Introduction
Aspergillosis is frequent, economically important, fungal disease of different avian and mammal species caused by fungi of the genus Aspergillus. The disease is characterized by inflammatory changes in the respiratory system and sometimes has generalized onset when more organ systems are affected. Clinical signs of aspergillosis depend upon which organ or system organ are involved and whether infection is localized or generalized. Pathomorphological substrate of this disease is mycotic granuloma (aspergiloma) which is commonly found in lungs and air saccs. Numerous reports have described encephalitic or meninogoencephalitic aspergillosis in turkey poults, ducklings and chickens (Kneževic and Matejic, 1996; Ivetic et al., 2003; Ozmen and Dorrestein, 2004; Singh et al., 2009, Kureljušic et al., 2011a). Olias et al. (2010) have described a case of articular aspergillosis of hip joints in turkeys. Comparative experimental study of the susceptibility of different species of poultry to infection with Aspergillus fumigatus has shown that turkeys are more susceptible than chickens. Primarily, there are two forms of aspergillosis in poultry: acute and chronic. Acute aspergilossis is usually characterized by severe outbreaks in young birds and high morbidity and high mortality. The mortality rate may range from 70 to 90%. Chronic aspergillosis occurs in adult breeder birds (particulary turkeys). It is very difficult to diagnosticate this infection by clinical examination because in many cases more organ systems are affected. It is known from literature that the diagnosis of fungal infections of poultry often be placed only after histopathological examination of organs of infected poultry (Richard et al., 1984, Saif, 2008). For diagnostic of mycotic infection is necessary to use more methods including clinical examination of flock, macroscopic examination of carcasses, isolation of pathogens, as well as histochemical methods for detection tissue lesions and fungal elements. Histochemical methods which can be used are routine hematoxylin-eosin staining method, periodic acid-Schiff (PAS) and specific staining method for fungi Grocott methenamine silver method (GMS) (Ozmen and Dorrestein, 2004). There is also a possibility to use immunohistochemical methods for direct in situ evidence of etiological agent (Jensen et al., 1997).
Materials and methods
In this paper, we examined the carcasses of 12 turkey poults, three weeks old which originated from one farm in Serbia. The mortality rate in the flock was 7,2%. Clinical examination of the flock revealed moderate depression of many turkey poults and nervous symptoms which were expressed in the form of ataxia, torticollis, paresis and paralysis of legs and wings. Food consumption was reduced leading to reduced weight gain. After necropsy, for histopathological and mycological examination samples of lung and brain tissue were taken. Samples for histopathological analysys were fixed in 10% buffered formalin, routinely processed and embedded in paraffin blocks. Paraffin sections about 5 μm were stained with hematoxylin-eosin (HE), periodic acid Schiff (PAS) and Grocott methenamine silver (GMS) methods. Samples for mycological examination were inoculated onto Sabouraud dextrose agar and incubated at temperature of 250C under aerobic conditions for isolation of infectious agents. To avoid bacterial contamination in the substrate was added 20 IU/ml penicillin G and 40 mg/ml streptomycin sulfate. Macroscopic and microscopic examination of colonies was performed according to Quinn (2002). Samples of feed and litter were also taken for mycological analysis.
Results and discussion
External examination of turkey poults carcasses indicates less feathering of the skin especially in the region of chest.
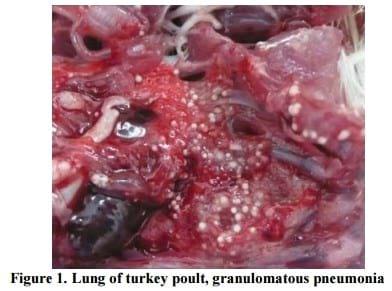
In ten out of twelve necropsied turkey poults granulomatous pneumonia with multiple yellowish-white granulomas, one to three millimeters in diameter were found (Figure 1).
In addition, multifocal granulomatous airsaculitis of thoracic and abdominal air sacs was also observed. In advanced cases of aspergillosis, the organism can sporulate on the surface of the caseous lesions and on the walls of the thickened air sacs, as evidenced by visible greenish-gray mold growth. That was also found in one case of aspegillosis in black swan (Kureljušic et al., 2011b), but in this case there were no such lesions. In 3 out of twelve necropsied turkey poults solitary yellowish-white granuloma of irregular shape, three to five millimeters in diameter on sagital section of the cerebrum or cerebellum were found (Figure 2).
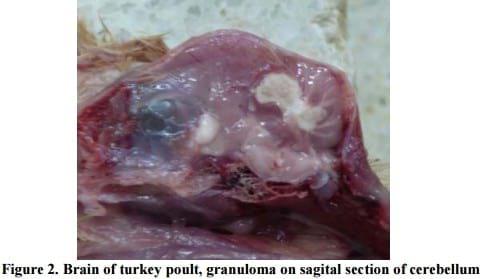
In two out of twelve cases granulomas were found on the cranial part of the cerebrum, and in one case granuloma was on the central part of cerebellum. Data from literature indicate that granulomas caused by Aspergillus fumigatus (aspergilomas) usually occur in the lungs and air sacs although there are data on the occurrence of aspergilomas in the cerebellum, only in turkey poults (Schulz, 1991; Ozmen and Dorrestein, 2004; Saif 2008, Kureljušic et al, 2011a). Similar to our observations, the granulomas occur either in cerebrum or the cerebellum (Saif, 2008). Rarely, aspergillomas occur simultaneously in both parts of brain (Saif, 2008). In addition to this finding, in turkey poults that were necropsied, hyperemia of cerebral blood vessels was found. In other organs there were no macroscopically visible changes.
Mycological examination revealed that the ethiological agent is fungus Aspergillus fumigatus. Growth in the medium was observed after 24 hours, and after four days, grown colonies were white and about 2 cm in diameter. By the seventh day the colony diameter increased to 3.5 cm, and there was a color change in the central part of the colonies from blue-green to the gray-green, while the edges remained white (Figure 3). Based on macroscopic and microscopic characteristics of colonies, the culture was identified as Aspergillus fumigatus.

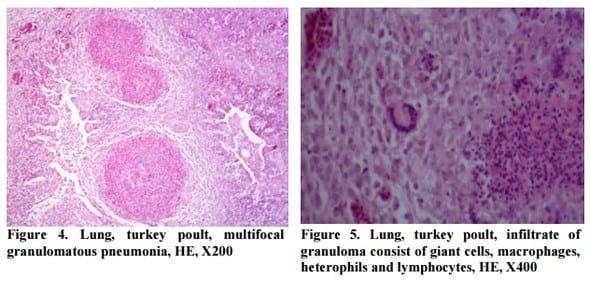
For the evaluation of histopathological changes and demonstration of fungal hyphae, three stain methods were used: haematoxylin-eosin (HE), Grocott methenamine silver and PAS method. Histological finding in the lung and in the brain was the same. Granulomatous reactions with central caseous necrosis were observed in the HE stained slides. There was surrounding region of proliferation including giant cells, macrophages, heterophils and lymphocytes and outer capsule of connective tissue (Figure 4, Figure 5).
The fungal hyphae were either hardly visible or not visible in HE slides, while septed and arborized hyphae were easily demonstrated in all samples by Grocott methenamine silver and PAS stain, predominantly in central parts of granuloma (Figure 6, Figure 7).
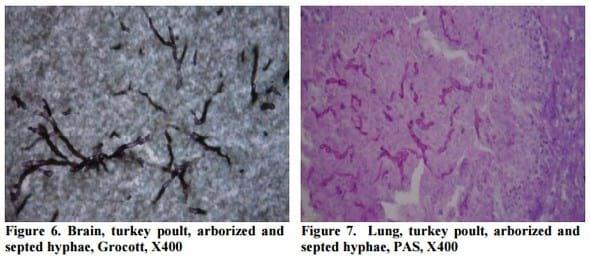
In some cases, in the sections of brain and lung stained by HE method, granulomatous changes were characterized only by presence of smaller or larger number of necrotic foci, which are infiltrated with macrophages, giant cells and heterophil granulocytes. These changes are described in the literature as early initial lesions (Jensen et al., 1997). In three out of twelve cases in HE stained sections, fungal hyphae were not apparent. Grocott and PAS methods in all twelve cases revealed septed and arborized hyphae. Thus, the specific staining methods revealed fungal hyphae even if minimal histopathological changes occurs. Based on this finding it can be assumed that the occurrence of lesions in the lung and brain depends not only on the presence of infectious agents, but also on the reaction of the immune system of the host. PAS and Grocott method are described in the literature as a very reliable method for direct visualization of the different fungal elements in tissues (Ozmen and Dorrestein, 2004; Singh et al., 2009).
Mycological analysys of litter revealed Aspergillus fumigatus while samples of feed were negative for this microorganism. It is known from literature that contaminated litter is often the source of Aspergillus fumigatus (Saif 2008).
Conclusion
Microscopic finding of typical mycotic granulomas in the lung and brain with central caseous necrosis and fungal hyphae surrounded by macrophages, giant cells, lymphocytes, heterophils and capsule of connective tissue pointed to aspergillosis. For diagnostic of mycotic infection is necessary to use more methods including clinical examination of flock, macroscopic examination of carcasses, isolation of pathogens, as well as histochemical methods for detection of tissue lesions and fungal elements.
Acknowledgment: This work was financed by the Ministry of Education and Science, Republic of Serbia, project number III 46009
References
IVETIC V., VALTER D., PAVLOVIC I., MILJKOVIC B., MASLIC-STRIŽAK D., ILIC Ž., SAVIC B., STANOJEVIC S., SPALEVIC LJ.(2003): Atlas bolesti živine, Naucni institut za veterinarstvo Srbije, Beograd
JENSEN H.E., CHRISTENSEN J.P., BISGAARD M., NIELSEN O.L.(1997): Immunohistochemistry for diagnosis of aspergillosis in turkey poults, Avian Pathology 26, 5-18
KNEŽEVIC N., MATEJIC M.(1996): Bolesti pernate živine, Veterinarska komora Srbije, Beograd
KURELJUŠIC B., SAVIC B., PRODANOVIC R., ÐEKIC J., ADAMOV V., JAKIC-DIMIC D., MILJKOVIC B., RADANOVIC O., IVETIC V.(2011A): Primena razlicitih histohemijskih metoda u dijagnostici aspergiloze mozga kod curica, Veterinarski glasnik 65, 1-2, 43-50
KURELJUŠIC B., PRODANOVIC R., KURELJUŠIC J., JEZDIMIROVIC N., RADANOVIC O. (2011B): Prikaz slucaja aspergiloze kod crnog labuda (Cygnus atratus), Živinarstvo 1/2, 21-26
OLIAS P., HAUCK R., WINDHAUS H., VAN DER GRINTEN E., GRUBER A.D., HAFEZ H.M. (2010): Articular aspergillosis of hip joints in turkeys, Avian Disease 54, 1098-1101
OZMEN O., DORRESTEIN M.G.(2004): Observations of aspergillosis in the brains of turkey poults using different histopathological staining techniques, Biotechnic and Histochemistry 79, 2, 95-99
QUINN P.J., MARKEY B.K., CARTER M.E., DONNELY W.J.C., LEONARD F.C.(2002): Veterinary microbiology and microbial disease, Blackwell Science Ltd., Iowa State University Press
RICHARD J.L., THURSTON J.R., PEDEN W.M., PINELLO C.(1984): Recent studies on aspergillosis in turkey poults, Mycopathologia 87, 3-11
SAIF Y.M.(2008): Diseases of poultry, twelfth edition, Blackwell Publishing, Ames, Iowa, USA SCHULZ L.CL.(1991): Pathologie der Haustiere, Teil 1, Gustav Fischer Verlag, Jena
SINGH S., BORAH M.K., SHARMA D.K., JOSHI G.D., GOGOI R.(2009): Aspergillosis in turkey poults, Canadian Journal of Veterinary Pathology 33, 2, 220-221