Inhibitory Effect of PgAFP and Protective Cultures on Aspergillus parasiticus Growth and Aflatoxins Production on Dry-Fermented Sausage and Cheese
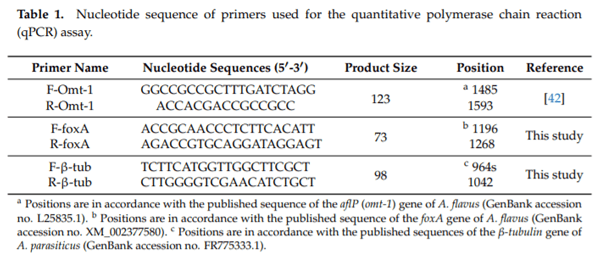
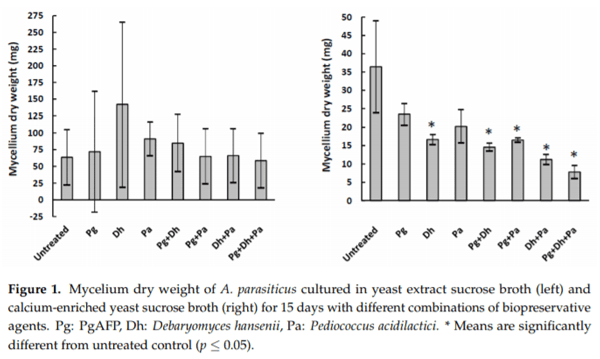
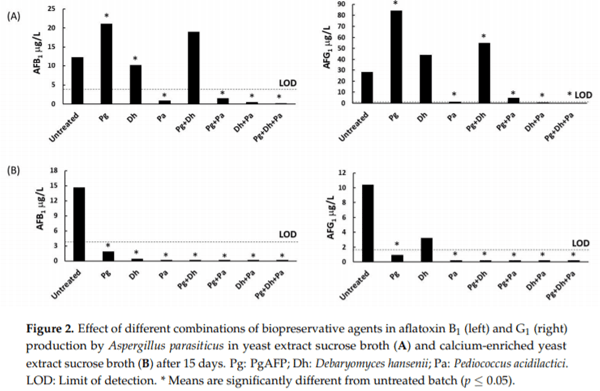
Aflatoxigenic molds can grow and produce aflatoxins on dry-fermented meat and cheese. The small, basic, cysteine-rich antifungal protein PgAFP displays a time-limited inhibitory ability against unwanted molds by increasing reactive oxygen species (ROS), which can lead to increased aflatoxin production. However, calcium abolishes the inhibitory effect of PgAFP on certain Aspergillus spp. To maximize the antifungal effect, this protein may be combined with protective cultures. Yeasts and lactic acid bacteria may counteract the impact of calcium on PgAFP fungal inhibition. The objective of this work was to study the effect of PgAFP and different combined treatments with Debaryomyces hansenii and/or Pediococcus acidilactici against growth of and aflatoxin production by an aflatoxigenic strain of Aspergillus parasiticus in both culture media and dry-fermented foods with low or high calcium levels. Aflatoxins production was increased by PgAFP but dramatically reduced by P. acidilactici in low calcium culture medium, whereas in the Ca-enriched culture medium, all treatments tested led to low aflatoxins levels. To study whether PgAFP and the protective microorganisms interfere with ROS and aflatoxin production, the relative expression of genes foxA, which is involved in peroxisomal β-oxidation, and aflP, which is required for aflatoxin biosynthesis, were evaluated. The aflatoxin overproduction induced by PgAFP seems not to be linked to peroxisomal β-oxidation. The combination of PgAFP and D. hansenii provided a successful inhibitory effect on A. parasiticus growth as well as on aflatoxin production on sliced dry-fermented sausage and cheese ripened up to 15 days, whereas P. acidilactici did not further enhance the protective effect of the two former agents. Therefore, the combined treatment of PgAFP and D. hansenii seems to provide a promising protective mean against aflatoxin-producing A. parasiticus on dry-fermented foods.
Keywords: Aflatoxin; Aspergillus parasiticus; antifungal protein; dry-fermented sausage; cheese; biopreservation; Debaryomyces hansenii
1. Alapont, C.; López-Mendoza, M.C.; Gil, J.V.; Martínez-Culebras, P.V. Mycobiota and toxigenic Penicillium species on two Spanish dry-cured ham manufacturing plants. Food Addit. Contam. A 2014, 31, 93–104. [CrossRef] [PubMed]
2. Blanco, J.L.; Domínguez, L.; Gómez-Lucía, E.; Garayzabal, J.F.F.; Goyache, J.; Suárez, G. Experimental aflatoxin production in Manchego-type cheese. J. Appl. Bacteriol. 1988, 64, 17–26. [CrossRef] [PubMed]
3. Hymery, N.; Vasseur, V.; Coton, M.; Mounier, J.; Jany, J.L.; Barbier, G.; Coton, M. Filamentous fungi and mycotoxins in cheese: A review. Compr. Rev. Food Sci. F 2014, 13, 437–456. [CrossRef]
4. Lie, J.L.; Marth, E.H. Formation of aflatoxin in cheddar cheese by Aspergillus flavus and Aspergillus parasiticus. J. Dairy Sci. 1967, 50, 1708–1710. [CrossRef]
5. Taniwaki, M.H.; Hocking, A.D.; Pitt, J.I.; Fleet, G.H. Growth of fungi and mycotoxin production on cheese under modified atmospheres. Int. J. Food Microbiol. 2001, 68, 125–133. [CrossRef]
6. Markov, K.; Pleadin, J.; Bevardi, M.; Vah?ci´c, N.; Sokoli´c-Mihalak, D.; Frece, J. Natural occurrence of aflatoxin B1 , ochratoxin A and citrinin in Croatian fermented meat products. Food Control 2013, 34, 312–317. [CrossRef]
7. Pleadin, J.; Malenica, M.; Vah, N.; Milone, S.; Safti, L. Survey of aflatoxin B1 and ochratoxin A occurrence in traditional meat products coming from Croatian households and markets. Food Control 2015, 52, 71–77. [CrossRef]
8. Rodríguez, A.; Rodríguez, M.; Martín, A.; Delgado, J.; Córdoba, J.J. Presence of ochratoxin A on the surface of dry-cured Iberian ham after initial fungal growth in the drying stage. Meat Sci. 2012, 92, 728–734. [CrossRef] [PubMed]
9. Rodríguez, A.; Rodríguez, M.; Martín, A.; Núñez, F.; Córdoba, J.J. Evaluation of hazard of aflatoxin B1 , ochratoxin A and patulin production in dry-cured ham and early detection of producing moulds by qPCR. Food Control 2012, 27, 118–126. [CrossRef]
10. Delgado, J.; Acosta, R.; Rodríguez-Martín, A.; Bermúdez, E.; Núñez, F.; Asensio, M.A. Growth inhibition and stability of PgAFP from Penicillium chrysogenum against fungi common on dry-ripened meat products. Int. J. Food Microbiol. 2015, 205, 23–29. [CrossRef] [PubMed]
11. Marx, F. Small, basic antifungal proteins secreted from filamentous ascomycetes: a comparative study regarding expression, structure, function and potential application. Appl. Microbiol. Biotechnol. 2004, 65, 133–142. [CrossRef] [PubMed]
12. Tóth, L.; Kele, Z.; Borics, A.; Nagy, L.G.; Váradi, G.; Virágh, M.; Takó, M.; Vágvölgyi, C.; Gallgóczy, L. NFAP2, a novel cysteine-rich anti-yeast protein from Neosartorya fischeri NRRL 181: isolation and characterization. AMB Express 2016, 6, 75. [CrossRef] [PubMed]
13. Huber, A.; Hajdu, D.; Bratschun-Khan, D.; Gáspári, Z.; Varbanov, M.; Philippot, S.; Fizil, A.; Czajlik, A.; Kele, Z.; Sonderegger, C.; et al. New antimicrobial potential and structural properties of PAFB: A cationic, cysteine-rich protein from Penicillium chrysogenum Q176. Sci. Rep. 2018, 8, 1751. [CrossRef] [PubMed]
14. Tóth, L.; Váradi, G.; Borics, A.; Batta, G.; Kele, Z.; Vendrinszky, A.; Tóth, R.; Ficze, H.; Tóth, G.; Vagvölgy, C.; Marx, F.; Galgóczy, L. Anti-candidal activity and functional mapping of recombinant and synthetic Neosartorya fischeri antifungal protein 2 (NFAP2). Front. Microbiol. 2018, 9, 393. [CrossRef] [PubMed]
15. Rodríguez-Martín, A.; Acosta, R.; Liddell, S.; Núñez, F.; Benito, M.J.; Asensio, M.A. Characterization of the novel antifungal protein PgAFP and the encoding gene of Penicillium chrysogenum. Peptides 2010, 31, 541–547. [CrossRef] [PubMed]
16. Delgado, J.; Owens, R.A.; Doyle, S.; Asensio, M.A.; Núñez, F. Impact of the antifungal protein PgAFP from Penicillium chrysogenum on the protein profile in Aspergillus flavus. Appl. Microbiol. Biotechnol. 2015, 99, 8701–8715. [CrossRef] [PubMed]
17. Chen, Z.; Ao, J.; Yang, W.; Jiao, L.; Zheng, T.; Chen, X. Purification and characterization of a novel antifungal protein secreted by Penicillium chrysogenum from an Arctic sediment. Appl. Microbiol. Biotechnol. 2013, 97, 10381–10390. [CrossRef] [PubMed]
18. Galgóczy, L.; Kovács, L.; Karácsony, Z.; Virágh, M.; Hamari, Z.; Vágvölgyi, C. Investigation of the antimicrobial effect of Neosartorya fischeri antifungal protein (NFAP) after heterologous expression in Aspergillus nidulans. Microbiology 2013, 159, 411–419. [CrossRef] [PubMed]
19. Kaiserer, L.; Oberparleiter, C.; Weiler-Görz, R.; Burgstaller, W.; Leiter, E.; Marx, F. Characterization of the Penicillium chrysogenum antifungal protein PAF. Arch. Microbiol. 2003, 180, 204–210. [CrossRef] [PubMed]
20. Delgado, J.; Owens, R.A.; Doyle, S.; Núñez, F.; Asensio, M.A. Quantitative proteomics reveals new insights into calcium-mediated resistance mechanisms in Aspergillus flavus against the antifungal protein PgAFP in cheese. Food Microbiol. 2017, 66, 1–10. [CrossRef] [PubMed]
21. Jayashree, T.; Subramanyan, C. Oxidative stress as a prerequisite for aflatoxin production by Aspergillus parasiticus. Free Radical Biol. Med. 2000, 29, 981–985. [CrossRef]
22. Reverberi, M.; Fabbri, A.A.; Zjalic, S.; Ricelli, A.; Punelli, F.; Fanelli, C. Antioxidant enzymes stimulation in Aspergillus parasiticus by Lentinula edodes inhibits aflatoxin production. Appl. Microbiol. Biotechnol. 2005, 69, 207–215. [CrossRef] [PubMed]
23. Reverberi, M.; Punelli, M.; Smith, C.A.; Zjalic, S.; Scarpari, M.; Scala, V.; Fanelli, C. How peroxisomes affect aflatoxin biosynthesis in Aspergillus flavus. PLoS ONE 2012, 7, e48097. [CrossRef] [PubMed]
24. Maggio-Hall, L.A.; Wilson, R.A.; Keller, N.P. Fundamental contribution of beta-oxidation to polyketide mycotoxin production in planta. Mol. Plant Microbe Int. 2005, 18, 783–793. [CrossRef] [PubMed]
25. Roze, L.V.; Chanda, A.; Laivenieks, M.; Beaudry, R.M.; Artymovich, K.A.; Koptina, A.V.; Linz, J.E. Volatile profiling reveals intracellular metabolic changes in Aspergillus parasiticus: veA regulates branched chain amino acid and ethanol metabolism. BMC Biochem. 2010, 11, 33. [CrossRef] [PubMed]
26. Lozano-Ojalvo, D.; Rodríguez, A.; Bernáldez, V.; Córdoba, J.J.; Rodríguez, M. Influence of temperature and substrate conditions on the omt-1 gene expression of Aspergillus parasiticus in relation to its aflatoxin production. Int. J. Food Microbiol. 2013, 166, 263–269. [CrossRef] [PubMed]
27. Yu, J.; Chang, P.; Ehrlich, K.C.; Cary, J.W.; Bhatnagar, D.; Cleveland, T.E.; Payne, G.A.; Linz, J.E.; Woloshuk, C.P.; Bennett, J.W. Clustered pathway genes in aflatoxin biosynthesis. Appl. Environ. Microbiol. 2004, 70, 1253–1262. [CrossRef] [PubMed]
28. Mendonça, R.C.S.; Gouvêa, D.M.; Hungaro, H.M.; Sodré, A. de F.; Querol-Simon, A. Dynamics of the yeast flora in artisanal country style and industrial dry cured sausage (yeast in fermented sausage). Food Control 2013, 29, 143–148. [CrossRef]
29. Núñez, F.; Rodríguez, M.M.; Córdoba, J.J.; Bermúdez, M.E.; Asensio, M.A. Yeast population during ripening of dry-cured Iberian ham. Int. J. Food Microbiol. 1996, 29, 271–280. [CrossRef]
30. Andrade, M.J.; Thorsen, L.; Rodríguez, A.; Córdoba, J.J.; Jespersen, L. Inhibition of ochratoxigenic moulds by Debaryomyces hansenii strains for biopreservation of dry-cured meat products. Int. J. Food Microbiol. 2014, 170, 70–77. [CrossRef] [PubMed]
31. Núñez, F.; Lara, M.S.; Peromingo, B.; Delgado, J.; Sanchez-Montero, L.; Andrade, M.J. Selection and evaluation of Debaryomyces hansenii isolates as potential bioprotective agents against toxigenic penicillia in dry-fermented sausages. Food Microbiol. 2015, 46, 114–120. [CrossRef] [PubMed]
32. Peromingo, B.; Núñez, F.; Rodríguez, A.; Alía, A.; Andrade, M.J. Potential of yeasts isolated from dry-cured ham to control ochratoxin A production in meat models. Int. J. Food Microbiol. 2018, 268, 73–80. [CrossRef] [PubMed]
33. Li, H.; Zhang, S.; Lu, J.; Liu, L.; Uluko, H.; Pang, X.; Lv, J. Antifungal activities and effect of Lactobacillus casei AST18 on the mycelia morphology and ultrastructure of Penicillium chrysogenum. Food Control 2014, 43, 57–64. [CrossRef]
34. Effat, B.A.; Ibrahim, G.A.; Tawfik, N.F.; Sharaf, O.M. Comparison of antifungal activity of metabolites from Lactobacillus rhamnosus, Pediococcus acidilactici and Propionibacterium thoenii. Egypt. J. Dairy Sci. 2001, 29, 251–262.
35. Montiel, R.; Bravo, D.; Medina, M. Commercial biopreservatives combined with salt and sugar to control Listeria monocytogenes during smoked salmon processing. J. Food Prot. 2013, 76, 1463–1465. [CrossRef] [PubMed]
36. Mandal, V.; Sen, S.K.; Mandal, N.C. Production and partial characterisation of an inducer-dependent novel antifungal compound(s) by Pediococcus acidilactici LAB 5. J. Sci. Food Agr. 2013, 93, 2445–2453. [CrossRef] [PubMed]
37. Acosta, R.; Rodríguez-Martín, A.; Martín, A.; Núñez, F.; Asensio, M.A. Selection of antifungal protein-producing molds from dry-cured meat products. Int. J. Food Microbiol. 2009, 135, 39–46. [CrossRef][PubMed]
38. Lowry, O.H.; Rosebrough, N.J.; Farr, L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [PubMed]
39. Long, G.L.; Winefordner, J.D. Limit of detection, a closer look at the IUPAC definition. Anal. Chem. 1983, 55, 712–724.
40. Currie, L.A. Nomenclature in evaluation of analytical methods including detection and quantification capabilities. Anal. Chim. Acta 1999, 391, 105–126. [CrossRef]
41. Rodríguez, A.; Medina, Á.; Córdoba, J.J.; Magan, N. The influence of salt (NaCl) on ochratoxin A biosynthetic genes, growth and ochratoxin A production by three strains of Penicillium nordicum on a dry-cured ham-based medium. Int. J. Food Microbiol. 2014, 178, 113–119. [CrossRef] [PubMed]
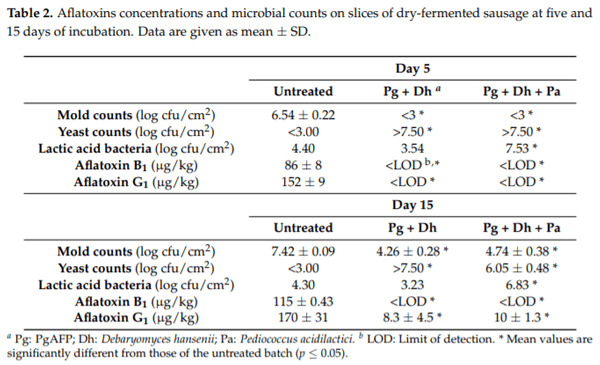
42. Rodríguez, A.; Rodríguez, M.; Luque, M.I.; Martín, A.; Córdoba, J.J. Real-time PCR assays for detection and quantification of aflatoxin-producing molds in foods. Food Microbiol. 2012, 31, 89–99. [CrossRef] [PubMed]
43. Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-??CT Method. Methods 2001, 25, 402–408. [CrossRef] [PubMed]
44. Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Prot. 2008, 3, 1101–1108. [CrossRef]
45. Cizeikiene, D.; Juodeikiene, G.; Paskevicius, A.; Bartkiene, E. Antimicrobial activity of lactic acid bacteria against pathogenic and spoilage microorganism isolated from food and their control in wheat bread. Food Control 2013, 31, 539–545. [CrossRef]
46. Liu, P.; Luo, L.; Long, C. Characterization of competition for nutrients in the biocontrol of Penicillium italicum by Kloeckera apiculata. Biol. Control 2013, 67, 157–162. [CrossRef]
47. Andrade, M.J.; Córdoba, J.J.; Casado, E.M.; Córdoba, M.G.; Rodríguez, M. Effect of selected strains of Debaryomyces hansenii on the volatile compound production of dry fermented sausage “salchichón”. Meat Sci. 2010, 85, 256–264. [CrossRef] [PubMed]
48. Demasi, T.W.; Wardlaw, F.B.; Dick, R.L.; Acton, J.C. Nonprotein nitrogen (NPN) and free amino acid contents of dry, fermented and nonfermented sausages. Meat Sci. 1990, 27, 1–12. [CrossRef]
49. Hierro, E.; de la Hoz, L.; Ordóñez, J.A. Contribution of microbial and meat endogenous enzymes to the lipolysis of dry fermented sausages. J. Agric. Food Chem. 1999, 45, 2989–2995. [CrossRef]
50. Dalié, D.K.D.; Deschamps, A.M.; Richard-Forget, F. Lactic acid bacteria. Potential for control of mould growth and mycotoxins: A review. Food Control 2010, 21, 370–380. [CrossRef]
51. Schmidt-Heydt, M.; Stoll, D.; Schütz, P.; Geisen, R. Oxidative stress induces the biosynthesis of citrinin by Penicillium verrucosum at the expense of ochratoxin. Int. J. Food Microbiol. 2014, 192, 1–6. [CrossRef] [PubMed]
52. Kim, J.H.; Yu, J.; Mahoney, N.; Chan, K.L.; Molyneux, R.J.; Varga, J.; Bhatnagar, D.; Thomas, E.; Cleveland, T.E.; Nierman, W.C.; Campbell, B.C. Elucidation of the functional genomics of antioxidant-based inhibition of aflatoxin biosynthesis. Int. J. Food Microbiol. 2008, 122, 49–60. [CrossRef] [PubMed]
53. Scherm, B.; Palomba, M.; Serra, D.; Marcello, A.; Migheli, Q. Detection of transcripts of the aflatoxin genes aflD, aflO, and aflP by reverse transcription-polymerase chain reaction allows differentiation of aflatoxin-producing and non-producing isolates of Aspergillus flavus and Aspergillus parasiticus. Int. J. Food Microbiol. 2005, 98, 201–210. [CrossRef] [PubMed]
54. Roze, L.; Laivenieks, M.; Hong, S.-Y.; Wee, J.; Wong, S.-S.; Vanos, B.; Linz, J. Aflatoxin biosynthesis is a novel source of reactive oxygen species. A potential redox signal to initiate resistance to oxidative stress? Toxins 2015, 7, 1411–1430. [CrossRef] [PubMed]
55. Binder, U.; Bencina, M.; Eigentler, A.; Meyer, V.; Marx, F. The Aspergillus giganteus antifungal protein AFPNN5353 activates the cell wall integrity pathway and perturbs calcium homeostasis. BMC Microbiol. 2011, 11, 209. [CrossRef] [PubMed]
56. European Commission. Commission regulation (CE) 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Offic. J. Eur. Union 2006, L364, 5–24.
57. Miller, J.D. Factors that affect the occurrence of fumonisin. Environ. Health Persp. 2001, 109 (Suppl. 2), 321–324. [CrossRef]
58. Payne, G.A.; Hagler, W.M., Jr. Effect of specific amino acids on growth and aflatoxin production by Aspergillus parasiticus and Aspergillus flavus in defined media. Appl. Environ. Microbiol. 1983, 46, 805–812. [PubMed]
59. Rüegg, M.; Blanc, B. Influence of water activity on the manufacture and aging of cheese. In Water Activity. Influences on Food Quality; Rockland, L.B., Stewart, G.F., Eds.; Academic Press, Inc.: New York, NY, USA, 1981; pp. 791–893.
60. Leistner, L.; Rödel, W.; Krispien, K. Microbiology of meat and meat products in high- and intermediate-moisture ranges. In Water Activity. Influences on Food Quality; Rockland, L.B., Stewart, G.F., Eds.; Academic Press, Inc.: New York, NY, USA, 1981; pp. 791–893.
61. Troller, J.A.; Christian, J.H.B. Water Activity and Foods; Academic Press, Inc.: New York, NY, USA, 1978.
62. Peromingo, B.; Rodríguez, A.; Bernáldez, V.; Delgado, J.; Rodríguez, M. Effect of temperature and water activity on growth and aflatoxin production by Aspergillus flavus and Aspergillus parasiticus on cured meat model systems. Meat Sci. 2016, 122, 76–83. [CrossRef] [PubMed]
63. Sedaghat, H.; Eskandari, M.H.; Moosavi-Nasab, M.; Shekarforoush, S.S. Application of non-starter lactic acid bacteria as biopreservative agents to control fungal spoilage of fresh cheese. Int. Dairy J. 2016, 56, 87–91. [CrossRef]







.jpg&w=3840&q=75)