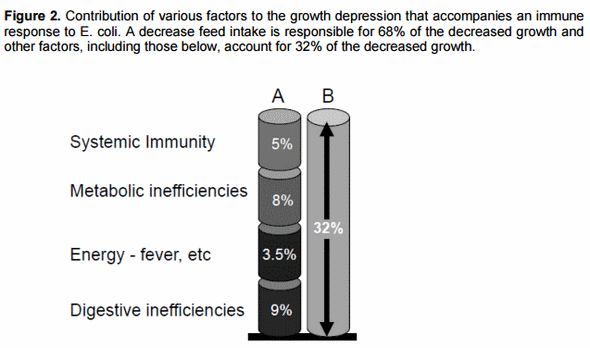
Indirect estimates were made by quantifying the magnitude of growth depression that occurs during the periods of time that growing broiler chicks mount an initial innate response and also a subsequent adaptive immune response. We estimate that a robust acute phase immune response against a simulated infection with dead Escherichia coli decreases growth by about 25% but there is no decrease in growth during the subsequent adaptive response. About two-thirds of the growth depression during the acute phase response is due to a decrease in appetite and about a third is due to nutrient diversions or losses related to the immune response.
Direct estimates were made by quantifying the whole body dynamics and nutrient content of the myriad of cells and proteins responsible for protective immunity during the innate and adaptive responses to a simulated infection with E. coli (Iseri and Klasing, 2013; 2014). Although energy expenditure or any one of the dozens of dietary essential nutrients might be used as a metric for nutritional expenditures by the immune system relative to other tissues, the essential amino acid lysine was initially used as a reference nutrient. This is because lysine is the reference amino acid in the ideal protein system used commonly in non-ruminant nutrition because it functions almost exclusively as a substrate for protein synthesis and cannot be stored or synthesized.
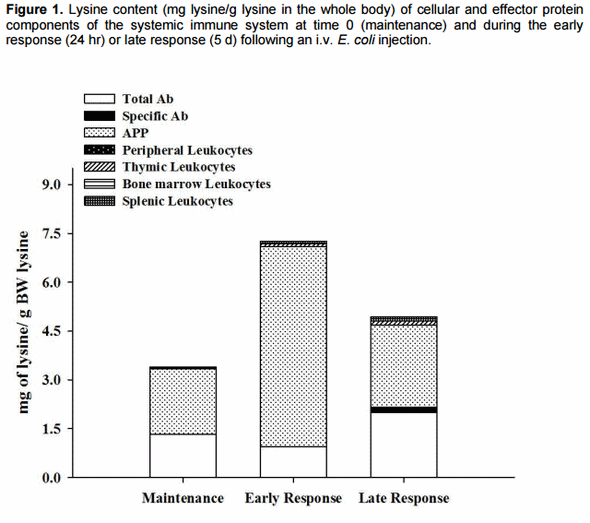
The studies by Iseri and Klasing examined the amount of lysine in 6 different leukocyte types in 5 different tissues (blood, spleen, bursa, thymus, bone marrow) and 12 protective protein/immunoglobulin pools, all at several time points. The immune system has both systemic and mucosal components; however, we limited this investigation to the systemic system due to the extreme difficulty of quantifying the diffusely organized mucosal immune system. A summary of the data is shown in Figure 1 and indicates that the amount of lysine in protective proteins, such as the acute phase proteins and immunoglobulins, greatly exceed that in the cellular component of the immune system, regardless of whether the immune system is responding or not. During the acute phase of the immune response the liver hypertrophies markedly for the rapid production of acute phase proteins. Because the liver is recruited to become part of the immune defenses during the acute phase response, it is the most expensive part of the response. The amount of lysine needed for the adaptive phase of the response (antibody production and new lymphocytes) is much less than that needed for the acute phase of the response and is incurred following the acute phase response (i.e. after 3 days). During the transition from the acute phase response to the time when the adaptive response begins to utilize significant quantities of lysine, the size of the liver and levels of protective proteins return toward normal. The lysine liberated from protein catabolism of hepatic tissue and acute phase proteins would provide a surplus of lysine to provision the anabolic processes of the adaptive response.
The amount of lysine consumed by the immune system during a robust response accounts for only a five per cent decrease in growth, which is not sufficient to account for the 25% decrease that is observed during the response. This means that the cost of an immune response is mostly due to protective processes and physiological adjustments that are unrelated to the needs of leukocytes or the production of protective proteins. Even when the hypertrophy of the liver and the massive production of acute phase proteins are included, the amount of nutrients diverted to protective processes accounts for very little of the depression in growth or reproduction that occurs during the response.
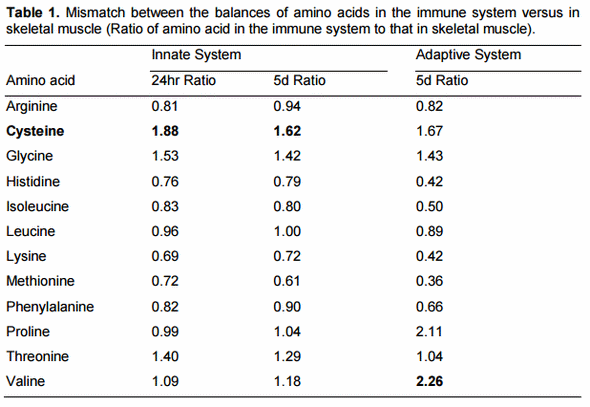
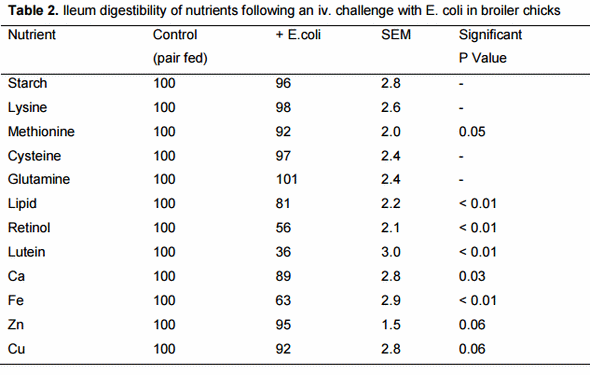
More recently we have examined the ideal balance of amino acids for the immune response to a pathogen and found that lysine needs are lower for immunity relative to growth or egg production and use of sulfur amino acids, especially cysteine, gives a better estimate (Table 1). This large difference in the balance of amino acids needed for the immune response relative to accretion of body tissue or egg protein greatly increases the protein cost of an immune response. Ongoing research indicates that fever, decreased intake of food, and less efficient digestion that accompanies a robust immune response are, together, more important than the diversion of nutritional resources to the immune system (Figure 2). Quantitatively, a decrease in digestion of nutrients, especially fat and some amino acids (Table 2) is the most important physiological change when the nutritional impact is used as a metric.
Appropriation of nutrients when the immune system responds
In the initial stages of an immune response against a novel pathogen, phagocytes are the early responders and release pro-inflammatory cytokines in sufficient amounts that they have endocrine-like effects throughout the body. This cytokine storm induces metabolic changes, including increased protein degradation and insulin resistance, which divert nutrients from skeletal muscle and other tissues so that they become available for the increased demands of the liver and responding leukocytes (Sirimongkolkasem, 2007). In the case of amino acids, the balance of essential and semi-essential amino acids is very different in leukocytes, protective proteins and the hypertrophying liver compared to the balance in muscle and other tissues. Recent work indicates that cysteine is the most limiting amino acid during the acute phase response in chickens (Table 1. Iseri and Klasing, 2013; Sirimongkolkasem, 2007) and also in rats (Breuille et al., 2006; Breuille and Obled, 2000). This is due to a mismatch between muscle cysteine release and hepatic demand for the markedly enhanced production of acute phase proteins and glutathione, which serves as an antioxidant.
Presented at CLANA 2016 in Cancun, Mexico.
References
Ardia, D., Parmentier, H.K., Vogel, L., 2010. The role of constraints and limitation in driving individual variation in immune response. Functional Ecology 25, 61-73.
Breuille, D., Bechereau, F., Buffiere, C., Denis, P., Pouyet, C.and Obled, C. 2006. Beneficial effect of amino acid supplementation, especially cysteine, on body nitrogen economy in septic rats. Clin. Nutr., 25, 634-642.
Breuille, D.and Obled, C. 2000. Cysteine and glutathione in catabolic states. Nestle Nutrition Workshop Series. , 3, 173-191.
Iseri, V., and K. Klasing. 2013. Dynamics of the systemic components of the chicken (Gallus gallus domesticus) immune system following activation by Escherichia coli; implications for the costs of immunity. Developmental & Comparative Immunology 40: 248-257.
Iseri, V., and K. Klasing. 2014. Changes in the Amount of Lysine in Protective Proteins and Immune Cells after a Systemic Response to Dead Escherichia coli: Implications for the Nutritional Costs of Immunity. Integrative and Comparative Biology 54: 922-930.
Medzhitov, R., 2001. Toll-like receptors and innate immunity. Nat Rev Immunol 1, 135-145. Schmid-Hempel, 2011. Ecological Immunology, Evolutionary Parasitology. Oxford University Press, Oxford, pp. 98-140.
Sirimongkolkasem, P. 2007. Amino acid partitioning during the acute phase response. . Ph.D Thesis, UC Davis, Davis, CA.
Related topics:
Authors:
UC Davis - University of California
Influencers who recommended :
Jose Mauro Arrieta Acevedo, Mostafa Abd ElrahimRecommend
Comment
Share
11 de enero de 2018
Many thanks, Professor Kirk, for this interesting and sensitive discussion. As a matter of fact, immunity and nutrition are closely related and inseparable.Healthy birds exhibit superb immune status against various diseases/pathogens by eating adequately to maintain and meet their nutritional/productive requirements. On the other hand, poor feeds or feeds that are deficient in essential nutrients are capable of jeopardizing the immune capabilities or abilities of apparently healthy birds to resisting dormant or invading pathogens.
Recommend
Reply
10 de enero de 2018
The relationship between immune system and nutrition cannot be over emphasized and cannot be separated. This is because the immune system is the defense engine while nutrition is needed to support or activate it, so that, it can be very effective to fight disease for birds.
Recommend
Reply

Would you like to discuss another topic? Create a new post to engage with experts in the community.










.jpg&w=3840&q=75)