A rapid test for avian influenza detects swine influenza virus
Published: October 14, 2016
By: G. M. Nava 1, R. Merino 2, R. Jarquin 3, N. Ledesma 2, I. Sanchez-Betancourt 2, E. Lucio 4, E. Martínez 2, M. Escorcia 2.
/ 1 Facultad de Química, Universidad Autónoma de Querétaro, Querétaro, México; 2 Facultad de Medicina Veterinaria y Zootecnia, Universidad Nacional Autónoma de México, México, DF, México; 3 Department of Food Science and Technology, University of Tennessee, Knoxville, TN, USA; 4 Investigación Aplicada, Tehuacán, Puebla, México.
The A (H1N1) pdm09 influenza pandemic and, most recently, the A(H3N2) variant outbreak in several areas of the USA are examples of swine influenza viruses infecting humans. These cases highlight the need for reliable and rapid diagnostic tests to elucidate the epidemiology and evolution of swine influenza viruses (Smith and others 2009, Centers for Disease Control and Prevention 2012a, b, c). Currently, there are numerous commercial kits based on rapid-immunomigration techniques available for a fast detection of avian influenza viruses (Chen and others 2010). These rapid-immunomigration kits use specific antibodies against nucleoprotein (NP) of type A influenza viruses. Because the NP proteins are highly conserved between influenza viruses (Shu and others 1993, Li and others 2009), it is of relevance to assess if rapid-immunomigration kits designed for avian influenza are effective to detect influenza viruses in swine populations. Thus, the main goal of the present study was to evaluate the sensitivity and specificity of a commercial kit intended for avian samples, for samples obtained from backyard and commercial farm pigs.
All procedures in this study were performed following the Good Laboratory Practices and its recommended biosecurity guidelines (Centers for Disease Control and Prevention 2012a, b, c). Handling and sampling of animals were performed as indicated in the Mexican Official Regulation 062-ZOO-1999, which outlines technical specifications for the reproduction, care and use of laboratory animals (FMVZ 2012).
For the present study, we collected 48 nasal swabs from backyard pigs without clinical signs of respiratory disease from six different locations in a municipality, and 1513 nasal swabs from pigs with varying degrees of respiratory clinical signs from 17 commercial farms. Swabs from commercial farms were pooled for pigs of comparable age and housed in the same pen. This sample pooling generated a total of 226 samples. Samples were collected as recommended by the Manual of Diagnostic Tests and Vaccines for Terrestrial Animals 2012, and were kept frozen at −70°C until further processing (OIE 2012). Two reference antigens were used as positive controls, A/ swine/New Jersey/11/76 (H1N1), GenBank accession number K00992; and A/swine/Minnesota/9088-2/98 (H3N2), GenBank accession number AF153234. These viruses were kept as stock at −70°C, in chicken-embryo allantoic fluid (H1N1) or Madin-Darby Canine Kidney (MDCK) cell-line (H3N2). Presence of influenza viruses in swab samples was evaluated by rapid-immunomigration test using a commercial-kit (Flu Detect, Synbiotics Corporation; California, USA) following the manufacturer’s instructions. Detection of influenza virus was also performed by virus isolation (VI) and haemagglutination (HA) assays (OIE 2012). Samples positive in rapid-immunomigration and VI-HA assays were then tested by reverse transcription-PCR (RT-PCR). Viral RNA was extracted with the QIAamp Viral RNA mini kit (Qiagen, California, USA) as recommended by the manufacturer. RNA was amplified using primers targeting haemagglutinin genes H1 and H3 (H1-forward: 5′-GGGCAGTCAGGATATGACAGCT-3′ and H1-reverse: 5′-ATTGCCCCCAGGGAGACCAACA-3′, generating an amplicon of 528 bp; and H3-forward: 5′-TATGCCTGGTTTTCGCTCAA-3′ and H3-reverse: 5′-TTCGGGATTACAGTTTGTTG-3, producing an amplicon of 698 bp). Both primers-pairs were validated using negative (blue-eye disease virus) and positive controls (reference avian-influenza viruses). The RT-PCR reaction was carried out with the Super Script One Step RT-PCR with Platinum Taq kit (Invitrogen, California, USA) using a Perkin Elmer Cetus-480 thermocycler. The cycling protocol was as follows: 1 cycle 50°C (30 minutes), 94°C (2 minutes), 45 cycles of 94°C (15 seconds), 54.5°C (1 minutes), 72°C (1.45 minutes), followed by one cycle of 72°C (10 minutes). Specificity of the PCR assay was confirmed by visualising single bands corresponding to DNA fragments of the expected size via ethidium bromide/agarose gel electrophoresis, and by Sanger sequencing of the PCR products.
Diagnostic sensitivity and specificity of the rapid-immunomigration test were estimated by comparing the results obtained via VI-HA and RT-PCR assays. Agreement between assays was established by calculating the κ value coefficient with the Win Episcope V.2.0 software (Thrusfield and others 2001). Interpretation of the κ value is as follows: <0=none, 0.00–0.20=minimum, 0.21–0.40=regular, 0.41–0.60=good, 0.61–0.80=excellent and 0.81–1.00=almost perfect (Morilla and Gonzalez 1998, Viera and Garret 2005).
Analysis of samples from backyard pigs without respiratory clinical signs identified seven influenza-positive samples by the rapidimmunomigration test; from these, only one was confirmed as positive in both, VI-HA and RT-PCR assays. Sensitivity of immunomigration test was 100 per cent, meanwhile the specificity was 87 per cent. Estimated κ value was 0.45, defined as good agreement between tests. In samples from commercial farms, animals with respiratory clinical signs, five influenza-positive samples were detected in the immunomigration test, and all of them were confirmed as positive in VI-HA and RT-PCR assays. In this case, sensitivity and specificity of the immunomigration test was 100 per cent with a κ value equal to 1.0, corresponding to an agreement between assays classified as almost perfect (Table 1). Together, these results indicate that this rapidimmunomigration test is an effective biomedical tool for influenza A virus surveillance in animal populations. In fact, it has been shown that the rapid-immunomigration test is a good surveillance tool for detection of influenza viruses in farm and waterfowl avian populations (Chua and others 2007, Felt and others 2008, Loth and others 2008, Spackman and others 2009), as well as experimental ferrets (Wan and others 2008, Song and others 2009). It is worth pointing out that the specificity and κ values were lower in samples from pigs without respiratory clinical signs than samples from animals with signs of respiratory disease. This lower performance in apparently healthy animals could be associated with samples recovered from animals with low influenza activity (Chua and others 2007). The sensitivity of influenza antigen-detection methods is directly influenced by 2008). Thus, sensitivities are higher in samples obtained from sick and dead animals than samples from apparently healthy animals (Chua and others 2007, Marche and Van den berg 2010, Sanchez and others 2010).
Therefore, influenza virus surveillance from clinically healthy populations should be taken with caution. In conclusion, the present study indicates that the rapid-immunomigrationtest is a rapid and reliable surveillance tool for detection of influenza viruses in farm and backyard animal populations due to its sensitivity and specificity. More important, this diagnostic tool is easy to use, fast and straightforward in field conditions. We believe this diagnostic assay could be an important tool to perform rapid animal population screens in regional or national influenza virus surveillance programmes. | Table 1: Comparison of results from three tests for detection of influenza A virus in nasal swab samples from pigs with or without signs of respiratory disease. 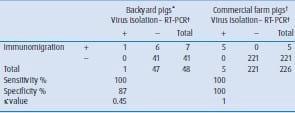 *Backyard pigs without clinical signs †Pigs from commercial farms showing respiratory disease signs ‡Presence of virus was confirmed by isolation and subtype genetic typification by RT-PCR RT-PCR, reverse transcription-PCR |
Acknowledgements
The authors thank Dr Humberto Ramírez Mendoza (FMVZ-UNAM) for providing the H1N1 and H3N2 viruses. Also, thanks to Dr Patty Miller for helping with the English editing of this manuscript.
Funding This project was supported by the Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica (PAPIIT) IN 201110 from the Universidad Nacional Autónoma de México (UNAM) and by the SSA/IMSS/ISSSTE/CONACYT Salud-2009-CO2-126619 project.
This article was originally published in Veterinary Record 2013 173: 424, doi:10.1136/vr.101527. This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 3.0) license (http://creativecommons.org/licenses/ by-nc/3.0/).
References
1. CENTERS FOR DISEASE CONTROL AND PREVENTION. (2012a) Influenza A (H3N2) variant virus. http://www.cdc.gov/flu/swineflu/h3n2v-outbreak.htm
2. CENTERS FOR DISEASE CONTROL AND PREVENTION. (2012b) Good laboratory biosafety influenza virus. http://www.cdc.gov/h1n1flu/guidelines_labworkers. htm
3. CENTERS FOR DISEASE CONTROL AND PREVENTION. (2012c) Prevention and control of influenza: Recommendations of the Advisory Committee on Immunization Practices. http://www.cdc.gov/vaccines/acip/index.html
4. CHEN, Y., XU, F., GUI, X., YANG, K., WU, X., ZHENG, Q., GE, S., YUAN, Q., YEO, A. E., ZHANG, J., GUAN, Y., CHEN, H. & XIA, N. (2010) A rapid test for the detection of influenza A virus including pandemic influenza A/H1N1 2009. Journal of Virological Methods 167, 100–102
5. CHUA, T. H., ELLIS, T. M., WONG, C. W., GUAN, Y., GE, S. X., PENG, G., LAMICHHANEX, C., MALIADIS, C., TAN, S. W., SELLECK, P. & PARKINSON, J. (2007) Performance evaluation of five detection tests for avian influenza antigen with various avian samples. Avian Diseases 51, 96–105
6. DUGAN, V. G., CHEN, R., SPIRO, D. J., SENGAMALAY, N., ZABORSKY, J., GHE DIN, E., NOLTING, J., SWAYNE, D. E., RUNSTADLER, J. A., HAPP, G. M., SEENNE, D. A., WANG, R., SLEMONS, R. D., HOLMES, E. C. & TAUBENBERGER, J. K. (2008) The evolutionary genetics and emergence of avian influenza viruses in wild birds. PLoS Pathogens 4, e1000076
7. FACULTAD DE MEDICINA VETERINARIA Y ZOOTECNIA (FMVZ) (2012). Norma Oficial Mexicana NOM-062-ZOO-1999 Especificaciones técnicas para la producción, cuidado y uso de los animales de laboratorio. http://www.fmvz.unam.mx/fmvz/ principal/archivos/062ZOOPDF
8. FELT, S. A., SAAD, M. D & YINGST, S. L. (2008) A multi-species outbreak of highly pathogenic avian influenza (H5N1) in Grd Jotyar, Iraq: a detailed report. Libian Journal of Infectious Diseases 2, 45–51
9. LI, Z., WATANABE, T., HATTA, M., WATANABE, S., NANBO, A., OZAWA, M., KAKUGAWA, S., SHIMOJIMA, M., YAMADA, S., NEUMANN, G. & KAWAOKA, Y. (2009) Mutational analysis of conserved amino acids in the influenza A virus nucleoprotein. Journal of Virology 83, 4153–4162
10. LOTH, L., PRIJONO, W. B., WIBAWA, H. & USMAN, T. B (2008) Evaluation of two avian influenza type A rapid antigen tests under Indonesian field conditions. Journal of Veterinary Diagnostic Investigation: Official Publication of the American Association of Veterinary Laboratory Diagnosticians, Inc 20, 642–644
11. MARCHE, S. & VAN DEN BERG, T. (2010) Evaluation of rapid antigen detection kits for the diagnosis of highly pathogenic avian influenza H5N1 infection. Avian Diseases 54, 650–654
12. MORILLA, G. A. & GONZÁLEZ, V. D. (1998) Concordancia entre dos pruebas (Kappa). In Introducción al Diagnóstico Inmunológico De Las Enfermedades De Los Animales Domésticos. Ed G. A. Morilla. México, DF: INIFAP-SAGARPA, 34–35
13. OIE: WORLD ORGANIZATION FOR ANIMAL HEALTH. (2012). Swine Influenza. http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.08.08_SWINE_ INFLUENZA.pdf
14. SANCHEZ, M. D. M., CARREON, N. R. & PALACIOS, A. J. M. (2010) Comparison of two diagnostic methods for the detection of the porcine influenza virus. Veterinary Mexicana 41, 45–58
15. SHU, L. L., BEAN, W. J. & WEBSTER, R. G. (1993) Analysis of the evolution and variation of the human influenza A virus nucleoprotein gene from 1933 to 1990. Journal of Virology 67, 2723–2729
16. SMITH, G. J., VIJAYKRISHNA, D., BAHL, J., LYCETT, S. J., WOROBEY, M., PYBUS, O. G., MA, S. K., CHEUNG, C. L., RAGHWANI, J., BHATT, S., PEIRIS, J. S., GUAN, Y. & RAMBAUT, A. (2009) Origins and evolutionary genomics of the 2009 swineorigin H1N1 influenza A epidemic. Nature 459, 1122–1125
17. SONG, H., Wan, H., ARAYA, Y. & PEREZ, D. R. (2009) Partial direct contact transmission in ferrets of a mallard H7N3 influenza virus with typical avian-like receptor specificity. Virology Journal 6, 126
18. SPACKMAN, E., PANTIN-JACKWOOD, M. J., SWAYNE, D. E. & SUAREZ, D. L. (2009) An evaluation of avian influenza diagnostic methods with domestic duck specimens. Avian Diseases 53, 276–280
19. THRUSFIELD, M., ORTEGA, C., DE BLAS, I., NOORDHUIZEN, J. P. & FRANKENA, K. (2001) WIN EPISCOPE 2.0: improved epidemiological software for veterinary medicine. The Veterinary Record 148, 567–572
20. VIERA, A. J. & GARRET, J. M. (2005) Understanding interobserver agreement: the Kappa statistic. Family Medicine 37, 360–363
21. WAN, H., SORRELL, E. M., SONG, H., HOSSAIN, M. J., RAMIREZ-NIETO, G., MONNE, I., STEVENS, J., CATTOLI, G., CAPUA, I., CHEN, L. M., DONIS, R. O., BUSCH, J., PAULSON, J. C., BROCKELL, C., WEBBY, R., BLANCO, J., AL-NATOUR, M. Q. & PEREZ, D. R. (2008) Replication and transmission of H9N2 influenza viruses in ferrets: evaluation of pandemic potential. PLoS ONE 3, e2923
Related topics:
Authors:
Merial Mexico
UNAM - Universidad Nacional Autónoma de México
UNAM - Universidad Nacional Autónoma de México
Show more
Recommend
Comment
Share
Food & Allied Group of Companies
19 de octubre de 2016
Very interesting. It is practical for the diagnostic laboratories which can use one kit for both diseases.
Recommend
Reply

Would you like to discuss another topic? Create a new post to engage with experts in the community.









