I. Introduction
The UK review of antimicrobial resistance presented now outdated 2014 data on AMR consequences for human life (O’Neill, 2016). They estimated that 700,000 people die every year from AMR, with concerning devastating effects on infant mortality. In India, 60,000 newborn babies die each year from antibiotic-resistant neonatal infections. This problem is more prominent in less developed countries but by no means restricted to them. In the US, there are more than two million AMR infections per year, costing the US health system 20 billion USD. The committee of experts suggested a strong response to AMR (Figure 1); however, other than agriculture responding with efforts to remove antibiotics from production altogether, other directives are falling behind.
II. Major contributors to AMR rise
a) Human and hospital waste
The sewage microbial community is comprised mainly of human intestinal bacteria and some bacteria that grow on the sewage equipment and system. Antibiotics (ABs) are developed to be resilient to metabolic degradation and active during biological transit time. It is estimated that 50-80% of antibiotics is excreted in urine and 4-30% in faecal material (Riaz et al., 2020). This indicates that all unmetabolised antibiotics used by the human population, at home and in hospitals, end up in the sewage system. The sewage is saturated with antimicrobial-resistant bacteria (ARB) and their antimicrobial-resistant genes (ARGs). The troubling issue is the abundance of mobile genetic elements (MGEs), such as plasmids, which serve as vectors transferring ARGs between bacterial strains in the sewage environment. Sewage is the breeding ground for new ARB via ARG transfer.
Figure 1 – Recommendations from The Review on Antimicrobial Resistance (O'Neill, 2016)
Unfortunately, wastewater treatment plants (WWTPs) were not designed to remove or reduce AMR contamination. On the contrary, some multiple drug-resistant (MDR) species of Escherichia, Shigella and Klebsiella increase two-fold from raw to treated water. Similarly, total MDR Enterobacteriaceae surged from 5.5 to 14.1% in the treated wastewater. Species like methicillin-resistant Staphylococcus aureus (MRSA) are abundant in raw and treated sewage (Krzemiński et al., 2020). Hospitals often have their own waste treatment. However, that is reserved for medical waste rather than the general waste from toilets, kitchen and laundry. There is a whole new level of AMR enrichment in a hospital environment. Here, it is important to note that all hospital waste eventually ends up in sewage and WWTP.
b) AMR from agriculture
Four major manure types are used in agriculture: bovine, horse, pig and poultry. Although animal manures enrich the soil with essential and rare plant nutrients, they also contain biological impurities that include bacteria, fungi, helminths, parasites and other intestinal and environmental biological contaminants. Residual concentration of antibiotics in manure may not be very high and is reported as commonly ~1–10 mg/kg (Krzemiński et al., 2020); it was estimated that, in the last 50 years, more than one million tonnes of antibiotics entered the soil via manure (Allen et al., 2010).
The animal manure contains both urinary and faecal excrements, and both are used to excrete unmetabolised AB. In addition to adding antibiotics to the soil via manure, we also add ARB and ARGs. The survival time of pathogens in the soil is up to ten years and, on plant surfaces, up to one year (Krzemiński et al., 2020). Defra project by UK Centre for Ecology & Hydrology (Nicholson et al., 2016) estimated that approximately 96 million tonnes of farm manure are used annually in the UK and, with this volume of manure applied worldwide, antibiotic accumulation in the soil is inevitable.
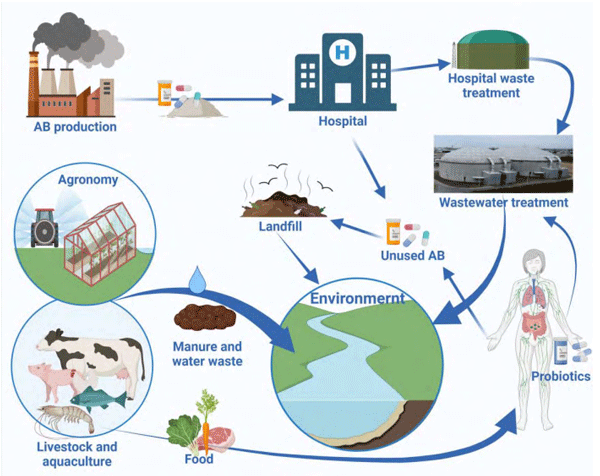
Although manure is a major source of antibiotics, ARG and ARB in the soil, in the USA and many other countries, all manure is considered an organic product and can be used on crops grown organically, thus introducing residual excreted antibiotics into organic plants and allowing AMB to colonise plants and ARGs exchange with native farm soil microbiota. As a result, many organic plants carry an abundance of AMR (Roberts, 2019). Ultimately all of these biological contaminants end up in food and in waterways (Figure 2).
There is another dimension to the presence of AMR in chicken sheds – farm employees welfare and safety! A good example is the UK use of vancomycin-similar drug avoparcin in poultry that resulted in the development of vancomycin-resistant Enterococcus faecium (VRE) in chickens. VRE was then found in processed meat, and farmers and abattoir staff who slaughtered the VRE+ animals, some of whom had to be hospitalised with the VRE infection (Cetinkaya et al., 2000; van den Bogaard et al., 2002). Additionally, van den Bogaard et al. (2002) reported that AMR for nearly all antibiotics was higher in broiler farmers than in egg farmers and slaughterhouse workers. This is in agreement with the higher use of antibiotics in broilers. Moreover, it was reported by multiple investigators that, in cattle and swine, faecal AMR decreases as food animals age, but this was not observed in short-lived poultry (Gaire et al., 2020).
Despite much attention to livestock animals and manure, one of the biggest AMR offenders is aquaculture. Aquaculture is the fastest-growing food-producing area in the world, and by many, considered the future of the food industry. Integrated aquaculture that involves feeding fish livestock manure is a traditional practice of the smaller Asian farms. Tetracyclines were extensively used in aquaculture, and aquaculture is mainly blamed for the broad tet-genes distribution in the world. In 2017 there were 59 tetracycline resistance genes identified even in the world’s most pristine remote environments like Antarctica and the Arctic (Roberts, 2019). It was estimated that 70% of an enormous amount of antibiotics given to seafood ends up in waterways. In addition to using and releasing massive amounts of antibiotics into waterways, the aquaculture AMR bio-waste also contaminates adjacent soils and waterways sediments where it is actively concentrated.
With the rise in seafood consumption, seafood-borne bacterial diseases are becoming more severe health hazards worldwide and, due to antibiotic use in production, it resulted in numerous AMR disease outbreaks in the USA (Elbashir et al., 2018). However, there is a considerable discrepancy in antibiotic usage in different countries. For example, antibiotics are used in a wide range – from 1g/t in Norway to 700 g/t in Vietnam (Defoirdt et al., 2011). Asian countries yield two-thirds of global food fish production, yet concerning levels of resistance to clinically critical antibiotics and high levels of foodborne zoonotic pathogens are evident (Schar et al., 2021), and this resulted in accumulated resistance along with Asia's major river systems, especially in China and India (Schar et al., 2021). Furthermore, to close the toxic cycle, AMR, ARB and ARGs are regularly present in drinking water (Roberts, 2019).
c) AMR in the soil
Soil represents the richest microbiota system, abundant with bacterial and fungal communities, and it is the origin of many clinical antibiotics (Afzaal et al., 2020). Similarly to waterways, the soil acts as a major sink that accepts the AMR from major contaminants like hospital waste, wastewater treatment, aquaculture and manure. Although some antibiotics can chemically bond with the soil and persist, soil factors such as soil texture and AMR gene stability play a role in AMR persistence in the soil (Macedo et al., 2020). In addition to antibiotics, many metals in the soil that generate toxic metal resistant bacteria and toxic metal resistant genes show potent cross- and co-resistance to antibiotics and can result in AMR gene emergence without any exposure to antibiotics (Yazdankhah et al., 2018). This is further complicated with both manure and inorganic fertilisers.
d) AMR in the food
AMR pathogens in human food present a major risk for public health. Food is contaminated by the presence of AB and ARB on food due to the use of antibiotics in production and via cross-contamination with ARB during food processing (Verraes et al., 2013). This is of special concern with seafood (Elbashir et al., 2018). ARB and ARG have been identified in tap water, milk, meat, vegetables, and processed and unprocessed foods (Roberts, 2019); thus, the food chain is one of the principal means of AMR transmission. Although often present in the very low amount on meat, ARBs can often amplify to high numbers due to poor meat handling, storage and hygiene practices (Plaza-Rodriguez et al., 2021).
While ARB and AMR in the meat are a consequence of the contamination in slaughterhouses and processing, vegetables are known to actively uptake antibiotics and AMR genes from the soil or irrigation water (Azanu et al., 2016), especially if manure is used for fertilising (Tasho and Cho, 2016). Wang et al. (2015) and many others reviewed in Tasho and Cho (2016), identified highly abundant ARGs on vegetables growing on soil with more than three years of manure usage.
Although very little attention is given to wild animals, the levels of AMR infiltration into their water, soil and plants suggests that they could not remain unaffected. Even in wild animals that have minimal contact with humans and live in generally considered pristine environments, ARB, like highly multiresistant MRSA, were detected in deer, boar, wolves, foxes, pigeons, pheasants and birds of prey (reviewed in Silva et al., 2020).
Fruit and vegetables are declared safe for human use if they have antibiotic residue under the selected maximum threshold. Similarly, fish and seafood heavily treated with antibiotics can be used after being housed in antibiotic-free water for some time, referred to as the withdrawal period (Bhattacharjee, 2016). Unfortunately, the simple antibiotic residue is a much smaller problem than ARB and ARGs in food. Additionally, washing fruit and vegetables in drinking tap water is not going to help since an abundance of researchers detected AMR in tap water (Roberts, 2019).
e) AMR in the air
Although it may seem unmanageable, AMR and ARGs have been isolated from the air, especially in highly industrialised countries like China. Li and colleagues (Li et al., 2018) carried out a well-known study investigating air samples from 19 major cities worldwide and screening 30 ARGs (Li et al., 2018). The ARGs were detected to vary by nearly 100-fold in different cities. The highest number of ARGs was detected in Bejing, and San Fransico had the highest ARG abundance. Clinically critical antibiotic resistance genes such as vancomycin were detected in air samples (Li et al., 2018), indicating that urban air represents a health risk associated with continual exposure to airborne ARGs. Li and colleagues also analysed bacterial communities in the air, their networks and contributions to ARGs. They found 50 genera in the urban air, including Corynebacterium, Escherichia/Shigella, Streptococcus and other potentially pathogenic genera. Many other authors came to similar conclusions (Krzemiński et al., 2020). Unlike AMR in soil, the AMR in air and water are mobile, and diffusion, floods and rain play a role in delivering AMR, ARB and ARGs to the remote and "untouched" ecosystems.
f) AMR in probiotics
Although often ignored, it is well established that most probiotics carry AMR and ARGs. This is well investigated and comprehensively reviewed (Gueimonde et al., 2013; Li et al., 2020). The probiotics carry ARGs, because AMR profile was not required for their registration and, even if it was, AMR profile was until recently done using PCR on a hand-picked subset of genes or disc diffusion tests, methodologies inferior to shotgun metagenomic sequencing and blasting against databases that now have well over 3000 fully annotated ARGs. Some researchers that tackled this issue suggest that it is suitable for species like Lactobacillus to be antibiotic-resistant so they can survive, maintain and help to restore healthy gut after antibiotic treatments (Gueimonde et al., 2013). However, authors universally agree that ARGs on mobile genetic elements (such as plasmids) constitute a significant problem because of resistance spread to other species (Zheng et al., 2017; Baumgardner et al., 2021). Liu confirmed the presence of resistance to AB, including clinically critical, in commercial probiotics, identifying vancomycin, rifampicin, streptomycin, bacitracin, and erythromycin ARGs (Liu et al., 2009), and suggesting a re-evaluation of probiotic safety and proposing new regulations.
Probiotics are continually added into the intestinal community due to regular use in both humans and supplemented animals (Zheng et al., 2017) and the effects of accumulation of AMR is not yet investigated. Baumgardner and colleagues (Baumgardner et al., 2021) performed a comprehensive study to determine if transferable ARGs are present in 47 commercially available veterinary probiotics, including products marketed for cattle, dogs, camelids, cats, goats and horses. 97% of the 47 probiotics contained at least one mobile AMR gene, and 82% contained two or more. This study is highly relevant to poultry and other livestock, and it confirms the risk for transmission of these mobile AMR genes into meat products, manure and environment. Considering the scale of production and administration of probiotics, they also strongly amount to AMR enrichment in manure, and thus soil, water, and the environment. In a survey of fermented food starter cultures from 90 different sources in Switzerland, Kastner et al. (2006) confirmed AMR in the most common starter culture species. A recent study by Montassier published in Nature Microbiology (Montassier et al., 2021) brings this story to another level. An administration of ABs resulted in the increase of the lower GI tract resistome. However, the addition of probiotics further exacerbated AMR and ARGs expansion in the gut mucosa by promoting the bloom of strains carrying vancomycin resistance genes but not resistance genes carried by the probiotic strains! This opens an interesting new area of research on identifying supplements that enlarge or reduce resistome in human and animal intestines. For example, the faecal transplant can assist in the reduction of ARGs in the gut (Montassier et al., 2021). The vicious cycle of AMR spread on a global scale is presented in Figure 2.
Figure 2 – Vicious cycle of AMR spread and expansion in the environment
III. Future directions
The estimated death toll from rising AMR is in millions per year (O’Neill, 2016), and medical practitioners are constantly reporting new all-resistant strains of pathogens while AMR related annual mortality is on a steep rise (O’Neill, 2016). There are numerous suggestions to improve monitoring, surveillance and diagnostics; however, efforts in reducing the load of AMR in the environment are urgently needed. One of the emerging methods is bioremediation using living organisms that have a remarkable ability to remove or reduce ARG load in treated samples.
Bacteria and other microorganisms such as protozoa and yeast are the most promising and widely used for AMR and AB bioremediation. However, this is based on complex microbial interactions and is not yet fully understood or optimised. To assist bioremediation, microorganisms must be able to survive and thrive under extreme conditions (oxidative pressure, nutrient depletion, osmotic stress) (reviewed in Apreja et al., 2021). It was reported that a combination of bacteria and microalgae could efficiently remove a range of antibiotics from the sludge, where the symbiotic interactions between bacteria and microalgae played a major role in the kinetics of antibiotic removal (da Silva Rodrigues et al., 2021). Although this area of research is relatively new, modern methodologies such as shotgun sequencing of antibiotic-resistant isolates revealed new enzymes capable of bioremediation (dos Santos et al., 2015). They opened an opportunity for the rapid development of bacterial libraries capable of removing ABs from the environment. Bacillus species, alone or in combination with other microorganisms, are among the most promising candidates (Al-Gheethi et al., 2019).
Phytoremediation is the removal of antibiotics using algae. Cyanobacteria are among the most interesting: they use light as an energy source and CO2 as a carbon source, also helping in carbon sequestration during the bioremediation process. Additionally, they are hardy, capable of growing under extreme conditions, and capable of fixing up atmospheric nitrogen. Successful examples of algal bioremediation of antibiotics are well presented and reviewed (Grimes et al., 2019; Xiao et al., 2021; Zhou et al., 2021). It is, however, important to monitor the production of algal toxins before selecting the algae of choice for bioremediation.
Mycoremediation and phytoremediation are up-and-coming remediation techniques using fungi and plants to remove ABs from the environment. We previously discussed how plants accumulate AB from the soil; although this is an issue for vegetable and other edible plants production, it is excellent in bioremediation.
IV. Conclusions
There is no doubt that AMR is one of the major global health issues. AMR is a natural phenomenon that has existed for as long as microorganisms. The preservation of intestinal content in both naturally and deliberately mummified human remains confirmed the presence of resistance genes to most known antibiotics, including most recently discovered (reviewed in Santiago-Rodriguez et al., 2017). This became an excuse for many to ignore the current global AMR pollution putting aside the fact that never in history have we had millions of tonnes of antibiotics pumped into the environment. According to WHO predictions, we are heading towards post-antibiotic era where things like c-section, organ transplants or chemotherapy will be hazardous procedures.
Presented at the 33th Annual Australian Poultry Science Symposium 2022. For information on the next edition, click here. 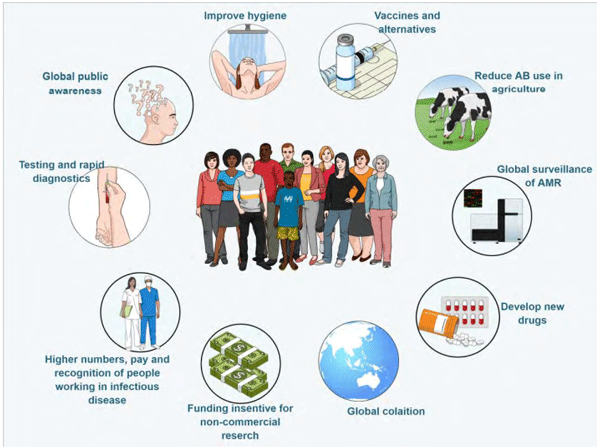









.jpg&w=3840&q=75)