Research Note: Storage period and prewarming temperature effects on synchronous egg hatching from broiler breeder flocks during the early laying period
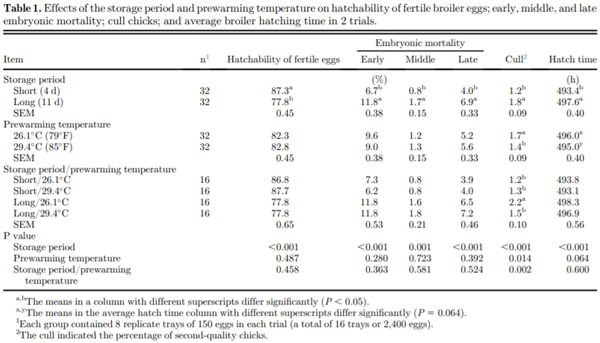
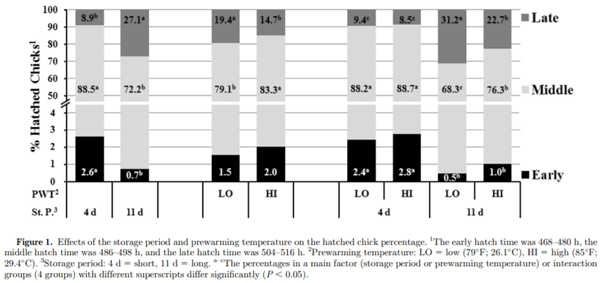
The effects of the storage period and prewarming temperature on embryonic mortality, hatchability, and synchronous hatching of broiler eggs were investigated. Eggs were obtained from commercial flocks of Ross 308 broiler breeders at 27 and 28 wk of age for trials 1 and 2, respectively. In both trials, 2,400 eggs were stored for 4 d (short) or 11 d (long) at 18°C (64.4°F) and 75% RH and were randomly assigned to 2 groups at either a prewarming temperature of 26.1°C (79°F, low) or 29.4°C (85°F, high) for 8 h before setting. The eggs were transferred from setters to hatching baskets at 444 h (18.5 d) of incubation. The hatched chicks were counted at 6-h intervals between 468 h and 516 h of incubation and categorized as early, middle, or late hatching. The eggs stored for 4 d hatched earlier than the eggs stored for 11 d (P < 0.05). An increased prewarming temperature (29.4°C) resulted in a 1.0-h shorter incubation duration, but this difference was not significant (P = 0.064). An interaction between the storage period and prewarming temperature was observed for middle- and late-hatched chicks (P < 0.05). No interactions between the storage period and prewarming temperature were observed for hatchability of fertile eggs or embryonic mortality; however, a significant interaction was found between the storage period and prewarming temperature on the second-quality chick percentage (P < 0.05). The eggs stored for 11 d had a significantly reduced hatchability of fertile eggs owing to increased embryonic mortality than short-stored eggs (P < 0.05). The interaction effect indicated that eggs held for 8 h with prewarming at 29.4°C after 11 d of storage had more middle- and fewer late-hatched chicks and improved chick quality than those that received the 26.1°C prewarming treatment (P < 0.05), but no significant difference was found among the prewarming treatments for eggs stored for 4 d. This study demonstrated that prolonged egg storage resulted in reduced hatchability, increased incubation duration, and an asynchronous hatching time. Moreover, increasing the prewarming temperature could be used to promote uniformity among embryos through synchronous hatch-ing, thus improving broiler flock uniformity and performance of the prolonged stored eggs.
Key words: prewarming temperature, egg storage, synchronous hatching, embryonic mortality, early laying period.
Becker, W. A., and G. E. Bearse. 1958. Pre-incubation warming and hatchability of chicken eggs. Poult. Sci. 37:944–948.
Brake, J. 1996. Optimization of egg handling and storage. World Poult. Misset 12:33–39.
Bruzual, J. J., S. D. Peak, J. Brake, and E. D. Peebles. 2000. Effects of relative humidity during the last five days of incubation and brooding temperature on performance of broiler chicks from young broiler breeders. Poult. Sci. 79:1385–1391.
Careghi, C., K. Tona, O. Onagbesan, J. Buyse, E. Decuypere, and V. Buggeman. 2005. The effects of the spread of hatch and inter-action with delayed feed access after hatch on broiler performance until seven days of age. Poult. Sci. 84:1314–1320.
Decuypere, E., and H. Michels. 1992. Incubation temperature as a management tool: a review. Worlds Poult. Sci. J. 48:28–38.
Dymond, J., B. Vinyard, A. D. Nicholson, N. A. French, and M. R. Bakst. 2013. Short periods of incubation during egg storage increase hatchability and chick quality in long-stored broiler eggs. Poult. Sci. 92:2977–2987.
Ebeid, T. A., F. A. Twfeek, M. H. Assar, A. M. Bealish, R. E. Abd El-Karim, and M. Ragab. 2017. Influence of pre-storage incubation on hatchability traits, thyroid hormones, antioxidative status and immunity of newly hatched chicks at two chicken breeder flock ages. Animal 11:1966–1974.
Elibol, O., B. Hodgetts, and J. Brake. 2002. The effect of storage and pre-warming periods on hatch time and hatchability. Avian Poult. Biol. Rev. 13:243–244.
Elibol, O., and J. Brake. 2008. Effect of egg position during three and fourteen days of storage and turning frequency during subsequent incubation on hatchability of broiler hatching eggs. Poult. Sci. 87:1237–1241.
Elibol, O., M. Gucbilmez, and J. Brake. 2009. Effect of rate of pre-incubation temperature increase on hatchability of broiler hatch-ing eggs. Poult. Sci. 88:46 (Abstr.).
Fasenko, G. M. 2007. Egg storage and the embryo. Poult. Sci. 86:1020–1024.
Gucbilmez, M., S. Ozlu, R. Shiranjang, O. Elibol, and J. Brake. 2013. Effects of preincubation heating of broiler hatching eggs during storage, flock age, and length of storage period on hatchability. Poult. Sci. 92:3310–3313.
Hodgetts, B. 1999. Incubation and hatching. Page 53 in The Poultry Production Guide. K. Naheeda, ed. Elsevier Intl., Doetichem, the Netherlands.
Hudson, B. P., B. D. Fairchild, J. L. Wilson, W. A. Dozier, and R. J. Buhr. 2004. Breeder age and zinc source in broile breeder hen diets on progeny characteristics at hatching. J. Appl. Poult. Res. 13:55–64.
Kamanli, S., I_. Durmus, and H. Ayg€oren. 2009. Effect of different temperature and period pre-warming on hatching traits. J. Poult. Res. 8:20–22.
Lin, Y. M., S. Druyan, S. Yahav, and J. Brake. 2017. Thermal treatments prior to and during the beginning of incubation affects development of the broiler embryo and yolk sac membranes, and live performance and carcass characteristics. Poult. Sci. 96:1939–1947.
Mather, C. M., and K. Laughlin. 1976. Storage of hatching eggs: the effect on total incubation period. Br. Poult. Sci. 17:471–479.
Mayes, F., and M. Takeballi. 1984. Storage of the eggs of the fowl (Gallus domesticus) before incubation: a review. Worlds Poult. Sci. J. 40:131–140.
Meijerhof, R., J. Noordhuizen, and F. Leenstra. 1994. Influence of pre-incubation treatment on hatching results of broiler breeder eggs produced at 37 and 59 weeks of age. Br. Poult. Sci. 35:249–257.
Molenaar, R., I. A. M. Reijrink, R. Meijerhof, and H. van den Brand. 2010. Meeting embryonic requirements of broilers throughout incubation: a review. Braz. J. Poult. Sci. 12:137–148.
Nasri, H., H. van den Brand, T. Najjar, and M. Bouzouaia. 2020. Egg storage and breeder age impact on egg quality and embryo devel-opment. J. Anim. Physiol. Anim. Nutr. 104:257–268.
Nicholson, D. 2012. Improving hatchability after longer periods of egg storage. Intl. Hatch. Pract. 26:23–24.
Ozlu, S., O. Elibol, and J. Brake. 2018a. Effect of storage temperature fluctuation on embryonic development and mortality, and hatch-ability of broiler hatching eggs. Poult. Sci. 97:3878–3883.
Ozlu, S., R. Shiranjang, O. Elibol, and J. Brake. 2018b. Effect of hatching time on yolk sac percentage and broiler live performance. Braz. J. Poult. Sci. 20:231–236.
Ozlu, S., A. U ar, R. Banwell, and O. Elibol. 2019. The effect of increased concentration of carbon dioxide during the first 3 days of incubation on albumen characteristics, embryonic mortality and hatchability of broiler hatching eggs. Poult. Sci. 98:771– 776.
Piestun, Y., S. Druyan, J. Brake, and S. Yahav. 2013. Thermal treatments prior to and during the beginning of incubation affect phenotypic characteristics of broiler chickens posthatching. Poult. Sci. 92:882–889.
Pokhrel, N., E. B.-T. Cohen, O. Genin, M. Ruzal, D. Sela-Donenfeld, and Y. Cinnamon. 2018. Effects of storage condi-tions on hatchability, embryonic survival and cytoarchitectural properties in broiler from young and old flocks. Poult. Sci. 97:1429–1440.
Reijrink, I., R. Meijerhof, B. Kemp, E. Graat, and H. van den Brand. 2009. Influence of prestorage incubation on embryonic development, hatchability, and chick quality. Poult. Sci. 88:2649– 2660.
Reijrink, I. A. M., D. Berghmans, R. Meijerhof, B. Kemp, and H. van den Brand. 2010. Influence of egg storage time and preincubation warming profile on embryonic development, hatchability, and chick quality. Poult. Sci. 89:1225–1238.
Renema, R. A., J. J. R. Feddes, K. L. Schmid, M. A. Ford, and A. R. Kolk. 2006. Internal egg temperature in response to pre-incubation warming in broiler breeder and Turkey eggs. J. Appl. Poult. Res. 15:1–8.
Shiranjang, R., S. Ozlu, and O. Elibol. 2018. The effect of egg storage period, hatching time, and initial brooding litter temperature on performance of chicks from young breeder. Pages 50–55 in Proc. Intl. Poult. Sci. Cong., Turkey, May. World’s Poultry Science Association Turkish Branch, Nigde, Cappadocia-Turkey.
Sunde, M. L., and H. R. Bird. 1959. The effect of pullet maturity on fertility and hatchability of eggs. Poult. Sci. 38:272–279.
Tona, K., F. Bamelis, B. De Ketelaere, V. Bruggeman, V. M. B. Moraes, J. Buyse, O. Onagbesan, and E. Decuypere. 2003a. Effects of egg storage time on spread of hatch, chick quality, and chick juvenile growth. Poult. Sci. 82:736–741.
Tona, K., R. Malheiros, F. Bamelis, C. Careghi, V. Moraes, O. Onagbesan, E. Decuypere, and V. Bruggeman. 2003b. Effects of storage time on incubating egg gas pressure, thyroid hormones, and corticosterone levels in embryos and on their hatching parameters. Poult. Sci. 82:840–845.
Tona, K., O. Onagbesan, B. De Ketelaere, E. Decuypere, and V. Bruggeman. 2004. Effects of age of broiler breeders and egg storage on egg quality, hatchability, chick quality, chick weight, and chick posthatch growth to forty-two days. J. Appl. Poult. Res. 13:10–18.
van Roovert-Reijrink, I. A. M., C. W. van der Pol, R. Molenaar, and H. van den Brand. 2018. Effect of warming profile at the onset of incubation on early embryonic mortality in long stored broiler eggs. Poult. Sci. 97:4083–4092.
Willemsen, H., M. Debonne, Q. Swennen, N. Everaert, C. Careghi, H. Han, V. Bruggeman, K. Tona, and E. Decuypere. 2010. Delay in feed access and spread of hatch: Importance of early nutrition. World’s Poult. Sci. J. 66:177–188.
Wilson, H. 1990. Physiological requirements of the developing em-bryo: temperature and turning. Pages 145–156 in Avian Incuba-tion. S. G. Tullet, ed. Butterworths, London, UK.
Yousaf, A., A. Jabbar, and Y. Ditta. 2017. Effect of pre-warming on broiler breeder eggs hatchability and post-hatch performance. J. Anim. Health Pro. 5:1–4.


United States






