Introduction
Feed management is a key factor affecting water quality (Boyd and Tucker 1998, Jory 1995) and production economics in aquaculture (Jolly and Clonts 1993). Not only is the feed the initial 0source of physiological wastes, but it accounts for 55% to 60% of the operation costs in intensive systems and around 40% in semi-intensive systems (Chanratchakool et al. 1994, Lovell 1998). Survival and growth have the greatest impact on the economic performance of shrimp production, and correct feeding is essential for both (Wyban et al. 1989). Feed management strategies should therefore be aimed at optimizing feed inputs, reducing feed conversion ratios and reducing the potential impact on the culture and effluent water (Jory et al. 2001). In recent years, the intensity of shrimp production systems has increased, resulting in higher stocking densities and greater feed inputs, which in turn commonly result in a high FCR (Peterson 1999, Peterson and Walker 2002). Quite often, failures are blamed on PL quality, feed, water quality and /or disease; although in most cases the origin of the problem is poor feed management (Cruz 1991, Piedad-Pascual 1993).
Environmental, social and economic considerations drive the need to improve feed management and feed formulations. Shrimp pond water can impact the environment because only a portion of the nutrients in the feed are consumed, assimilated and retained as tissue growth. Briggs and Funge-Smith (1994) indicated that 24% of the nitrogen and 13% of the phosphorus from the feed was retained by the shrimp. Similarly, Green et al. (1997) reported 37% of the applied nitrogen and 20% of the phosphorus was retained by the shrimp. The subsequent nutrient loading on the culture environment results in costs associate with aeration, water exchange and environmental impacts. A large unused fraction remains in the water column or as settled solids. The unutilized nitrogen and phosphorus promotes eutrophication in the pond system as well as in neighboring waters if effluents are released from the pond system (Boyd and Musig 1992, Dierberg and Kiattisimkul 1996, Boyd and Tucker 1998). Negative effects are not isolated but promote diseases and other water quality related problems, which affect production (Goddard 1996, Boyd and Tucker 1998, Jory et al. 2001, Samocha et al. 2001). In terms of social issues, there is considerable concern with regards to the use of industrial fish meals to produce seafood products. Commercial shrimp feed typically contain fish meal at 25% to 50% of the total diet (Dersjant-Li, 2002; Tacon and Barg, 1998). The high fish meal content of feeds as well as typical industrial feed conversion ratios of over 2:1 (authors estimate) make shrimp feed a primary consumer of fish meal resources. The poor feed conversion, reliance on expensive high fish meal diets in combination with decreasing prices for shrimp, have resulted in considerable economic and social pressures on shrimp farming.
If shrimp farmers are to continue to run a profitable business they must improve economic efficiencies as well as their image as a sustainable industry. Improvements in feed management will go a long way in reducing the reliance on fish meal (less feed means less fish meal used to produce a pound of shrimp), reduce environmental impacts (reduced feed inputs, result in lower nutrient inputs and reduced requirement for aeration or water exchange) and improved economics as the investments in feed per unit of shrimp produced are improved. Unfortunately, feed management is a poorly understood “art” because numerous factors influence not only growth but also the size of the population which is quite difficult to determine. Because of the difficulties in determining biomass, a poor understanding of daily nutrient requirements, as well as the desire of farmers to obtain maximum growth they often overfeed and have relatively por FCR. Consequently, there is a need to re-assess current feeding protocols and develop new feeding strategies.
There are a variety of methods used to estimate feed inputs. Irrespective of the methods employed, all feeding protocols should at least include the following considerations. First, feed utilization should be planned and daily feed inputs should be limited. Daily inputs must be reasonable and should consider the growth of the shrimp as well as nutrient recycling capacity of the pond ecosystem. Growth is easily estimated based on weekly sampling of the shrimp. However, feed inputs are often overestimated due to unrealistic expectations of future shrimp growth as well as survival. Placing upper limits on daily feed inputs based on historical growth rates helps limit expectations. Daily feed inputs should not exceed the ability of the pond to assimilate byproducts and to maintain a sufficient level of dissolved oxygen. When feeding rates increase beyond the natural carrying capacity of the pond, water quality deteriorates (Cole and Boyd 1986). Low dissolved oxygen levels are usually the first indication that the carrying capacity has been exceeded (Hopkins et al. 1992). Although carrying capacity will vary with local environmental factors, a number of authors have presented guidelines. In ponds without aeration, suitable feeding rates have been found to be around 25-30 kg/ha/day (Tucker et al. 1979). With nighttime mechanical aeration, feeding rates can be sustained in a range up to 100- 120 kg/ha/day (Boyd 1989, Boyd and Tucker 1998). Even if adequate aeration is provided to prevent dissolved oxygen depletion, other limiting factors such as high levels of ammonia may limit feed input and production (Boyd and Tucker, 1998). Water exchange has been used to minimize water quality problems encountered during shrimp production (Clifford 1992; Chien 1992; Hopkins et al. 1993). This practice should be minimized since exchanging water is a very inefficient method of managing water quality and pond effluents can have negative impact on receiving waters and will eventually inhibit production (Pruder 1992; Ziemman et al. 1992; Boyd and Musig 1992; Hopkins et al. 1995; Teichert-Coddington 1995; Teichert-Coddington et al. 1999; Boyd 2000; Lawrence et al. 2001).
The second consideration should be the number of feedings and feed distribution. Multiple daily feedings are desirable because shrimp eat slowly and almost continuously (Lovett and Felder 1990). Disintegration of the feed particles and a loss of nutrients such as water soluble vitamins, free amino acids, some minerals and attractants can be expected when feed pellets are exposed to water (Goddard 1996). Increased feeding frequency reduces nutrient leaching and improves feed utilization efficiency (Wyban and Sweeney 1989; Villalon 1991; Robertson et al. 1993; Velasco et al. 1999; Nunes and Parsons 2000). Although increasing the number of feedings is helpful, this increases labor costs and there is a limit to the number of feedings that are practical. Wyban and Sweeney (1989) reported that six feedings per day did not improve growth hence, moderate feed inputs of 2-4 times /day (depending on the size of the shrimp) is often recommended.
Environmental aspects should also be considered when determining daily feed inputs. Water temperatures and dissolved oxygen levels are primary factors influence feeding activity, metabolism and growth, and thus influence both the types and quantities of feeds used (Goddard 1996). Low light levels due to cloudy conditions limit algae photosynthetic activity, hence lowering DO concentrations. Warm, sunny, or calm days may cause algal blooms and crashes, pond stratification, and high temperatures that affect shrimp feeding activity. Consequently, weather conditions should be taken into account and daily rations reduced or eliminated in anticipation of water quality problems. Bottom soil conditions influence water quality and aquatic production in ponds (Hajek and Boyd 1994; Boyd 1995). Feed input is the major cause of deterioration in pond bottom soils. Feces and unconsumed feed pellets increase the amount of organic material on the bottom. When excessive organic matter accumulates in the interstices between soil particles, it inhibits oxygen diffusion into the soil and generates anaerobic conditions (Boyd 1995, Lin 1995). Anaerobic soils lead to high concentrations of metabolites such as NH3, NO2 and H2S (Boyd, 1992; Fast and Boyd 1992; Hopkins et al. 1994.), which are toxic to shrimp (Boyd 1995; Muir and Owens 1997). Sulfide oxidation demands higher dissolved oxygen, which exacerbates the problem. Even if anaerobic conditions do not extend far away from the soil surface, they can cause problems if the soil-water boundary is disturbed. Prevention of problems requires that the overlying water be maintained at 70% saturation (Ritvo et al. 2000). Deteriorated pond bottom conditions can be minimized by conducting periodic assessments to detect the build up of uneaten feeds, and then responding to excess build up with appropriate pond management.
Natural food also plays an important role in shrimp culture as it provides an important yet variable food source. In properly prepared ponds, post-larvae utilize mostly natural foods during the first few weeks after stocking and therefore may not require additional feed (Chanratchakool et al. 1994). Penaeid shrimp cultured in extensive aquaculture ponds, fed almost totally on natural pond biota, have yielded several hundred kg/ha (Pruder 1987). Moss et al. (1992) reported that shrimp in these systems consume detrital aggregates, microalgae, nematodes, copepods, amphipods, polychaetes, moluscs, and other invertebrates. In more intensive ponds, with yields from several hundred to several thousands kg/ha annually, the contribution of natural pond biota to shrimp growth is still important, even though significant quantities of pelleted feed may be provided (Rubright et al.1981). Anderson et al. (1987) demonstrated that 53-77% of L. vannamei growth in semi-intensive systems (20/m2 in cages) was derived from natural food organisms, while formulated feeds accounted for the remaining 30-40%. Bianchi et al. (1990) demonstrated that 70-80% of the weight gain of L. vannamei reared under laboratory conditions came from consumption of bacterial flock. Even under grow-out conditions, shrimp growth is enhanced by unknown growth factors produced autochthonously in the intensive shrimp pond (Leber and Pruder 1988; Moss et al. 1992; Moss 1995).
Even under the best of circumstances, feed management for commercial shrimp ponds is a complex problem. Feed inputs, have a direct effect on nutrient loading of the pond, growth performance of the shrimp and economic returns on production. Under-feeding generally results in good utilization of the feed and low nutrient loading of the system but produces poor growth of the shrimp and lower yields. Overfeeding results in increased nutrient loading of the culture system and maximum growth rates, only if water quality is not inhibited. If overfeeding is excessive, this can result in reduced yield due to deterioration of water and pond bottom quality. Despite the importance of feed management, there are few studies that have been conducted using replicated ponds and standardized conditions. The following case study describes systematic improvement in feed management that we have employed over the last 6 years.
Production Trials and Results
Over the last six years (2000 - 2005) we have carried out a variety of studies aimed to evaluate marine shrimp production in semi-intensive ponds conditions. During this time we have utilized a number of feeding strategies and protocols. Methodologies and results from these studies will be summarized and discussed emphasizing the specifics of feed management for each study.
General Management
All production trials described in this paper were conducted at the Claude Peteet Mariculture Center using high health shrimp obtained from commercial hatcheries, reared in a nursery system for about three weeks and then stocked into production ponds at 35 shrimp/m2. Ponds used for the grow-out phase were 0.1 ha in surface area (46 x 20 m) (35,000 shrimp/pond), with a 1.0 m average depth and lined with a 1.52 mm thick high-density polyethylene. Each pond bottom was covered with a 25-cm deep layer of sandy-loam soil and equipped with a 20-cm diameter screened standpipe and a concrete catch basin. Prior to use, pond soils were dried and tilled to allow oxidation and mineralization of organic matter. Two to four weeks before stocking, ponds were filled with brackish water from the Intracostal Canal between Mobile and Perdido Bay. Unless specified, the shrimp were fed twice a day with commercially produced high quality shrimp feeds containing 35% protein.
During each production trial, dissolved oxygen, temperature, salinity and pH values were measured after sunrise (5:00-5:30 h) and at night (20:00-22:00 h). Afternoon parameters were also monitored in order to determine extreme DO and temperature conditions. Total ammonianitrogen (TAN) and Sechii disk readings were determined on a weekly basis. In order to maintain minimum dissolved oxygen levels of 2.5 mg/L, each pond was provided with a base aeration capacity of 10hp/ha. Spiral paddle wheel aerators of 1-hp (7.5kW/ha) (Little John Aerator, Southern Machine Welding Inc. Quinton, AL) or propeller aspirators aerators of 1-hp (11.2 Ampers) or 2-hp (20 Ampers) (Aire-O2, Aeration Industries International, Inc. Minneapolis, Minnesota) were used for this purpose. When required, additional aeration (up to 30 hp/ha) was used to maintain adequate DO levels. Ponds were managed with a minimal wáter Exchange strategy, therefore, there were no regular water exchanges. However, because of heavy rains associated with storms, the water level of the ponds was reduced as necessary, to avoid the risk of flooding. Additionally, the week prior to harvest, ~30% of the water from the ponds was replaced to reduce the chance of algae crashes and to encourage the shrimp to molting.
Year 2000a. Effects of Nursery on the Growth and Survival of Litopenaeus Vannamei in Culture Ponds. (Garza et al., 2004)
This study (Garza et al., 2004) evaluated the effects nursery duration (Direct Stocking (DS), 10 days (N10) and 20 days (N20)), on the survival and production of L. vannamei during subsequent pond culture. Feed inputs for this study were calculated using a fixed FCR of 1.6, an expected weekly growth based on the average weight gain of shrimp during the previous two weeks, and an assumed survival of 70% for the DS treatment and 85% for the N10 and N20 treatments. As an example of this strategy, a pond with an estimated population of 30,000 shrimp at a given week, with an average weight gain of 1 gram/wk during the previous two weeks and an expected feed conversion of 1.6:1, would require a total feed input of 6.85 kg/day for the subsequent week (30,000 shrimp x 1g per wk x 1.6 / 7 days).
Direct stocked shrimp were fed at a rate of 10 kg/ha/day for the first ten days and at a rate of 20 kg/ha/day for the next ten days. After 20 days from stocking in the ponds, feeding was based on estimated biomass. After ten days of nursing, the second group of shrimp (N10) were stocked into the ponds and feed was offered at a rate of 20 kg/ha/day for the first ten days, after which they were fed based on estimated biomass. N20 shrimp were fed based on estimated biomass as soon as they were stocked in the ponds.
Results at harvest for the grow-out of the DS, N10, and N20 treatments were: 15.01, 13.90 and 12.77 g/shrimp for mean weight; 67, 77, and 79 % for survival and 3701, 3934, and 3709 kg/ha for mean yields, respectively. Mean growth per week was 0.94, 0.87, and 0.80 g/week and mean FCR's were 1.97, 2.03, and 2.12, respectively. No statistical differences were found in survivals, average weights, yields, growth per week, or FCR, among the treatments. This growth rate and FCR is typical of previous production trial at this facility.
Year 2000b. Effects of Feed Management on the Growth, Survival and Feed Conversion of Litopenaeus Vannamei in Culture Ponds. (Garza 2001)
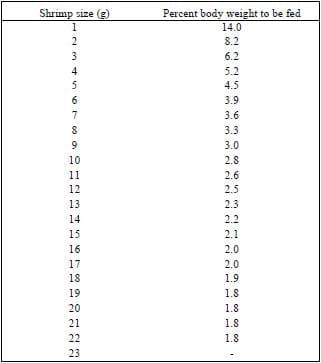
Run in parallel to the previous nursery study, a second study (Garza et al., 2001) was carried out to evaluate the effects of two feed management protocols on the production parameters of L. vannamei raised in ponds. In the first protocol, the N10 treatment from the previous study was used (Fixed FCR treatment). Shrimp in this second protocol were fed at a rate of 20 kg/ha/day for the first ten days after stocking and feed inputs subsequently adjusted considering a Fixed FCR of 1.6, an expected growth based on the average growth of the previous weeks and an assumed survival of 85%. In the second protocol, feed inputs were calculated according to a feeding table published by a commercial feed company (Table 1) and adjusted on a weekly basis using feeding trays (FT treatment).The FT method does not target for any specific FCR, rather, it targets a daily ration, based on percentage of body weight that needs to be fed to the shrimp at a specific point of time depending on the shrimp’s individual weight.
Table 1. Feeding table as recommended by Nicovita, S.A., Lima, Peru for L. vannamei and L. stylirostris cultured at 30 PL/m2. (Clifford 1992).
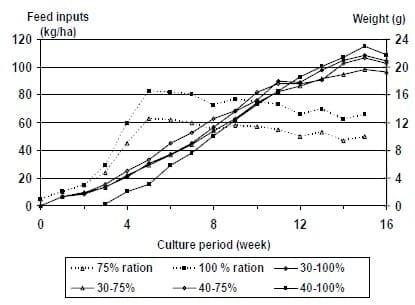
In this study feed inputs followed different patterns (Figure 1) and were dependent on management techniques. Although feed inputs were similar during the first two weeks, by the third week, the Fixed FCR ponds were receiving approximately 80 kg/ha/day. Feed inputs in this treatment stayed relatively consistent throughout the remainder of the study, fluctuating between 80 and 100 kg/ha/day. Feed inputs for ponds fed using the FT protocol, increased gradually during the first part of the study, reaching 80 kg/ha/day by the fifth week and continued to increase until the end of the study. Feed inputs during the final three weeks of the study were between 135 and 150 kg/ha/day. When compared to the commercial recommendations, feed inputs were substantially reduced towards the end of the production cycle using the Fixed FCR protocol.
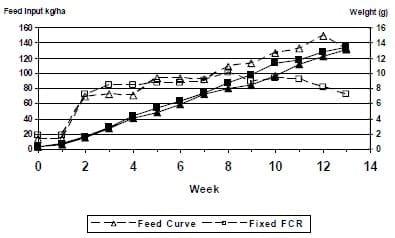
Figure 1: Feed inputs (dashed line) and average weights (solid line) of L. vannamei offered feed (35 % protein) based on two feeding protocols and stocked at 35 shrimp/m2 in 0.1 ha ponds (Year 2000).
At harvest, survivals were significantly higher in ponds where the standard feeding tables and feed trays were used (93%) than in ponds fed using FCRs to determine feed input (77%). Yields were also significantly higher in ponds fed according to the FT protocol (4,420 kg/ha) than in ponds fed using the Fixed FCR schedule (3,747 kg/ha). It should be noted that the increased survival and yields were primarily due to a pond that was overstocked and had over 100% survival. Exclusion of this pond results in similar values that are not significantly different. Despite variations in the previous parameters, there were no differences in final weights, growth rates and FCR Average individual weight were 13.9 g for the shrimp fed using the Fixed FCR method, and 13.6 g for the shrimp fed using the FT protocol. The final FCR was 2.03 for both treatments. This value was higher than expected for the Fixed FCR method because the actual survivals were lower than had been predicted.
The FT method using trays in combination with a feeding table, needed considerably more labor and monitoring, but provided a way to react to shifts in “consumption”. Albeit our experience with feed trays is that they do not truly reflect consumption. However, this gives a general understanding if feed is consumed. The Fixed FCR method was easy to implement and the time consumed analyzing feeding changes was minimal. The weakest point of this technique would be that it depends on more accurate prediction of survivals. Higher survivals (than predicted) could cause this method to under-feed, while lower survival (than predicted) would generate FCRs higher than expected, possibly resulting in overfeeding and deterioration of water and bottom quality in the most extreme cases. On the other hand Feed Tables without the use of feed trays would also have the same problem.
What was most clearly observed during this experiment is the need to refine methods for accurate survival rate estimates and that historical production data could be used to further refine feeding curves. The results clearly show that despite vary different feed inputs towards the end of the growth trial, there were no notable differences in shrimp growth or performance. Additionally, although the use of feeding trays and a feed table resulted in higher feed inputs, the resulting FCR was exactly the same. As there was little response of the shrimp, this was our first indication that feed inputs were in excess of nutritional requirements and that further studies should be conducted to optimize feed inputs.
Lessons from year 2,000 experience.
Using very general numbers, the over estimation of feed requirements can be confirmed using a few back of the envelope calculations. For example, we can estimate the expected feed conversion of a given feed by making a few assumptions about nutrient retention. For this example we will use protein and assume that shrimp contain 15 % protein on a wet weight basis and that using a typical feed 30 % of the dietary protein is retained as new tissues in the shrimp (which contains about 15 % protein). So if we produce 1000g shrimp it would contain 150g protein (1000 x 0.15). If 30% is retained from the feed we would need to feed 500g protein (150g / 0.3) to the shrimp which means that for a 35% protein diet we would need to feed 1.4 kg of feed (500g /0.35). Hence, in the theoretical world we should have a FCR of 1.4:1. In fact if we consider, that there are numerous studies that indicate that natural productivity contributes almost half of the carbon or nitrogen assimilated by the shrimp we should be getting FCR of less than 1.4. These numbers encouraged us to look at alternative feeding strategies in an attempt to improve our feed management.
Year 2001. Influence of Indoor Nursery Densities on Final Pond Production of the Pacific White Shrimp Litopenaeus vannamei. (Zelaya, 2004)
The objective of this study was to evaluate the influence of stocking rates during nursery on production characteristics during the nursery and the subsequent pond production. Replicated nursery tanks were stocked at 25, 38 and 65 Pls /L, low density (LD), medium density (MD) and high density (HD) treatments, respectively. The nursery period was 21 days for all tanks and then distributed in the ponds. Feed inputs were adjusted weekly based on the estimated shrimp biomass and assuming a survival of 83%. Feed calculations also incorporated Feed Conversion Rates (FCR < 2:1). Feed trays were used to help evaluate consumption of the feed. For the first two weeks of the grow-out phase, ponds were fed at a rate of 8 kg/ha. Beginning the third week, feeding rates were based on 15% of the estimated biomass and then gradually reduced each week as the shrimp biomass increased, following guidelines from a feeding table. Maximum feeding rates were set at 180 kg/ha.After 12 weeks of pond production, shrimp mean average weights were 12.5, 13.6 and 10.7 g, survivals were 89%, 68% and 83%, FCR were 1.6, 2.1 and 2.1, and average yields were 4,091, 3,193 and 2,914 kg/ha, for shrimp originating from HD, MD and LD nursery treatments, respectively. No significant difference in final average weight or FCR was found. Significantly greater yields and lower observed FCR were obtained in production ponds stocked with shrimp from the HD nursery tanks.
Findings of this experiment suggests that PL nursery densities in the range of 25 to 65 PL/L with good management practices have limited influence on subsequent growth and survival of shrimp during grow-out. It is interesting to note that during this trial we allowed feed inputs to reach 180 kg/ha without improvements in survival or growth rates as compared to previous year. Also, indicating that feed inputs may be excessive.
Year 2002. Influence of Indoor Nursery Densities on Final Pond Production of the Pacific White Shrimp Litopenaeus vannamei. (Zelaya, 2004)
The objective of this study was to evaluate the influence of post larvae age at stocking on the subsequent grow-out of L. vannamei in ponds. Shrimp were nursed either for 21 days,14 days or directly stocked into the ponds, corresponding to treatments N21, N14 and DS, respectively. Feed adjustments were made weekly based on estimated shrimp biomass as well as observations from feed trays. Feed calculations incorporated feed conversion rates (FCR < 2:1) and an assumed 30% mortality for the 16-week culture period. Feed consumption was monitored with feeding trays. For the first two weeks of the grow-out phase, ponds were fed at a rate of 8 kg/ha. Beginning the third week, feeding rates were based on 15% of the estimated biomass and then gradually reduced each week as the shrimp biomass increased. Biomass was estimated based on the number of shrimp stocked, weekly sample weight and an assumed mortality of 30% fractionated over the 16-week grow-out period. Maximum feeding rates were set at 140 kg/ha, which were lower than the used on the previous year. Because of warm water temperatures and higher standing crops, feeding rates were reduced during the last two weeks of production.
At the end of the 16- week pond production period, mean average weights were 15.4, 16.9 and 14.9 g, survivals were 63%, 62% and 64%, FCR were 2.7, 2.5 and 2.7, average yields were 3,592, 4,005 and 3,374 kg/ha, for N21, N14 and DS treatments, respectively. No significant differences were found among treatments in any of these results. Results indicate that regardless of nursing PL for 14 or 21 days, nursed juveniles did not differ significantly in production characteristics, from shrimp stocked directly from the hatchery. One benefit of head-starting PL was that it extended the period of time which PL could be acquired from a hatchery, avoiding the risk of a possible shortage in supply as the warmer season begins. Once again, although we reduce our feed inputs from traditional levels we did not see an improvement in FCR, survival or growth rates of the shrimp.
Year 2003. Evaluation of Feed Management Strategies for Litopenaeus vnnamei under Pond Production Conditions. (Zelaya, 2004)
Based on poor feed conversion values from the previous year, we decided to evaluate the influence of feed loading on shrimp production. Depending on the point of view some people suggest that feed inputs should be high during the mid portion of the production cycle to promote maximum growth but then feed inputs should be reduced towards the end of the cycle. This assumes that overfeeding early in the production cycle is less likely to cause problems; whereas, over feeding late in the production cycle, is more likely to cause water quality problems. Contrary to this thought, it can also be argued that through the early stages of production, the contribution from natural production is high and feed inputs should be reduced whereas late in the production cycle natural productivity is limited and feed inputs should be maximized
In an attempt to determine the effects of feed management on shrimp production this study evaluated three different feeding strategies (Zelaya et al., 2005). The first was designated as an intermediate feed (IF) strategy, which was based on historical results at this facility. The other two incorporated 25% reductions and increases of this rate. The EAF (Early Aggressive) feeding strategy incorporated a high feed input early in the cycle to maximize early growth of the shrimp with feeding rates reduced during the second half of the cycle when water quality could be more unstable. The LAF (Late Aggressive Feeding) treatment minimized early feed inputs when natural productivity should be adequate and maximized feed inputs late in the cycle when natural productivity was more likely be limited.
At the conclusion of the 18-week pond production period, production characteristics for shrimp reared on the EAF, LAF, and IF feeding protocols were: mean average shrimp weights of 15.2 g, 15.7 g, and 16.2 g; survivals of 81%, 79% and 78%; FCR of 1.5, 2.0 and 1.8; and average yields of 4,328, 4,384 and 4,398 kg/ha. Feed inputs as well as sample weights of the shrimp are presented in Figure 2. There were no significant differences found among treatments for survival, final weight or total yield. Significant differences for the three treatments were found for FCR, with the best FCR found in EAF and the poorest in the LAF treatments. Despite the shifts in feed inputs (25% from the mean), there was no apparent shift in growth rates of the shrimp. This lack of response would indicate that the lower feed inputs are probably more appropriate and that the lowest feed inputs utilized in this study are closer to the required feed inputs for our production systems.
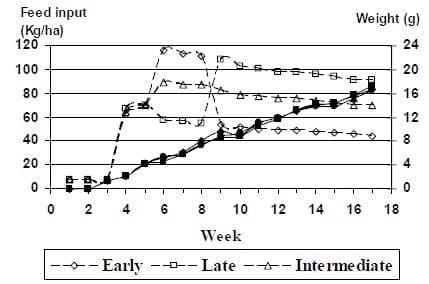
Figure 2: Weekly shrimp weights (solid line) of L. vannamei reared at 35 shrimp/m2 in 0.1 ha ponds and offered various rations (dashed line) using three different feeding protocols.
Observed yields and growth rates were consistent with those of previous studies at this facility. McGraw (2000) reported yields of 3,975 kg/ha with 61% survival; and Garza (2001), who evaluated several feeding methods, reported 3,747 kg/ha with 77% survival. Despite considerable differences in feed inputs, the three protocols resulted in similar growth patterns and none showed a growth decline during the culture period.
Historical Comparison of Results
this facility we found that when total feed inputs were 6,000-6,500 kg/ha with survivals of 68- 88%, FCR averaged around 1.65-2.1; with feed inputs in the range of 9,000-9,600 kg/ha and survivals of 62-64%, then FCR averaged 2.5-2.7; and in this trial with feed inputs of 6,500 (EAF)-7,960 (IF) kg/ha and survivals of 81-77.8%, FCR averaged 1.5-1.8. In cases where feed inputs were 9,070 kg/ha (LAF) even with survivals of 80% FCR averaged 2.0. This suggests that even under close pond management and acceptable survivals, total feed offerings above 8,000 kg/ha/day tend to hurt FCR as they did not improve growth or survival. This is further supported by FCR from our previous years production in which shrimp were stocked at the same density and managed under similar conditions but offered feed using a more liberal feeding protocols. When comparing FCR of the two trials, significantly lower FCR (P< 0.0001) were found for this study (mean FCR 1.77) as compared to that reported for the previous year (mean FCR 2.6). Given, that a number of feeding protocols have been utilized over the years and the fact that even the most conservative feed inputs utilize in this research did not result in notable differences in weekly growth as well as final production one can conclude that feed inputs may be in excess of requirement.
Lessons from years 2001 - 2003.
here is a strong influence of feed management on pond environment, production and economic success. Hence, it is critical to develop and implement feeding programs that take into consideration biological, environmental and economic factors. Under the reported conditions, the various feeding strategies did not profoundly influence weekly growth rates of the shrimp, final yields or gross returns. The tested manipulations of feed inputs did not seem to result in major differences in production but the more conservative feed input did result in a significant improvement in FCR and economic returns.
Year 2004. Variable Feed Allowance with Constant Protein Input for Pacific White Shrimp Litopenaeus Vannamei under Semi-Intensive Conditions in Tanks and Ponds. (Venero, 2006)
Based on the previous years work we felt that we were close to the appropriate feed input and that in addition to looking at a more conservative feed input we should also look at nutrient density. It is well established that growth rate is dependent on nutrient intake and hence, shrimp should respond similarly if nutrient intake is controlled through variable feed allowances. To demonstrate variable feed allowance for shrimp under practical pond conditions, Juvenile L. vannamei were reared in 12 ponds for 114-121 days and fed two practical diets formulated to contain 30 and 40% crude protein. To equalize nutrient intake (protein) two different feeding levels (75, and 100%) were utilized. Once the shrimp reached approximately 1 g, three feeding treatments were assigned to the 12 ponds, allowing four replicate ponds per treatment. The first treatment (30-100%) received a 30% CP feed at a typical feed rate (100% ration) to obtain an expected FCR of 1.8 and a growth rate of 1 g/wk, which was based in previous production records in the same experimental units with the same species. This was essentially, an more conservative feeding protocol based on the previous years data. The second treatment (30-75%) was fed the 30% CP diet, but it was offered at 75% of the feed allowance of a typical ration. The third treatment (40-75%) consisted of feeding a 40% CP diet at 75% of the feed allowance. This matched the total protein inputs of treatment 1 (30-100%). Two weeks after the initial stocking two additional ponds were added to the study. This allowed an observational review of the fourth treatment, which received the 40% CP diet fed at 100% ration.
All ponds were fed equally during the first 18 days of culture (Figure 3). During the first seven days, 1 kg of 35%-CP feed was fed daily to each pond. From day 8 to day 18 shrimp were fed 1.5 kg per day of the same feed. Feeding the treatment diets started at day 19 at half of the total feed input per treatment and continues until day 22. The complete daily ration, for the assigned treatment, was fed after day 23 of pond culture. The amount of daily feed input in each pond was calculated based on the ration for each treatment (75 or 100%) and adjusted weekly for mortalities (assuming an expected mortality of 30% over a 17-week grow-out period).
At the end of the trial (107-121 days) the final weight (19.7-21.7 g), FCR (1.0) and survival (75- 88%) between the treatments 30CP-100% and 30CP-75% were no significantly different. However, production was significantly higher for the treatment 30CP-100% than for the treatment 30CP-75% (6,482 versus 5,054 kg/ha). Although yield was higher for the 40CP-75% treatment than for the 30CP-75% treatment, it tended to be lower than that of the 30% CP diet offered at standard rate (30CP-100%). Although the 40-75% treatment was not included in the statistical analyses due to loss of two ponds, results from the two remaining ponds indicated that production was higher for the 40-75% treatment than for the 30-75% treatment, but it was lower than the 30-100% treatment.
Figure 3: Mean weights (solid line) of L. vannamei reared at 35 shrimp/m2 in 0.1 ha ponds and offered one of two dietary protein levels (30 and 40%) and two feed inputs (dashed lines).
This study demonstrates that increasing the nutrient density (protein content) of the shrimp feed allows a reduction of feed inputs without affecting the growth performance of shrimp. Unfortunately, due to lost of some experimental units in the pond trial results from the pond trial could not be verified statistically. However, tank studies that were run simultaneously, clearly indicated that growth was correlated to protein intake. Hence, reducing feed inputs for nutrient dense diets is a valid technique. Based on work with other species, lower amounts of feed and better feed management would lead to higher efficiency of nutrients with a subsequent reduction in wastes from the feed. This may improve the economic return in the farm and reduce the potential of water pollution and environmental impact on ecosystems receiving the effluents. The improved FCR, growth and net yields from the ponds were much better than previous years indicating that reductions of feed inputs were beneficial to the overall system.
Year 2005. Replacement of Fish Meal in Practical Diets for the Pacific white Shrimp (Litopenaeus Vannamei) Reared under Pond Conditions. (Amaya, 2006)
Based on the positive results of the 2004 work with reduced feed inputs and improved production results a similar feeding protocol was employed in the 2005 production season. This study evaluated a fish meal replacement strategy using vegetable protein sources in practical feeds for marine shrimp reared in ponds. Four commercially extruded diets formulated to contain 35% crude protein and 8% lipids were evaluated. These diets included varying levels of fish meal (9, 6, 3, and 0%) which was replaced by a combination of soybean meal (32.48, 34.82, 37.17 and 39.52% respectively) and corn gluten meal (0.00, 1.67, 3.17, and 4.84% respectively) to replace the protein originating from fish meal. In addition, dietary treatments maintained a minimum lysine and methionine plus cystine content of 5% and 3% of the total protein, respectively, and were supplemented with calcium phosphate to ensure adequate phosphorus supply. Feeds were commercially manufactured and offered as a sinkable, extruded 3 mm pellet. Feed inputs during the 1st, 2nd, 3rd and 4th weeks of pond culture were set at 10, 15, 30 and 60 kg of feed/ha/day, respectively (Figure 4). For the remainder of the growth trial, feed inputs were back-calculated, based on an expected weight gain of 1.5 g per week, a feed conversion of 1.2:1, and a total mortality of 30% (1.66%/wk) distributed over an 18-weeks culture period. Feeding during the first 19 days after stocking was done using the a commercial feed Treatment feeds were offered from day 20, when it was estimated that shrimp had reached 1 g of weight. Maximum feed inputs were set at 83.3 kg of feed/ha/day at the 5th week of culture
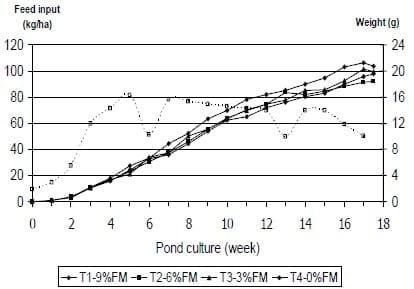
Figure 4: Mean weights (solid lines) of L. vannamei stocked at 35 shrimp/m2 in ponds and fed various diets over a 17.5 weeks experimental period. Feed input (dashed line) was determined based on expected: FCR 1:1.2, Wt Gain 1.5g/wk, 30% Mortality. Note dips in feed inputs are due to disturbances due to hurricanes.
At the conclusion of the experimental period, there were no significant differences (P>0.05) in shrimp production among the test diets. Mean final production, final weight, FCR and survival were evaluated at the end of the 18-week culture period, with final values ranging from 5,363 - 6,548 kg/ha, 18.4 - 20.7 g, 1.38 - 1.12 and 84.0 - 94.0 %, respectively. Although not significant, the economic analysis numerically showed a general increase in the partial gross returns of shrimp production, as higher levels of plant proteins sources were included in diets fed to marine shrimp. Production results obtained in this study are within the production range of comercial shrimp production in semi-intensive production systems. At the same facilities and with similar pond management, these production results were similar to those reported by Venero (2006) and improved those obtained by Zelaya (2005) and Garza (2004). Considering that all the test diets had a fixed level of poultry by-product meal (16%), it can be concluded that up to 71.5 % of the dietary protein can be provided by high quality plant protein sources (0% FM feed) in shrimp diets Results from this study demonstrate that in commercially manufactured shrimp feeds, fish meal can be completely removed from the formulation using alternative vegetable protein sources in combination with poultry by-product meal without compromising the production performance and economic returns of Litopenaeus vannamei reared in pond systems. It is likely that the positive response of shrimp fed the replacement feeds, was the result of a combination of adequate feed formulation and manufacture, accurate feed inputs and management of the pond ecosystem and the contribution of natural food organisms to the total feed intake.
Conclusions
The feed management strategy followed in this research has proved to be an effective alternative for shrimp production under pond conditions. In general, this management strategy involved the use of historical records to determine expected FCR, weekly growth and survival and a continuos evaluation and adjustment of feed input to those parameters based on the dynamic of environmental variables (eg., morning DO, temperature, cloudy days). The utilization of this feed management plan has increased progressively the production levels in the research facilities since its implementation. As previously presented, Garza (2001) compared growth between a traditional feed table and a fixed FCR and did not observe significant differences in growth and production and obtained a FCR of 2.03. On the other hand, following a similar feeding and pond management, Zelaya (2005) obtained a FCR between 1.5-2.0 when feed was offered between 60 to 91 kg/ha/day. Although he did not report significant differences among the treatments, the feed input were reduced by about 17% when compared with programs based on feeding tables and the FCR averaged 1.5-1.8 with production between 4,328 and 4,398 kg/ha. Similarly, in 2005 study (Amaya, 2006), production values (5,363-6,547kg/ha) and survivals (84 -94%), were greater than those of previous years.
There are a number of possible explanation of the improvement in production and FCR over the last few years. The response could be due to genetic improvements, general improvements in culture conditions and/or feed management. Figure 5 provides a graphical summary of the various feed inputs, given that the levels of feed inputs have been reduced from a high of 140- 180 kg/ha/day (Zelaya 2004), using feeding tables, to 60 kg/ha/day using a high protein diet (40% CP) (Venero, 2006) or 81kg/ha/day using a 35%CP diet (Amaya, 2006) feed management and subsequent improvements in water quality, are clearly the primary factors. It should be noted, that for this type of system a maximum daily feed load of 100-120 kg/ha/day has been recommended when night-time mechanical aeration is provided (Boyd, 1989; Boyd and Tucker, 1998). The maximum feed input in the last studies (2004-2005) was between 80-85 kg/ha/day which is below the maximum capacity of feed load suggested for ponds and values used in previous trials.
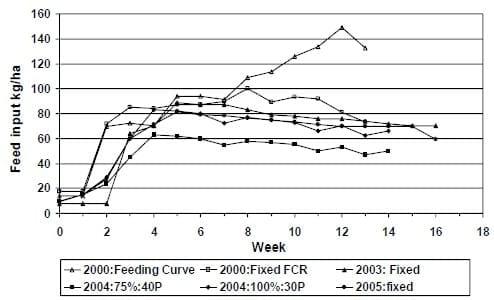
Figure 5: Summary of feed inputs (kg/ha) that have been used to rear L. vannamei in 0.1 ha research ponds stocked at 35 shrimp/m2. Note the 2000 feeding curve represents typical feed inputs at this facility prior to 2000.
Reducing the feed inputs ensures that all of the feed is consumed and that the shrimp utilize natural productivity to it’s full extent. The lower feed levels, in theory, improve water quality that might lead to the higher survival and production observed in these last years production data. Although, aeration time and dissolved oxygen levels have not been compared there is a general feeling that our aeration demand has been reduced and that we are seeing more stable ponds. Overall, every year we have been improving shrimp production as compared to production records from previous years in the same units. The reason for these enhanced results can not be exclusively associated to the modification feeding protocols. It can also be the result of other contributing factors and variables such as genetic pools of PL, climatic conditions, wáter quality, temperature, higher salinity and improved pond bottom conditions.
Acknowledgments
These studies were conducted by a number of graduate students from Auburn University, Department of Fisheries and Allied Aquacultures. The research was also supported by a dedicated staff from Auburn University and Alabama Marine Resources Division. The research was supported by a number of grants and was conducted at the Cluade Petite Mariculture Center in Gulf Shores, Al.
Presented at the VIII Simposium Internacional de Nutrición Acuícola. 15 - 17 Noviembre, 2006. Universidad Autónoma de Nuevo León, Monterrey, Nuevo León, México.
Literature cited
1. Amaya, E., 2006. Fish meal replacement in practical diets for pacific white shrimp (Litopenaeus vannamei) rearedin green water systems. Master of Science Thesis. Auburn University, Auburn, Alabama, USA.
2. Anderson, R. K., P. L. Parker and A. Lawrence. 1987. A 13/12C trace study of the utilization of presented feed by a commercially important shrimp Penaeus vannamei in a pond grow-out systems. Journal of the World Aquaculture Society 18: 148-155.
3. Bianchi, M., E. Bedier, A. Bianchi, A. M. Domenach, and D. Marty. 1990. Use of 15N labeled food pellets to estimate the consumption of heterotrophic microbial communities to penaied prawn diet in closed-system aquaculture. Pages 227-230 in R. Lesel, editor. Microbiology inpoecilotherms. Elsevier Science Publishers, B. V. (Biomedical Division), Amsterdam, The Netherlands.
4. Boyd, C. E. 1992. Shrimp pond bottom soil and sediment management. Pages 166-181 in J.Wyban, editor. Proceedings of the Special Session on Shrimp Farming. World Aquaculture Society, Baton Rouge, LA USA.
5. Boyd, C. E. 1995. Bottom Soils, Sediment and Pond Aquaculture. Chapman & Hall, New York, NY.
6. Boyd, C. E. 1989. Water quality management and aeration in shrimp farming . Alabama Agriculture Experiment Station , Auburn University, AL. Fisheries and Allied Aquaculture, Ser. No. 2, 83 pp.
7. Boyd, C. E. 2000. Farm effluent during drain harvest. Global Aquaculture Advocate 3 :26-27.
8. Boyd, C. E. and C. S. Tucker. 1998. Pond Aquaculture Water Quality Management. Kluwer Academics Publisher, Boston, Massachusetts, USA.
9. Boyd, C. E. and Y. Musig. 1992. Shrimp Pond effluents: observations of the nature of the problem on commercial farms. Pages 195-197 in J. Wyban, editor. Proceedings of the Special Session on Shrimp Farming. World Aquaculture Society, Baton Rouge, LA USA.
10. Briggs, M. R. P. and S. J. Funge-Smith. 1994. A nutrient budget of some intensive marine shrimp ponds in Thailand. Aquaculture Fish Management 25, 798-811.
11. Charatchakool, P., Turnbull, J. F., Funge-Smith, S., Limsuwan C., 1994. Health management in shrimp ponds. Aquatic animal health research institute. Department of Fisheries. Kasetsart University, Bangkok, Thailand
12. Chien, Y. H. 1992. Water Quality requirements and management for marine shrimp culture. Proceedings Special Session on Shrimp Farming. Pages 144-156 in J. Wyban, editor. Proceedings of the special session on Shrimp Farming. World Aquaculture Society, Orlando , Florida, USA.
13. Clifford, H. C. 1992. Marine Shrimp pond management: Review. Pages 110-137 in J. Wyban, editors. Proceeding of the Special session on Shrimp farming. The world Aquaculture Society. Baton Rouge,LA, USA.
14. Cole, B. A. and C. E. Boyd. 1986. Feeding rate, water quality, and channel catfish production in ponds. Progressive Fish Culturist 81:25-29.
15. Cruz, P. S. 1991. Shrimp feed Management : Principles and Practices. Kabukiran Enterprises, Inc. Davao City, Philippines.
16. Dersjant-Li, Y., 2002. The use of soy protein in aquafeeds. Avances en Nutricion Acuicola VI. Memorias del VI Simposium Internacional de Nutricion Acuicola. 3 al 6 de Septiembre del 2002. Cancun, Quintana Roo, Mexico.
17. Dierberg, F. E and W. Kiattisimkul. 1996. Issues, impacts, and implications of shrimp aquaculture in Thailand. Environmental Management 20:649-666.
18. Fast, A. W. and C. E. Boyd. 1992. Water circulation, aeration and other management practices. Pages 457-495 in A.W. Fast, L.J. Lester, editors. Marine Shrimp Culture: Principles and Practices. Elsevier, Amsterdam.
19. Garza, A. 2001. Influence of nursery and feed management on pond production of Litopenaeus vannamei. Master`s thesis. Auburn University, Auburn, Alabama.
20.Garza, A., Rouse, D.B., Davis, D.A., 2004. Influence of nursery period on the growth and survival of Litopenaeus vannamei under pond production conditions. J. World Aquaculture Society. 35 (3), 357-365.
21. Goddard, S. 1996. Feed Management in Intensive Aquaculture. Chapman Hall, New York, NY USA.
22. Green, B. W., D. R. Teichert-Coddington, C. E. Boyd, J. L. Harvin, H. L. Corrales, R. Zelaya, D. Martinez, and E. Ramirez. 1997. The effects of pond management strategies on nutrient budgets, Honduras. Pages 8-121 in B.Goetzel, editor. Pond Dynamics/ Aquaculture Collaborative Research Support Program, Fourteenth Annual Technical Report. PD/ A CRSP, Office of International Research and Development , Oregon State University , Corvallis, OR.
23. Hajek, B. F. and C. E. Boyd. 1994. Rating soil and water information for aquaculture. Aquacultural Engineering 13:115-128.
24. Hopkins, J. S., A. D. Stokes, C. L. Browdy, and P. A. Sandifer. 1992. The relationship between feeding rate, aeration rate and expected dawn dissolved oxygen in intensive shrimp ponds. Aquacultural Engineering, 10: 281-290.
25. Hopkins, J. S., M. R. DeVoe, A. F. Holland, C. L. Browdy, and A. D. Stokes. 1995. Environmental impacts of shrimp farming with special reference to the situation in the continental United States. Estuaries 18:25-42.
26. Hopkins, J. S., R. D. Hamilton, P. A. Sandifer, C. L. Browdy, and A. D. Stokes. 1993. Effect of water exchange rate on production, water quality, effluent characteristics and nitrogen budgets of intensive shrimp ponds. Journal of the World Aquaculture Society, 24 : 304-320.
27. Hopkins, J. S., P. A. Sandifer, and C. L. Browdy. 1994. Sludge management in intensive pond culture of shrimp: effect of management regime water quality, sludge characteristics, nitrogen extinction, and shrimp production. Aquaculture Engineering 13:11-30.
28. Jolly, C. M. and H. A. Clonts. 1993. Economics of Aquaculture. Food Product Press, Binghamton, NY.
29. Jory, D. D. 1995. Feed management practices for a healthy pond environment Pages 118-143 in C.L. Browdy and J. S. Hopkins, editors. Swimming through troubled water, Proceedings of the special session on shrimp farming , Aquaculture ’95. World Aquaculture Society, Baton Rouge, Louisiana, USA.
30. Jory, D., T. R. Cabrera, D. M. Dugger, D. Fegan, P. G. Lee, A.L. Lawrence, C. J. Jackson, R. P. McIntosh, and J. Castaneda. 2001. Perspectives on the application of closed shrimp culture systems. Pages 104-152 in C. L. Browdy and D.E. Jory, editors. The New Wave, Proceedings of the special session on sustainable Shrimp culture, Aquaculture 2001. The World Aquaculture Society, Baton Rouge, Louisiana Orlando, USA.
31. Lawrence, A., F. Castille, T. Samocha, and M. Velasco. 2001. Environmentally friendly or Least polluting feed and feed management for aquaculture.. Pages 84-96 in C. L. Browdy, and Darryl. E. Jory, editors. The New Wave, Proceedings of the special session on sustainable Shrimp culture, Aquaculture 2001. The World Aquaculture Society, Baton Rouge, Louisiana Orlando, USA.
32. Leber, K. M. and G. Pruder. 1988. Using experimental microcosms in shrimp research: the growth enhancing effect of shrimp pond water. Journal of the World Aquaculture Society. 19:197-203.
33. Lin, C. K. 1995. Progression of intensive marine shrimp culture in Thailand. in C.L. Browdy and J. S. Hopkins, editors. Swimming through troubled water, Proceedings of the special session on shrimp farming , Aquaculture ’95. World Aquaculture Society, Baton Rouge, Louisiana, USA.
34. Lovell, T. 1998. Nutrition and Feeding of Fish. Kluwer Academic Publishers, Norwell, Massachusetts.
35. Lovett, D. L. and D. L. Felder. 1990. Ontogeny of kinematics in the gut of the white shrimp Penaeus setiferus (Decapoda:Penaeidae). Journal of Crustacean Biology 10: 53-68.
36. McGraw, W. J. 2000. Effects of increased aeration and aerator type on shrimp production, water quality and pond soil. Doctoral Dissertation. Auburn University, Auburn, Alabama.
37. Moss, S. M. 1995. Production of growth-enhancing particles in a plastic-lined shrimp pond. Aquaculture 132:253- 260.
38. Moss, S. M., G. Pruder, K. M. Leber, and J. A. Wyban. 1992. The relative enhancement of Penaeus vannamei growth by selected fractions of shrimp pond water. Aquaculture 101: 229-239.
39. Muir, P. and Owens, L. 1997. Microbiology of prawn pond sediments- part 2. Austasia Aquaculture 11: 63-65.
40. Nunes, A. G. P. and G. J. Parsons. 2000. Size-related feeding and gastric evacuation measurements for the Southern brown shrimp Penaeus subtilis. Aquaculture 187:133-151.
41. Peterson, E. L. 1999. Benthic shear stress and sediment quality. Aquacultural Engineering 21:85-111.
42. Peterson, E. L. and M. B. Walker. 2002. Effect of speed on Taiwanese paddlewheel aeration. Aquacultural Engineering 26: 129-148.
43. Piedad-Pascual, F. 1993. Aquafeeds and feeding strategies in the Philippines. Pages 317-343 in M. B. New, A.G.J. Tacon, and I Csavas, editors. Farm-made aquafeeds. Proceeding of the FAO/AADCP, Bangkok, Thailand.
44. Pruder, G. D. 1987. Detrital and algal based food chains in aquaculture: a perspective. Pages 296-308 in D. J. W. Moriarty and R.S.V. Pullin, editors. Detritus and Microbial Ecology in Aquaculture. ICLARM Conference Proceedings, Manila, the Philippines.
45. Prder, G. D.1992. Marine shrimp pond effluent: characterization and environmental impact. Pages 187-189 in J.Wyban, editor. Proceedings of the special session on shrimp farming . World Aquaculture Society, Baton Rouge, Louisiana, USA.
46. Ritvo, G., J. Dixon, W. Neill, and T. Samocha. 2000. The effect of controlled soil sulfur concentration on growth and survival of Litopenaeus vannamei. Journal of the World Aquaculture Society 31:381-389.
47. Robertson, L., A. L. Lawrence and F. L. Castille. 1993. Effect of feeding frequency and feeding time on growth of Penaeus vannamei (Boone). Aquaculture and Fisheries Management 24:1-6.
48. Rubright, J. S., J. L. Harrell, H. W. Holcomb, and J. C. Parker. 1981. Responses of planktonic and benthic communities to fertilizer and feed applications in shrimp mariculture ponds. Journal of the World Mariculture Society 12:281-299.
49. Samocha, T. M., A. L. Lawrence, C. R. Collins, C.R. Emberson, J. L. Harvin, and P. M. Van Wyk. 2001. Development of integrated, environmentally sound, inland shrimp production technologies for Litopenaeus vannamei. Pages 64-75 in C. L. Browdy, and D.E. Jory, editors. The New Wave, Proceedings of the special session on sustainable Shrimp culture, Aquaculture 2001. The World Aquaculture Society, Baton Rouge, Louisiana Orlando, USA.
50. Tacon, A.G.J., Barg, U.C., 1998. Major challenges to feed development for marine and diadromous finfish and crustacean species. In: De Silva, S.S. (Ed.), Tropical Mariculture. Academic Press, San Diego, CA, USA, 171 - 208.
51. Teichert-Coddington, D. 1995. Estuarine Water Quality and Sustainable shrimp culture in Honduras. Pages 144- 156 in C.L. Browdy, and J. S. Hopkins, editors. Swimming through troubled water, Proceedings of the special session on shrimp farming, Aquaculture ’95. World Aquaculture Society, Baton Rouge, Louisiana, USA.
52. Teichert-Coddington, D. R., D. B. Rouse, A. Potts, and C. E. Boyd. 1999. Treatment of harvest discharge from intensive shrimp ponds by settling. Aquaculture Engineering 19:147-161.
53. Tucker, L. S., C. E. Boyd, and E. W. McCoy. 1979. Effect of feeding rate on water quality, production of channel catfish, and economic returns. Transactions of the American Fisheries Society 108: 389-396.
54. Velasco, M., A. L. Lawrence and F. L. Catille. 1999. Effect of variation in daily feeding frequency and ration size on growth of shrimp, Litopenaeus vannamei (Boone), in zero-water exchange culture tanks. Aquaculture 26:11-17.
55. Venero, J., 2006. Optimization of dietary nutrient inputs for Pacific white shrimp Litopenaeus vannamei. Doctoral dissertation. Auburn University, Auburn, Alabama, USA.
56. Villalon J. R. 1991. Practical manual for semi-intensive commercial production of marine shrimp. TAMU-SG-91- 501.Texas A&M University Sea Grant College Program, College Station, Texas, USA.
57. Wyban, J. A., G. D. Pruder and K. M. Leber. 1989. Paddlewheel effects on shrimp growth, production and crop value in commercial earthen ponds . Journal of the world Aquaculture Society 20:18-23
58. Wyban J. A., and J. N. Sweeney. 1991. Intensive shrimp production technology. The Oceanic Institute, Honolulu , Hawaii, USA.
59. Zelaya, O., 2005. An evaluation of nursery techniques and feed management during culture of marine shrimp Litopenaeus vannamei. Doctoral dissertation. Auburn University, Auburn, Alabama, USA.
60. Ziemann, D. A., W. A. Walsh, E. G. Saphore, and K. Fulton-Bennett. 1992. A survey of water quality characteristics of effluents from Hawaiian aquaculture facilities. Journal of the World Aquaculture Society 23:180-191.