Background
Thermophilic Campylobacters are the most common bacterial cause of diarrhoea in humans worldwide [1]. Enteric diseases caused by the thermophilic species C. jejuni, C. coli, C. lari, and C. upsaliensis range from asymptomatic infections to severe inflammatory bloody diarrhoea [2]. C. jejuni is often associated with the Guillain–Barré syndrome [3]. Virulence mechanisms in Campylobacteriosis are currently poorly understood.
Poultry and poultry products remain the most common source of foodborne human Campylobacteriosis [4, 5]. The natural habitat of thermophilic Campylobacter is the intestinal tract of healthy birds and raw meat that can be contaminated during the slaughtering process. Consumption of undercooked chicken meat or contaminated ready-to-eat food is the most common source of infection. Campylobacter are also found in pigs and cattle. Swine carcasses are often contaminated with faeces at the slaughter and processing facilities during the evisceration process, which ultimately leads to contaminated food products [6–8]. Compared to poultry, the relevance of swine in foodborne Campylobacteriosis is not well studied. However, a high incidence of Campylobacter on pork products at the retail level was found [9, 10].
South East Asia including Vietnam was often considered a hotspot for emerging infectious diseases [11]. Vietnam is currently a developing country and knowledge about Campylobacter and Campylobacteriosis is limited. Only few data exist about the prevalence of Campylobacter in children [12, 13] and adults [14]. The prevalence rates of Campylobacter in cases of diarrhea were between 2 and 4 % for children and <1 % for adults. In a study concerning the incidence of diarrhea in rural Vietnamese children [15] Campylobacter was the most frequently identified pathogen comprising 31 % of all isolates.
Fifteen to 32 % of meat samples in different regions of Vietnam contained thermophilic Campylobacter [16– 21]. Duck meat and pork were also contaminated with Campylobacter in 23.9 and 53.7 % of tested samples, respectively [21]. Bao et al. isolated thermophilic Campylobacters from 35.1 % of chicken carcasses in large and small abattoirs of Ho Chi Minh City and 67.9 % of the isolates belonged to the species C. jejuni [22]. However, Schwan investigated meat samples from markets in the Can Tho Province but found no Campylobacter spp. [19].
Several molecular biological methods for characterization and discrimination of Campylobacter isolates have been developed [23]. PCR and flaA typing were used as well as multi-locus sequence typing (MLST) and microarray assays for determination of relatedness among isolates [24, 25].
The molecular genetics of Campylobacter has been extensively studied but the pathogenesis of Campylobacter infections is not fully understood. A number of putative virulence and toxin genes that may contribute to pathogenicity in human Campylobacter infection have partly been identified and sequenced [26–29].
Flagella-mediated motility, adherence to intestinal epithelial cells, invasion and survival in the host cells as well as the ability to produce toxins are important virulence factors [27]. The involvement of the flaA gene in Campylobacter colonization has been shown [30]. Several Campylobacter cytotoxins have been identified [31] and the cytolethal distending toxin (CDT) has been characterized in detail [32, 33]. CDT is composed of three subunits and it has been suggested that CDT, amongst other functions, may play a role in adhesion and invasion [34]. Active CDT is lethal for host enterocytes [35, 36].
It was shown that 19–53 % of Campylobacter spp. strains contain plasmids of various sizes [37]. The plasmid-encoded virB11 gene is a marker potentially associated with the virulence of Campylobacter species [38].
A study with Vietnamese isolates dealt with the identification of possible virulence markers like a novel protein translocation system, the type-6 secretion system [39].
The antimicrobial resistance of Campylobacter isolates was investigated also in several studies [19, 20, 40]. High resistance rates in C. jejuni were determined against ciprofloxacin, nalidixic acid and tetracycline with 64, 46 and 68 %, respectively. Resistance against antibiotics in C. coli isolates was higher than in C. jejuni. All C. jejuni isolates were resistant to ciprofloxacin and nalidixic acid while, 83 % showed resistance to tetracycline [19]. These isolates were recovered from faeces. The broth microdilution method is an easy and reliable method for interpreting minimum inhibitory concentration (MIC) values for C. jejuni and C. coli which is also recommended by EUCAST [41–43]. The emergence of antimicrobial resistance in Campylobacter, particularly to fluoroquinolones, has showed the need for continued monitoring of Campylobacter resistance.
In this study, Vietnamese Campylobacter isolates were characterized to assess their genetic relatedness, potential virulence factors and antibiotic resistance profiles. The isolates were recovered from chicken and pig meat from two slaughterhouses in Hanoi. The investigation was done using different molecular biological tests, MLST, microarray analysis, and the antimicrobial susceptibility was assessed.
Methods
Campylobacter isolates
Campylobacter were isolated from 100 chicken meat and 50 pork samples of two slaughterhouses in Hanoi, Vietnam, following the International Standards Organization [ISO] 10272-1 (2006) guidelines [44] by the Institute of Veterinary Science in Hanoi, Vietnam, in 2009. Bacteria were stored using the Cryobank system (Mast Diagnostica, Reinfeld, Germany) and transferred to the National Reference Laboratory of Campylobacteriosis at the Institute of Bacterial Infections and Zoonoses of the Friedrich-Loeffler-Institut in Jena, Germany. Campylobacter isolates were sub-cultured on Mueller-Hinton Agar (Oxoid GmbH, Wesel, Germany) supplemented with 10 % bovine blood under microaerophilic conditions (5 % O2, 10 % CO2 and 85 % N2) at 42 °C for 48 h. Isolates were kept in cryovials at −80 °C.
DNA extraction
Genomic DNA was extracted from 48-h bacterial cultures on Mueller-Hinton blood agar plates using the High Pure PCR Template Preparation Kit™ according to the manufacturer’s instructions (Roche Diagnostics GmbH, Mannheim, Germany). Extracted DNA was quantified spectrophotometrically using a Nanodrop® ND-1000 (Fisher Scientific GmbH, Schwerte, Germany). DNA extracts were stored at −20 °C.
Species confirmation
Bacterial isolates were identified using a multiplex PCR assay [25] targeting the mapA and ceuE genes.
flaA-RFLP typing
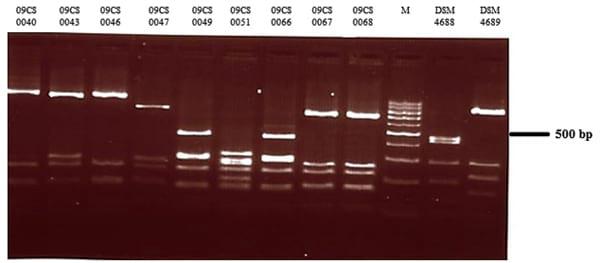
flaA-RFLP (flagellin A-restriction fragment length polymorphism) typing was performed as described previously [24]. Briefly, a part of the flaA gene of the isolates was amplified using primer pair flaA1-Wob/fla2-Wob (Jena Bioscience GmbH, Jena, Germany). The approximately 1700 bp amplicons were digested with DdeI (Roche Diagnostics GmbH) as recommended by the manufacturer. The DNA segments were analyzed after electrophoresis on a 1.5 % agarose gel by staining with ethidium bromide and visualization under UV light. Documentation was carried out using a Bio Imaging System (Syngene, Cambridge, UK).
Multilocus sequence typing
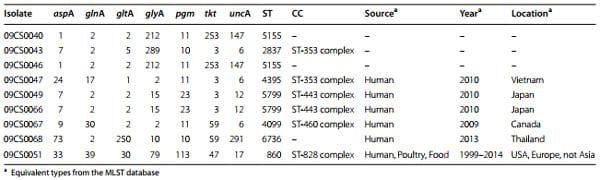
Seven housekeeping gene loci including aspA (aspartase A), glnA (glutamine synthetase), gltA (citrate synthase), glyA (serine hydroxyl methyl transferase), pgm (phosphor glucomutase), tkt (transketolase), and uncA (ATP synthase α subunit) were amplified by PCR as described previously [3]. PCR conditions were modified: after initial denaturation at 96 °C for 60 s followed 35 cycles of denaturation at 96 °C for 15 s, annealing at 50 °C for 1 min, and extension at 72 °C for 1 min. Amplicons were examined by gel electrophoresis on a 1.5 % agarose gel and purified with the QIAamp Gel Extraction Kit (Qiagen, Hilden, Germany) according to the recommendations of the manufacturer. Cycle sequencing was carried out using BigDye Terminator v1.1 Cycle Sequencing Kit (Applied Biosystems, Darmstadt, Germany). Analysis of sequencing products was done with a genetic analyzer ABI PRISM 3130 (Applied Biosystems).
Alleles, sequence types (STs), and clonal complexes (CCs) were assigned by submitting DNA sequences of amplicons to the MLST database available at the following website: http://pubmlst.org/Campylobacter.
Microarray DNA hybridization assay
DNA microarray analysis described here was based on the presence or absence of gene loci of Campylobacter jejuni isolates using the ArrayTube™ platform (Alere Technologies GmbH, Jena, Germany) [45]. Two types of microarrays with spotted probes were used to differentiate C. jejuni isolates: C. jejuni-1 and Campy-2. Sample processing was done using a commercial kit (Alere Technologies GmbH) according to the manufacturer’s instructions (www.alere-technologies.com). Briefly, 1 µg of genomic DNA was amplified and labelled by PCR with random primers and biotin-16-dUTP. Labelled DNA was hybridized to both microarrays for 1 h at 45 °C, washed, and quantified after colorimetric reaction using horseradish peroxidase and TrueBlue substrate. Hybridization signals were measured after 5 min precipitation with an ArrayTube transmission reader ATR-03 (Alere Technologies GmbH). Interpretation of array data was described by El-Adawy et al. [24]. SplitsTree analysis was done using BioNumerics (version 4.6; Applied Maths NV, SintMartens-Latem, Belgium).
Molecular biological characterization of Campylobacter isolates
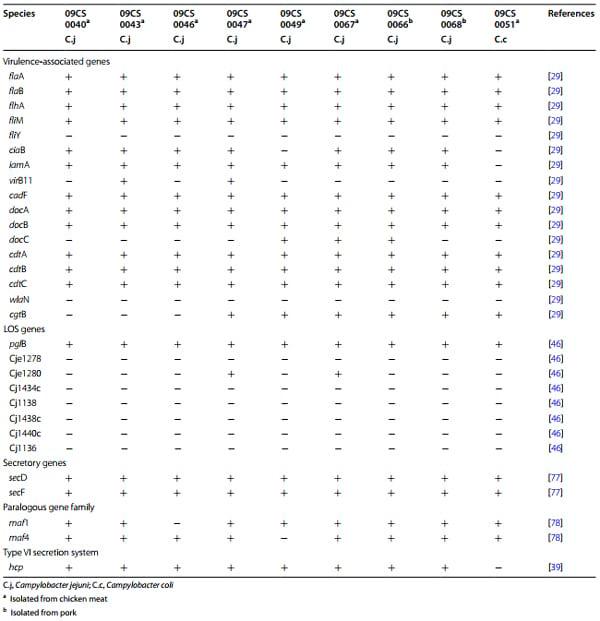
Detection of genes which have functions for motility, adhesion, colonization, invasion, toxin production, lipooligosaccharide (LOS) biosynthesis was carried out by PCRs as described previously [29, 46]. The presence of additional gene loci was detected as described in publications cited in Table 1.
Table 1 Presence of virulence-associated genes, lipooligosaccharide biosynthesis genes and other gene loci in Vietnamese Campylobacter isolates
Antimicrobial susceptibility testing and MIC determination
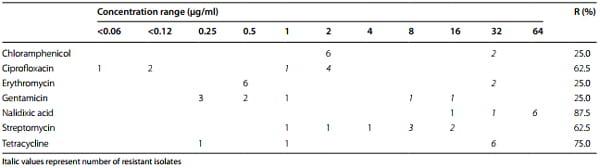
The broth micro-dilution test was performed with Sensititre Campylobacter plates EUCAMP 2 (MCS Diagnostics BV, RE Swalmen, The Netherlands). They consist of 96 round-bottom wells which are pre-coated with various concentrations of seven different clinically used antibiotics. The antimicrobial agents and their concentration ranges used in the test are given in Table 2. The susceptibility tests were performed according to CLSI guidelines [41]. Briefly, Campylobacter isolates were cultivated on Mueller-Hinton agar (Oxoid GmbH) supplied with 10 % bovine blood under microaerophilic conditions at 37 °C for 48 h. Bacterial colonies were suspended in NaCl solution (0.9 %) for matching turbidity of 0.5 McFarland units (Dr. Lange, CADAS 30 photometer, Berlin, Germany). One-hundred and fifty μl of the suspension were diluted in 10 ml Mueller-Hinton broth (Oxoid GmbH) resulting in a concentration range of 106 –107 colony forming units (cfu)/ml. Each well was dispensed with 100 µl of the suspension. The plates were sealed and incubated at 37 °C for 24 h under microaerophilic conditions. The results were obtained by reading either visually or photometrically (Tecan Deutschland GmbH, Crailsheim, Germany) using computer program easyWIN fitting (version V6.1, 2000). C. jejuni DSM 4688 (Deutsche Sammlung von Mikroorganismen und Zellkulturen GmbH, Braunschweig, Germany) and C. coli DSM 4689 were included in each batch of broth micro-dilution test for quality control.
Molecular biological detection of resistance determinants Erythromycin resistance
Point mutations at positions 2074 and 2075 in domain V of the 23S rRNA were confirmed as the most common mechanism for macrolide resistance in Campylobacter. The detection of point mutations was done by MAMAPCR assay as previously described [47].
Ciprofloxacin resistance
A single point mutation (Thr-86-Ile) in the quinolone resistance-determining region (QRDR) of gyrA was defined as source of high-level resistance to fluoroquinolones [48]. The MAMA-PCR was done to detect gyrA mutation in C. jejuni and C. coli isolates as described by [49, 50] with modified PCR cycling conditions.
Tetracycline resistance
tet(O) gene is strongly associated with tetracycline resistance in C. jejuni. Primer pair DMT1/DMT2 was chosen to detect this resistance determinant as described previously [51].
PCR conditions were identical with those described by El-Adawy et al. [52].
Table 2 MICs and resistance rate of Vietnamese C. jejuni isolates
Results
Campylobacter species identification
In total, 20 isolates suspected to be Campylobacter were cultivated (15 from chicken meat and 5 from pork) in Vietnam, saved cryo-conserved and transferred to Germany. However, only 9 isolates could be re-cultivated on Mueller-Hinton agar. Table 1 gives an overview of origin and species of cultivated Campylobacter isolates. Eight isolates belonged to C. jejuni and one isolate from chicken meat was identified as C. coli by multiplex PCR.
flaA-RFLP typing
Vietnamese Campylobacter isolates were characterized by flaA typing using the restriction enzyme DdeI (Fig. 1). The restriction profiles of C. jejuni yielded five different types.
MLST
Within the eight C. jejuni isolates, six different sequence types were identified (Table 3). Sequence type ST 5799 was found in C. jejuni isolates recovered from chicken and pork meat. C. jejuni sequence types ST 2837 and ST 4395 found in chicken belonged to the clonal complex ST-353. Four sequence types could be assigned to clonal complexes and two others (ST 5155 and ST 6736) were not assignable.
Campylobacter coli isolate 09CS0051 belonged to clonal complex ST-828.
Microarray DNA hybridization assay
The DNA microarray assay showed high significant genetic diversity among 8 C. jejuni isolates (Fig. 2). Isolates 09CS0040 and 09CS0046 were closely related and represented the same sequence type in MLST in which no assignment to an existing clonal complex was possible. Otherwise, isolates 09CS0049 and 09CS0066 belonged to the same sequence type (ST 5299) and CC (ST-443) proved to be different when considerably more gene loci were analyzed using the microarray. Likewise, isolates 09CS0043 and 09CS0047 showed large disparity in microarray analysis independent from their affiliation to ST-353 complex, whereby the different sequence types had to be considered.
Molecular biological detection of different virulence-associated and toxin genes
flaA, flaB, flhA and fliM as genes of the flagellar system of Campylobacter were found in all isolates, but fliY could not be detected by PCR (Table 1). The invasion-associated gene iamA was present in all C. jejuni isolates whereas ciaB was absent in one C. jejuni isolate. The gene virB11 was found in two isolates. All C. jejuni isolates carried cadF (an outer-membrane protein gene), cdtA, cdtB and cdtC-C (cytolethal distending toxin), docA (encoding a periplasmic cytochrome C peroxidase), and docB (encoding a methyl-accepting chemotaxis protein). Detection of docC (another methyl-accepting chemotaxis protein) was variable among the isolates (Table 1). It was detected in three out of eight isolates. wlaN (a beta-1,3 galactosyltransferase) which is responsible for a specific LOS structure was not identified in any of the isolates. In contrast, cgtB (another beta-1,3 galactosyltransferase gene) was found in five of eight C. jejuni isolates. The C. coli isolate 09CS0051 showed a difference to the C. jejuni strains. The invasion-associated genes ciaB and iamA were not detected by PCR. The presence of LOS biosynthesis genes was determined to characterize the Vietnamese Campylobacters. In all Campylobacter isolates pglB (encoding a putative oligosaccharyl transferase) was detected. Gene loci for putative galactosyltransferases (Cje1278, Cje1280, Cj1136, Cj1138, Cj1434c, Cj1438c, Cj1440c) were rarely found. Only Cje1280 was detected in two C. jejuni isolates.
Fig. 1 Agarose gel electrophoresis of flaA typing products of Vietnamese Campylobacter isolates digested with DdeI (M—100 bp DNA ladder)
Table 3 Results of MLST of Vietnamese Campylobacter isolates
Secretory genes secD and secF were detected in all Campylobacter isolates. Type-6 secretion system (T6SS), a novel class of protein translocation system, was identified over the haemolysin co-regulated protein (hsp) gene in all C. jejuni isolates.
The motility accessory factor (maf) family represents a new class of bacterial genes related to flagellar biosynthesis and phase variation. maf1 and maf4 were found in almost all isolates with the exceptions of 09CS0047 which lacked maf1 and 09CS0049 where maf4 could not be identified by PCR.
Antimicrobial susceptibility testing
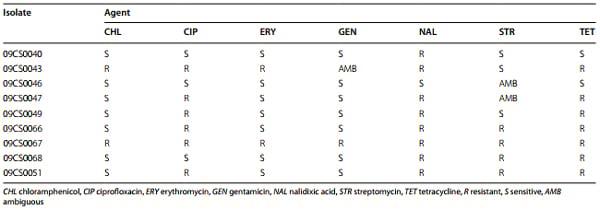
The results of antimicrobial susceptibility testing for seven antibiotic agents are given in Table 4. None of the Campylobacter isolates was fully susceptible to all investigated antibiotics. The C. jejuni isolates were highly resistant to ciprofloxacin, nalidixic acid, streptomycin and tetracycline with 62.5, 87.5, 62.5 and 75.0 % resistance, respectively. The resistance rate for chloramphenicol, erythromycin and streptomycin was low with 25.0 %. C. coli isolate 09CS0051 was resistant to ciprofloxacin, nalidixic acid, streptomycin and tetracycline.
Molecular biological assays for detection of ciprofloxacin, erythromycin, and tetracycline resistance determinants confirmed the results of phenotypic testing of antimicrobial resistance.
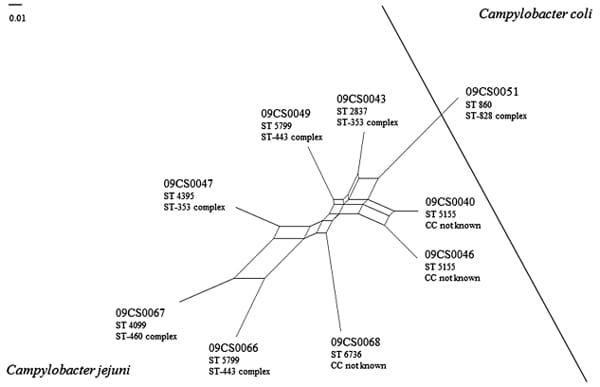
Fig. 2 SplitsTree analysis of Vietnamese Campylobacter isolates according to microarray investigation
Table 4 Resistance of Vietnamese Campylobacter isolates to various substances
Discussion
As in other countries, thermophilic Campylobacter are common bacterial agents in Vietnam which cause gastro-enteric illness in humans, especially in children [13, 15]. Meat and meat products serve as the main sources of human infections. Huong et al. showed that approximately 30 % of raw chicken samples from school and hospital canteens and retail markets in Hanoi city were contaminated with Campylobacter [17]. Other studies from different regions of Vietnam came to comparable results [20, 21]. Here, chicken and pork meat samples from a market in Hanoi were investigated for the presence of thermophilic Campylobacters. 13.3 % of the samples were Campylobacter positive. C. jejuni and C. coli were detected as contaminants of chicken and pork meat. In comparison, a report from Germany showed that 52.3 % of chicken carcasses in slaughterhouses and nearly 40 % of raw meat in retail were positive for these microorganisms [53]. It is possible that the lower percentage of Campylobacter findings in the meat samples from the Hanoi market is a result of slaughter procedures. In Vietnam the meat is not prepared in large slaughterhouses for retail as in Germany. Chicken and also pigs are slaughtered in low numbers and often directly on-site and the risk of contamination for example by generation of aerosols in slaughterhouses is smaller.
In this study, 20 Campylobacter isolates were obtained from meat samples in Vietnam. Unfortunately, after transfer to Germany only 9 isolates could be re-cultivated which were subject to further investigation. Eight of them were identified as C. jejuni and one as C. coli by mPCR. Several methods were used to type the isolates. As a rapid and simple method to illustrate heterogeneity within the C. jejuni isolates, flaA-RFLP typing was used. Four different strain types were detected by DdeI digestion of the amplified flaA gene. This enzyme was used because it showed the highest discriminatory power in former investigations [24] in comparison with AluI or Sau3AI. The digestion pattern of C. coli isolate 09CS0051 was completely different. Limitations of this typing method resulted from the use of only a very small part of the Campylobacter genome and difficulties in standardization of the analytical process. This complicates an inter-laboratory comparison of results between different laboratories.
Microarray analysis worked as a PCR-based comparative genomic fingerprinting (CGF) assay [54] and confirmed the heterogeneity of the C. jejuni isolates. An advantage of this method is the use of the whole genome data instead of only one or a few genes. The basis of this method is the detection of the presence or absence of several gene loci that are spread over the whole genome. SplitsTree analysis of the hybridization results showed high genetic diversity as no isolate is identical with another one. The C. coli isolate 09CS0051 was clearly distinct from the C. jejuni isolates. Additionally, sequence types and clonal complexes of the isolates determined by MLST are given in Fig. 2.
Both methods showed differences concerning the relatedness of different C. jejuni isolates among each other. Isolates 09CS0049 and 09CS0066 represented an identical sequence type and belonged to the same clonal complex but in microarray investigation they showed only poor relatedness. 09CS0043 and 09CS0047 were part of the clonal complex ST-353 but differed in the sequence type. Genetic relatedness based on microarray data was marginal. In contrast, two isolates (09CS0040 and 09CS0046) were found with identical sequence type and pattern in flaA typing after DdeI digestion and even microarray analysis showed a high degree of similarity.
The major advantage of MLST is the comparability of results independent from the laboratory and the local working conditions (technicians, machines etc.). The relatively high costs of this complex technique are outweighed by the hard facts that are obtained in the form of DNA sequences of seven house-keeping genes. In this study, six sequence types in the group of C. jejuni isolates were detected. These sequence types were compared with the database on the Campylobacter MLST Home Page (http://pubmlst.org/Campylobacter/). ST 2837 and ST 4395 belonged to clonal complex ST-353 whereby 09CS0047 (ST 4395) was identical with an isolate which was recovered from a stool sample of a hospital inpatient with gastroenteritis in Vietnam in 2010. 09CS0049 and 09CS0066 belonged to CC ST-443. Sequence type 5799 was previously isolated from human stool samples in Japan. Three isolates could not be assigned to any known clonal complex. The sequence type of isolate 09CS0068 had previously been discovered once in a human stool sample in Thailand, two others were not described yet. Isolate 09CS0067 represented sequence type 4099. This type belongs to the ST-460 complex and was previously identified in a human sample in Canada. C. coli isolate 09CS0051 belonged to sequence type 860 and ST-828. Identical isolates were found several times during the last two decades in Europe and the USA. Records from Asia are lacking until now. In summary, the investigated Vietnamese isolates in their majority seemed to represent strains typical for the Asian region. A route of infection of Campylobacter from meat to humans can be assumed.
The Campylobacter isolates were characterized regarding virulence factors associated with adhesion and invasion of host cells. All isolates harboured flagellin genes flaA, flaB, flhA and fliM. Similar observations have been reported previously [27, 55]. Molecular genetic approaches with defined mutants showed that flaA is essential for colonization [30]. The complex flagellum of Campylobacter species is encoded by two tandem-oriented flagellin genes (flaA and flaB). While the function of the flaA gene seems to be fully elucidated, there are many speculations about the function of the flaB gene, which may play a role in antigenic variation or influence the motility in various environmental conditions [56]. fliY, a gene of flagellar motor switch proteins, could not be detected.
The ciaB gene, coding for a Campylobacter invasion antigen, was present in most of the C. jejuni isolates. It was absent in 09CS0049 and C. coli 09CS0051. Another gene which is important in the invasion process of Campylobacter to host cells is iamA. It was detected in all C. jejuni isolates. Carvalho et al. described the detection of the iamA gene in 85 % of invasive C. jejuni but in non-invasive isolates it is rare [57]. Also the cadF gene was detected in all C. jejuni isolates. It encodes for an outer-membrane protein which mediates the binding of the bacteria to fibronectin [58]. Based on the results it can be concluded that these Vietnamese isolates represented invasive C. jejuni strains.
Cytolethal distending toxin causes direct DNA damage leading to induction of DNA damage checkpoint pathways [35]. The cdt gene cluster consists of 3 genes cdtA, cdtB and cdtC. The cdt genes were shown to be conserved among different Campylobacter strains [59]. Bang et al. observed that the presence of these genes in isolates from different sources exceeds 90 % [27]. In all Vietnamese C. jejuni strains isolated from chicken and pork meat the complete cdt gene cluster was observed. Rozynek et al. obtained results for C. jejuni strains isolated from children with diarrhea and found that cdtA, cdtB and cdtC were present in 98.4, 97.0 and 98.0 % of all isolates, respectively [60]. However, cdtC was not detected in C. coli isolate 09CS0051 from chicken meat which was in agreement with a previous study [60]. On the other hand, a similar frequency of cdt genes and the cdt gene cluster was observed in dog and chicken isolates [55]. In this study all investigated isolates harboured the cdtB gene. It is indeed generally accepted that the cdtB genes are widespread amongst poultry and cattle as well as in human isolates in Denmark, Japan, Poland, and Belgium [27, 60– 62]. However, low percentages of occurrence of cdtB have been reported in humans (28 %) and chickens (20 %) in India, which could be due to genetic reasons or variations in the isolates from different geographic areas [63].
Only a minority of C. jejuni isolates gave positive PCR results for virB11 encoding a putative component of a type IV secretion system. It is located in the pVir plasmid and could be involved in virulence [38]. The 25.0 % prevalence of the virB11 gene in C. jejuni isolates in this study is higher than 10.3 % in human isolates reported by Bacon et al., but much lower than in pig isolates (35.7 %). Until now, the role of the protein encoded by the virB11 gene in the invasion and colonization process of eukaryotic cells by Campylobacter species could not be elucidated.
Macrolides, quinolones and tetracycline are among the common antimicrobials recommended for testing, because they can be of therapeutic relevance in severe cases of infection. High levels of resistance of Campylobacter to tetracycline and ciprofloxacin were frequently reported but resistance to erythromycin and gentamicin remained low.
The antimicrobial susceptibility profiles among the Vietnamese isolates were analyzed based on the guidelines of CLSI (2008) [41]. In this study standardization of the protocol for the commercially available broth microdilution test as a method for the determination of the minimum inhibitory concentration (MIC) of antibiotics was done [52]. All isolates were sensitive to gentamicin and most of isolates (88.8 %) were sensitive to chloramphenicol, erythromycin and streptomycin. Similar results were reported in several previous studies [4, 42, 64–68]. In contrast to our findings, a previous study reported high resistance to streptomycin with 60.0 % [64]. The resistance rate to ciprofloxacin was 66.7 % which is in agreement with a previous study showing high resistance [51, 64], but in contrast to another study with only 9.5 % [69]. Resistance to nalidixic acid was 88.9 % which is similar to several aforementioned reports [51, 64, 68, 70]. However, other studies found either low resistance [71, 72] or none at all [65]. Resistances to tetracycline was higher (77.8 %) than previously reported (32.0 %) [72], but it was lower than in isolates recovered from conventionally grown turkeys [68].
The gene loci responsible for antibiotic resistance were detected in all resistant isolates to ciprofloxacin and erythromycin and 66.7 % of resistant isolates to tetracycline. Ciprofloxacin resistance among C. jejuni and C. coli isolates was conferred by threonine-to-isoleucine mutation of amino acid 86 of the gyrA protein (Thr-86- Ile), a finding that is in agreement with other previous studies [73–76]. Tetracycline resistance was attributed to the presence of the tet(O) gene [51]. All resistant isolates in this study were carrying tet(O); none of the susceptible isolates gave a positive result using specific PCR.
Conclusions
To the best of our knowledge we present here the first detailed characterization of Vietnamese Campylobacter isolates regarding genetic diversity, virulence-associated genes and antibiotic susceptibility. The limitation of our study is the small number of isolates. Further studies are needed to improve our knowledge about the epidemiology and relevance of Campylobacter for human health in Vietnam.
This article was originally published in Gut Pathogens (2016) 8:19 DOI 10.1186/s13099-016-0100-x. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/). References
1. Allos BM. Campylobacter jejuni infections: update on emerging issues and trends. Clin Infect Dis. 2001;32:1201–6.
2. Ketley JM. Pathogenesis of enteric infection by Campylobacter. Microbiol- ogy. 1997;143:5–21.
3. Dingle KE, Colles FM, Wareing DR, Ure R, Fox AJ, Bolton FE, et al. Multilo- cus sequence typing system for Campylobacter jejuni. J Clin Microbiol. 2001;39:14–23.
4. Hafez HM. Bacterial contaminations and risks from poultry meat and eggs. Arch Geflüglk. 2003;67:146–52.
5. EFSA. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2012. EFSA J. 2015;12:3547.
6. Weijtens MJ, van der Plas J, Bijker PG, Urlings HA, Koster D, van Logtestijn JG, et al. The transmission of Campylobacter in piggeries; an epidemio- logical study. J Appl Microbiol. 1997;83:693–8.
7. Gill CO, Badoni M, McGinnis JC. Assessment of the adequacy of cleaning of equipment used for breaking beef carcasses. Int J Food Microbiol. 1999;46:1–8.
8. Gill CO, McGinnis JC, Bryant J. Contamination of beef chucks with Escherichia coli during carcass breaking. J Food Prot. 2001;64:1824–7.
9. Duffy EA, Belk KE, Sofos JN, Bellinger GR, Pape A, Smith GC. Extent of microbial contamination in United States pork retail products. J Food Prot. 2001;64:172–8.
10. Pearce RA, Wallace FM, Call JE, Dudley RL, Oser A, Yoder L, et al. Prevalence of Campylobacter within a swine slaughter and processing facility. J Food Prot. 2003;66:1550–6.
11. Coker RJ, Hunter BM, Rudge JW, Liverani M, Hanvoravongchai P. Emerging infectious diseases in Southeast Asia: regional challenges to control. Lancet. 2011;377:599–609.
12. Bodhidatta L, Lan NT, Hien BT, Lai NV, Srijan A, Serichantalergs O, et al. Rotavirus disease in young children from Hanoi, Vietnam. Pediatr Infect Dis J. 2007;26:325–8.
13. My PV, Thompson C, Phuc HL, Tuyet PT, Vinh H, Hoang NV, et al. Endemic norovirus infections in children, Ho Chi Minh City, Vietnam, 2009–2010. Emerg Infect Dis. 2013;19:977–80.
14. Trang DT, Hien BTT, Molbak K, Cam PD, Dalsgaard A. Epidemiology and aetiology of diarrhoeal diseases in adults engaged in wastewater-fed agriculture and agriculture in Hanoi, Vietnam. Trop Med Int Health. 2007;12(Suppl. 2):23–33.
15. Isenbarger DW, Hien BT, Ha HT, Ha TT, Bodhidatta L, Pang LW, et al. Prospective study of the incidence of diarrhoea and prevalence of bacte- rial pathogens in a cohort of Vietnamese children along the Red River. Epidemiol Infect. 2001;127:229–36.
16. Carrique-Mas JJ, Bryant JE. A review of foodborne bacterial and parasitic zoonoses in Vietnam. EcoHealth. 2013;10:465–89.
17. Huong LQ, Hanh TT, Cam PD, Be NT. Study on the prevalence of Campylobacter spp. from chicken meat in Hanoi, Vietnam. Ann N Y Acad Sci. 2006;1081:273–5.
18. Ha TA, Pham T. Study of Salmonella, Campylobacter, and Escherichia coli contamination in raw food available in factories, schools, and hospital canteens in Hanoi, Vietnam. Ann N Y Acad Sci. 2006;1081:262–5.
19. Schwan P. Prevalence and antibiotic resistance of Campylobacter spp. in poultry and raw meat in the Can Tho Province, Vietnam. Examensarbete. 2010;49. ISSN 1652-8697. http://epsilon.slu.se.
20. Garin B, Gouali M, Wouafo M, Perchec AM, Tham MT, Ravaonindrina N, et al. Prevalence, quantification and antimicrobial resistance of Campylobacter spp. on chicken neck-skins at points of slaughter in 5 major cities located on 4 continents. Int J Food Microbiol. 2012;157:102–7.
21. Carrique-Mas JJ, Bryant JE, Cuong NV, Hoang NV, Campbell J, Hoang NV, et al. An epidemiological investigation of Campylobacter in pig and poultry farms in the Mekong delta of Vietnam. Epidemiol Infect. 2014;142:1425–36.
22. Bao VN, Fries R, Zessin KH, Kyule MN, Pinthong R, Baumann MPO. Salmonella and Campylobacter in broiler carcasses in Vietnam. In: Proceedings of the 11th international symposium veterinary epidemiology and economics. 2006. www.sciquest.org.nz.
23. Eberle KN, Kiess AS. Phenotypic and genotypic methods for typ- ing Campylobacter jejuni and Campylobacter coli in poultry. Poult Sci. 2012;91:255–64.
24. El-Adawy H, Hotzel H, Tomaso H, Neubauer H, Taboada EN, Ehricht R, et al. Detection of genetic diversity in Campylobacter jejuni isolated from a commercial turkey flock using flaA typing, MLST analysis and microarray assay. PLoS One. 2013;8:e51582.
25. El-Adawy H, Hotzel H, Tomaso H, Neubauer H, Hafez HM. Elucidation of colonization time and prevalence of thermophilic Campylobacter species during turkey rearing using multiplex polymerase chain reaction. Poult Sci. 2012;91:454–9.
26. Parkhill J, Wren BW, Mungall K, Ketley JM, Churcher C, Basham D, et al. The genome sequence of the food-borne pathogen Campylobacter jejuni reveals hypervariable sequences. Nature. 2000;403:665–8.
27. Bang DD, Nielsen EM, Scheutz F, Pedersen K, Handberg K, Madsen M. PCR detection of seven virulence and toxin genes of Campylobacter jejuni and Campylobacter coli isolates from Danish pigs and cattle and cytolethal distending toxin production of the isolates. J Appl Microbiol. 2003;94:1003–14.
28. Hänel I, Borrmann E, Müller J, Alter T. Relationships between bacte- rial genotypes and in vitro virulence properties of Campylobacter jejuni and Campylobacter coli isolated from turkeys. J Appl Microbiol. 2007;102:433–41.
29. Müller J, Schulze F, Müller W, Hänel I. PCR detection of virulenceassociated genes in Campylobacter jejuni strains with differential ability to invade Caco-2 cells and to colonize the chick gut. Vet Microbiol. 2006;113:123–9.
30. Nachamkin I, Yang XH, Stern NJ. Role of Campylobacter jejuni flagella as colonization factors for three-day-old chicks: analysis with flagellar mutants. Appl Environ Microbiol. 1993;59:1269–73.
31. Hänel I, Schulze F, Hotzel H, Schubert E. Detection and characterization of two cytotoxins produced by Campylobacter jejuni strains. Zentralbl Bakteriol. 1998;288:131–43.
32. Whitehouse CA, Balbo PB, Pesci EC, Cottle DL, Mirabito PM, Pickett CL. Campylobacter jejuni cytolethal distending toxin causes a G2-phase cell cycle block. Infect Immun. 1998;66:1934–40.
33. Pickett CL, Pesci EC, Cottle DL, Russell G, Erdem AN, Zeytin H. Preva- lence of cytolethal distending toxin production in Campylobacter jejuni and relatedness of Campylobacter sp. cdtB gene. Infect Immun. 1996;64:2070–8.
34. Jain D, Prasad KN, Sinha S, Husain N. Differences in virulence attributes between cytolethal distending toxin positive and negative Campylobacter jejuni strains. J Med Microbiol. 2008;57:267–72.
35. Lee RB, Hassane DC, Cottle DL, Pickett CL. Interactions of Campylobacter jejuni cytolethal distending toxin subunits CdtA and CdtC with HeLa cells. Infect Immun. 2003;71:4883–90.
36. Lara-Tejero M, Galán JE. CdtA, CdtB, and CdtC form a tripartite complex that is required for cytolethal distending toxin activity. Infect Immun. 2001;69:4358–65.
37. Bacon DJ, Alm RA, Hu L, Hickey TE, Ewing CP, Batchelor RA, et al. DNA sequence and mutational analyses of the pVir plasmid of Campylobacter jejuni 81-176. Infect Immun. 2002;70:6242–50.
38. Bacon DJ, Alm RA, Burr DH, Hu L, Kopecko DJ, Ewing CP, et al. Involve- ment of a plasmid in virulence of Campylobacter jejuni 81-176. Infect Immunol. 2000;68:4384–90.
39. Harrison JW, Dung TT, Siddiqui F, Korbrisate S, Bukhari H, Tra MP, et al. Identification of possible virulence marker from Campylobacter jejuni isolates. Emerg Infect Dis. 2014;20:1026–9.
40. Isenbarger DW, Hoge CW, Srijan A, Pitarangsi C, Vithayasai N, Bodhidatta L, et al. Comparative antibiotic resistance of diarrheal pathogens from Vietnam and Thailand, 1996–1999. Emerg Infect Dis. 2002;8:175–80.
41. Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial disk and dilution susceptibility tests for bacterial isolates from animals; approved standards. 3rd ed. M31-A3, vol. 28, no. 8; 2008.
42. Luber P, Bartelt E, Genschow E, Wagner J, Hahn H. Comparison of broth microdilution, E test, and agar dilution methods for antibiotic suscep- tibility testing of Campylobacter jejuni and Campylobacter coli. J Clin Microbiol. 2003;41:1062–8.
43. EUCAST. Breakpoint tables for interpretation of MICs and zone diameters. Version 3.1. 2013. http://www.eucast.org.
44. International Standards Organization: [ISO] 10272-1. Microbiology of food and animal feeding stuffs—horizontal method for detection and enumeration of Campylobacter spp. Part 1: detection method. 2006.
45. Taboada EN, Luebbert CC, Nash JH. Studying bacterial genome dynamics using microarray-based comparative genomic hybridization. Methods Mol Biol. 2007;396:223–53.
46. Müller J, Meyer B, Hänel I, Hotzel H. Comparison of lipooligosaccharide biosynthesis genes of Campylobacter jejuni strains with varying abilities to colonize the chicken gut and to invade Caco-2 cells. J Med Microbiol. 2007;56:1589–94.
47. Alonso R, Mateo E, Churruca E, Martinez I, Girbau C, Fernandez-Astorga A. MAMA-PCR assay for the detection of point mutations associated with high-level erythromycin resistance in Campylobacter jejuni and Campylobacter coli strains. J Microbiol Methods. 2005;63:99–103.
48. Payot S, Bolla JM, Corcoran D, Fanning S, Megraud F, Zhang Q. Mecha- nisms of fluoroquinolone and macrolide resistance in Campylobacter spp. Microbes Infect. 2006;8:1967–71.
49. Zirnstein G, Li Y, Swaminathan B, Angulo F. Ciprofloxacin resistance in Campylobacter jejuni isolates: detection of gyrA resistance mutations by mismatch amplification mutation assay PCR and DNA sequence analysis. J Clin Microbiol. 1999;37:3276–80.
50. Zirnstein G, Helsel L, Li Y, Swaminathan B, Besser J. Characterization of gyrA mutations associated with fluoroquinolone resistance in Campylobacter coli by DNA sequence analysis and MAMA PCR. FEMS Microbiol Lett. 2000;190:1–7.
51. Mazi W, Senok A, Al-Mahmeed A, Arzese A, Bindayna K, Botta G. Trends in antibiotic sensitivity pattern and molecular detection of tet(O)-mediated tetracycline resistance in Campylobacter jejuni isolates from human and poultry sources. Jpn J Infect Dis. 2008;61:82–4.
52. El-Adawy H, Hotzel H, Düpre S, Tomaso H, Neubauer H, Hafez HM. Deter- mination of antimicrobial sensitivities of Campylobacter jejuni isolated from commercial turkey farms in Germany. Avian Dis. 2012;56:685–92.
53. BVL: Berichte zur Lebensmittelsicherheit 2013, Zoonosen-Monitoring. ISBN 978-3-319-15379-7, 2015; 22.
54. Buchanan CJ, Yoshida C, Kruczkiewicz P, Mutschall SK, Ross SI, Chu J, et al. High-resolution comparative genomic fingerprinting (CGF) of C. jejuni using ArrayTube™ miniaturized diagnostic microarrays. In: Proceedings of the 16th internnational workshop on Campylobacter, Helicobacter, and related organisms (CHRO 2011) 2011, Vancouver, Canada, p. 164.
55. Krutkiewicz A, Klimuszko D. Genotyping and PCR detection of potential virulence genes in Campylobacter jejuni and Campylobacter coli isolates from different sources in Poland. Folia Microbiol (Praha). 2010;55:167–75.
56. Wassenaar TM, Bleumink-Pluym NM, Newell DG, Nuijten PJ, van der Zeijst BA. Differential flagellin expression in a flaA flaB+ mutant of Campylobacter jejuni. Infect Immun. 1994;62:3901–6.
57. Carvalho ACT, Ruiz-Palacios GM, Ramos-Cervantes P, Cervantes LE, Jiang X, Pickering LK. Molecular characterization of invasive and noninvasive Campylobacter jejuni and Campylobacter coli isolates. J Clin Microbiol. 2001;39:1353–9.
58. Konkel ME, Garvis SG, Tipton SL, Anderson DE Jr, Cieplak W Jr. Identifica- tion and molecular cloning of a gene encoding a fibronectin-binding protein (CadF) from Campylobacter jejuni. Mol Microbiol. 1997;24:953–63.
59. Fouts DE, Mongodin EF, Mandrell RE, Miller WG, Rasko DA, Ravel J, et al. Major structural differences and novel potential virulence mecha- nisms from the genomes of multiple Campylobacter species. PLoS Biol. 2005;3:e15.
60. Rozynek E, Dzierzanowska-Fangrat K, Jozwiak P, Popowski J, Korsak D, Dzi- erzanowska D. Prevalence of potential virulence markers in Polish Campylobacter jejuni and Campylobacter coli isolates obtained from hospitalized children and from chicken carcasses. J Med Microbiol. 2005;54:615–9.
61. González-Hein G, Huaracan B, Garcia P, Figueroa G. Prevalence of virulence genes in strains of Campylobacter jejuni isolated from human, bovine and broiler. Braz J Microbiol. 2014;44:1223–9.
62. Van Deun K, Haesebrouck F, Heyndrickx M, Favoreel H, Dewulf J, Ceelen L, et al. Virulence properties of Campylobacter jejuni isolates of poultry and human origin. J Med Microbiol. 2007;56:1284–9.
63. Rizal A, Kumar A, Vidyarthi AS. Prevalence of pathogenic genes in Campylobacter jejuni isolated from poultry and human. Internet J Food Saf. 2010;12:29–34.
64. Gu W, Siletzky RM, Wright S, Islam M, Kathariou S. Antimicrobial susceptibility profiles and strain type diversity of Campylobacter jejuni isolates from turkeys in eastern North Carolina. Appl Environ Microbiol. 2009;75:474–82.
65. Miflin JK, Templeton JM, Blackall PJ. Antibiotic resistance in Campylobacter jejuni and Campylobacter coli isolated from poultry in the South-East Queensland region. J Antimicrob Chemother. 2007;59:775–8.
66. Luber P, Wagner J, Hahn H, Bartelt E. Antimicrobial resistance in Campylobacter jejuni and Campylobacter coli strains isolated in 1991 and 2001–2002 from poultry and humans in Berlin, Germany. Antimicrob Agents Chemother. 2003;47:3825–30.
67. Luangtongkum T, Jeon B, Han J, Plummer P, Logue CM, Zhang Q. Antibiotic resistance in Campylobacter: emergence, transmission and persistence. Future Microbiol. 2009;4:189–200.
68. Luangtongkum T, Morishita TY, Ison AJ, Huang S, McDermott PF, Zhang Q. Effect of conventional and organic production practices on the preva- lence and antimicrobial resistance of Campylobacter spp. in poultry. Appl Environ Microbiol. 2006;72:3600–7.
69. Hariharan H, Sharma S, Chikweto A, Matthew V, DeAllie C. Antimicrobial drug resistance as determined by the E-test in Campylobacter jejuni, C. coli, and C. lari isolates from the ceca of broiler and layer chickens in Grenada. Comp Immunol Microbiol Infect Dis. 2009;32:21–8.
70. Luangtongkum T, Moroshita TY, El-Tayeb AB, Ison AJ, Zhang Q. Com- parison of antimicrobial susceptibility testing of Campylobacter spp. by the agar dilution and the agar disk diffusion methods. J Clin Microbiol. 2007;45:590–4.
71. Andersen SR, Saadbye P, Shukri NM, Rosenquist H, Nielsen NL, Boel J. Anti- microbial resistance among Campylobacter jejuni isolated from raw poul- try meat at retail level in Denmark. Int J Food Microbiol. 2006;107:250–5.
72. Andersen SR, Shukri NM, Boel J, Saadbye P. Metronidazole resistance in Campylobacter jejuni from poultry meat. J Food Prot. 2006;69:932–4.
73. Charvalos E, Peteinaki E, Spyridaki I, Manetas S, Tselentis Y. Detection of ciprofloxacin resistance mutations in Campylobacter jejuni gyrA by non- radioisotopic single-strand conformation polymorphism and direct DNA sequencing. J Clin Lab Anal. 1996;10:129–33.
74. Ruiz J, Goni P, Marco F, Gallardo F, Mirelis B, Jimenez De Anta T, et al. Increased resistance to quinolones in Campylobacter jejuni: a genetic analysis of gyrA gene mutations in quinolone-resistant clinical isolates. Microbiol Immunol. 1998;42:223–6.
75. Wang Y, Huang WM, Taylor DE. Cloning and nucleotide sequence of the Campylobacter jejuni gyrA gene and characterization of quinolone resist- ance mutations. Antimicrob Agents Chemother. 1993;37:457–63.
76. Wardak S, Szych J, Cieslik A. PCR-restriction fragment length polymor- phism assay (PCR-RFLP) as an useful tool for detection of mutation in gyrA gene at 86-THR position associated with fluoroquinolone resistance in Campylobacter jejuni. Med Dosw Mikrobiol. 2005;57:295–301.
77. Hänel I, Borrmann E, Müller J, Müller W, Pauly B, Liebler-Tenorio EM, et al. Genomic and phenotypic changes of Campylobacter jejuni strains after passage of the chicken gut. Vet Microbiol. 2009;136:121–9.
78. Karlyshev AV, Linton D, Gregson NA, Wren BW. A novel paralogous gene family involved in phase-variable flagella-mediated motility in Campylobacter jejuni. Microbiology. 2002;148:473–80.







.jpg&w=3840&q=75)