The Vibrio bacteria in shrimp: how a common pathogen in shrimp farming became the leading character in the EMS story.
During the past year, evidence has accumulated on how the ongoing outbreak of early mortality syndrome (EMS) or more precisely acute hepatopancreatic necrosis disease (AHPND) is caused by a specific strain of the Vibrio parahaemolyticus bacteria. That this newly emerged disease is caused by a well-studied bacterial species wide-spread in shrimp aquaculture for decades, came somewhat as a surprise. Indeed, a vast body of knowledge exists on Vibrio in aquaculture, and in other related fields, namely on V. fisheri and V. harveyi, and the human pathogens V. cholera and V. parahaemolyticus.
For more than 40 years, Vibrionaceae has been consistently identified as one of the dominant families in the natural intestinal flora of wild and farmed penaeid shrimp. As Vibrio has a natural place in the microflora of penaeid shrimp, it can by no means be considered automatically as an obligate pathogen.
However, shortly after shrimp farming intensified, reports of disease and mortality in shrimp caused by Vibrio began to spread. V. harveyi was one of the first and most frequently identified culprits, later joined by V. parahaemolyticus and others. The disease was baptised ‘vibriosis’, and one of its most typical disease signs was an increased luminescence. Since then, a lot has been written about virulence of Vibrio in shrimp.
Virulent Vibrio
It is known that virulence genes are spread among Vibrio via horizontal gene transfer. Plasmids or temperate bacteriophages transfer genetic material from bacteria to bacteria, leading to a virulence switch and the production of toxins. This is relatively well-understood from human epidemics of V. cholera and V. parahaemolyticus and also in marine luminous Vibrio, such as V. harveyi.
The types of toxins, how the genes are transmitted among Vibrio and how they cause clinical signs have been extensively described for humans and shrimp. In fact, several publications have already reported on necrosis of hepatopancreas cells and midgut cells of shrimp by toxigenic Vibrio isolates, mainly V. harveyi.
As such, we may ask whether the recent EMS/AHPND outbreak truly presents a totally new case, or whether this bacterial pathogen follows the classical concepts which have been described in the past. Proper identification of the causative agent according to modern standards and the nature of the virulence factor (probable toxin) will answer this question.
Most Vibrios which cause disease and mortality in shrimp have been labelled as opportunistic/secondary pathogens, which take advantage of unfavourable environmental conditions to overwhelm shrimp hosts with compromised immune systems. This is observed when culture conditions are sub-optimal and stressful. Several specific strains of Vibrio sp were identified as primary pathogens, capable of killing shrimp in hatcheries and grow-out ponds even under optimal conditions, and these have become a major source of concern.
It has been stated that EMS/AHPND is caused by a newly emerged, primary pathogenic V. parahaemolyticus, capable of adversely affecting perfectly healthy shrimp no matter how much has been the effort in the management of the farm. The fact is however, that the distinction between primary and secondary pathogens is artificial. In both cases, there is a classic interplay of bacterial virulence and infection pressure, host defence and environmental influences which ultimately decide in which direction the balance tips. In all cases published so far, high doses of Vibrio had to be inoculated to reproduce disease under laboratory settings.
The recent publication by the research group of Dr Timothy Flegel and Dr Chu-Fang Lo, one of the few publications which have appeared on the etiology of EMS/AHPND apart of that by the group of Dr Donald Lightner, has strengthened our existing views on this matter. It is becoming clear that there is not one epizootic, hypervirulent strain of V. parahaemolyticus which is causing EMS/AHPND, but that there have to be several strains and species involved with variable clinical presentations and virulence mechanisms. Moreover, the authors are pointing out that shrimp health status and gut microflora may impact disease outcome and that affected shrimp might recover under suitable management conditions.
A holistic management approach
The available information and experience on Vibrio has allowed us to design a holistic management approach which can successfully minimise the damage the bacteria inflicts on cultured shrimp. The approach of our company, INVE Aquaculture has always been holistic. First and foremost, basic good practices have to be established in the management of the aquaculture systems to provide an optimal and stable environment. Secondly, the host health is reinforced by optimising nutrition and supportive supplements for the immune system. Thirdly, on the level of infectious agents, we aim to reduce the presence of viruses and virulent bacteria, while preventing opportunistic bacteria from getting a chance to overwhelm the hosts. To the best of our current knowledge, this integrated plan is so far the only way to restrain EMS/AHPND.
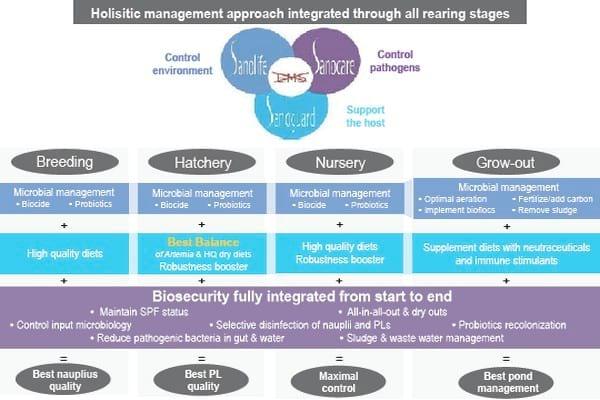
Figure 1. Holistic management approach integrated at all rearing stages
Breeding
A good breeding centre can only be operated under strict quarantine, with a surveillance program to maintain specific pathogen free (SPF) status. A team of experts has to plan and execute a long term program of selective breeding for desired traits, while keeping inbreeding under control. In the context of EMS, broodstock has been implicated as the cause of the problem, both that injudicious breeding which can lead to wide-spread inbreeding or that broodstock can be carriers of the EMS-causing bacterial strains.
Hence, we strongly advocate for disinfection of nauplii and materials used in the hatchery and farm in order to obtain real quarantine. In this context, further improvement of formulated diets for broodstock can allow the elimination of the risk of pathogen transfer via natural feeds.
Hatchery
As soon as shrimp larvae begin to feed, optimal nutrition becomes the main corner stone to ensure the best chances for survival and growth. Intuitively, most hatchery managers know that this is a complex process orchestrated by natural microbiological processes, and the administration of artificial feeds and live feeds (algae, Artemia).
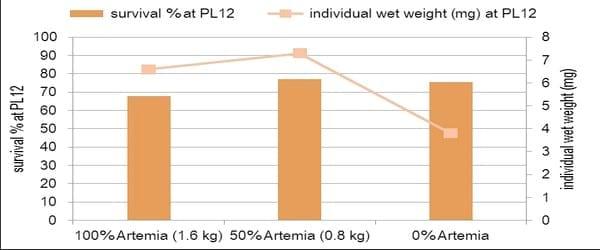
Our protocol aims to have as much control as possible over these factors by using probiotic bacteria, with the best balance of quality-controlled Artemia and dry diets. Together, this results in stable and controlled conditions, which renders the use of antibiotics unnecessary. Robustness of the post-larvae grown using this hatchery protocol is maximised by supplying specific health boosters, allowing them to cope better with stressors encountered during transport and when released in new environments. This can be evaluated objectively by recording their consistently better performance in stress tests.
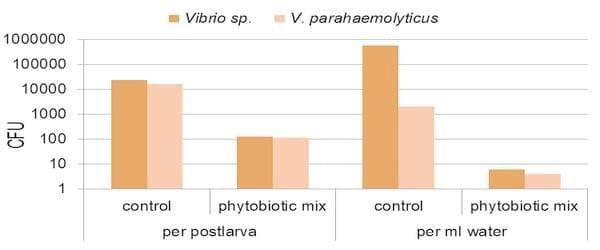
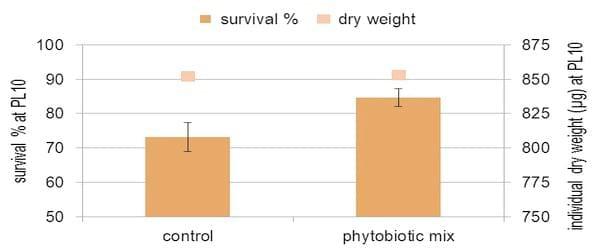
Recently, we have enhanced even further this stress resistance by applying a mixture of plant extracts during the hatchery cycle and in the transport water of post larvae. These specially selected plant molecules are not lethal to shrimp larvae, but boost their defence systems by inducing the production of heat-shock proteins. Additionally, they have a pronounced selective bactericidal effect, specifically against gram negative bacteria such as V. parahaemolyticus. As a result, post larvae with a superior quality can be delivered to grow-out ponds or nurseries.
Figure 2. The best balance between artificial diets and Artemia.
Figure 3. Reduction of Vibrio load by plant extracts during transport.
Figure 4. Enhanced survival during the hatchery cycle by applying plant extracts
Nursery
The practice of nursing post larvae to a larger size before stocking into ponds has been strongly encouraged since the onset of the EMS epizootic.
Although some have stated that this avoids early mortalities in ponds, others have reported that the practice does not result in any improvement. As information is being gathered, the explanations for differences in the effectiveness of using a nursery step has been emerging.
First and foremost, a good nursery requires a high investment. It has to be physically separated from the grow-out area and very strict biosecurity measures have to be in place. Due to the fear of EMS, many farmers have been making the mistake of leaving the shrimp for too long in the nursery, ignoring the carrying capacity of the system.
Furthermore, it has become clear that using cheap grow-out feeds undermines the success of a good nursery. The use of hatchery-grade diets does not only meet the nutritional requirements of young shrimp, the difference in ingredients also has a drastic effect on the microbial flora growing in the shrimp and on the faeces. Finally, robustness of the fry stocked at high densities should be maximised through addition of health boosters.
A good nursery protocol, be it in raceways or ponds, allows for better control and stabilisation of growth conditions, shorter cycles in open ponds, and more crops per year. But above all, in any shrimp nursery, well-trained staff and a good understanding of water management cannot be replaced by products with exaggerated claims or with the use of antibiotics.
Grow-out
The process of transferring shrimp from the nursery to the grow-out ponds offers a second opportunity to selectively disinfect against Vibrio and to boost the stress resistance of the shrimp. We see a big potential to optimise net profits from a shrimp crop with this practice, as even before EMS became a problem, the majority of shrimp mortality occurred during the first weeks after stocking.
Pond preparation should not be underestimated for grow-out and the same principles as for nursery should be applied, with attention to biosecurity and stabilising the chemistry and microbiology of the intake water. Applying disinfectant during pond preparation reduces the risk of horizontal transfer. However it is not sufficient and as a stand-alone treatment it might increase disease risk, as fast growing microorganism will fill up the empty niches.
What we support is a matured microbial ecosystem with a wide bacterial diversity. This microbial community will prevent V. parahaemolyticus from becoming one of the few dominant species. Therefore, a subsequent treatment with probiotic bacteria prevents opportunistic pathogens from blooming, colonising and invading the shrimp.
As we are dealing with an open system, algae play an important role in grow-out ponds. High quality fertilisers should be applied, in combination with a carbon source, in order to obtain the right C:N:P balance. After several weeks, depending on the stocking density, the organic load in the water will rise, and the algae phase will shift to the phase dominated by heterotrophic bacteria.
In this phase, three points are critical: aeration, pH and sludge management. The aerators have to be installed properly, so that they mix the whole pond (particularly important for biofloc systems), and maintain oxygen levels above 4 mg/L at all times.
The pH of the water has to be buffered by adequate amounts of alkalinity (>150 mg/L) and should not be allowed to fluctuate. Excess sludge has to be removed by regular siphoning. In the context of sludge management, many parallels can be made between the field of waste water management and the biofloc concept in aquaculture. In order to have as much control as possible over the dynamics of the spontaneous generation of bioflocs in the ponds, addition of selected Bacillus sp. and substrates is one of the few tools available to farmers.
The control strategies of V. cholera and V. parahaemolyticus from the point of view of human epidemics, center largely on waste water management, namely separating sewage and water sources. The cross-contamination of outlet water from shrimp farms to the inlet of the same or other farms has been a problematic issue for a long time already. With EMS/AHPND, this is more than ever a point to which farmers should pay particular attention. In order to prevent further spread and increase of the infection pressure in the environment surrounding shrimp farming areas, we advocate a proper treatment of pond water and sludge even after the crop has died due to an EMS outbreak.
Even if the environment of the shrimp is optimally managed, ubiquitous bacteria such as V. parahaemolyticus and V. harveyi might still have an opportunity to cause disease in shrimp. A major issue is that in grow-out, the feed constitutes a big part of the operation costs. Pressure on prices results in saving on ingredients, which leads to deficiencies in vitamins, oligo elements and bio-available ingredients. This is why grow-out diets have to be supplemented with nutraceuticals, enzymes and immune stimulants, which allow the shrimp to strengthen their natural barriers such as the cuticle, and to build up more reserves in energy and innate immunity to fight off invading bacteria.
Conclusion
The EMS crisis is a serious challenge for the shrimp farming industry and the supporting sectors. It is our firm belief that further integration and professional farm management are the way out of this crisis and to a sustainable future.
In our opinion, one single miracle cure product does not exist and certainly not a one-shot product which attacks V. parahaemolyticus. Opportunistic bacteria with transferable virulence genes have always been and will always be around in shrimp farming. The use of antibiotics or disinfection protocols without immediate recolonisation by probiotics is doomed. Our vision is that only a holistic approach, optimising environmental and post larvae quality, can successfully manage shrimp culture microbiology and curb EMS.
This article was originally published in Aquaculture Asia-Pacific, July/August 2014, Volume 10, Number 4.











.jpg&w=3840&q=75)


