Sel-Plex® maintains small intestine integrity in reovirusinfected broiler chickens
In 1974, when the US Food and Drug Administration (FDA) approved selenium as a feed supplement, inorganic selenium, primarily as sodium selenite, became the traditional source for dietary supplemental selenium for poultry and livestock (Leeson and Summers, 1991).
That decision in 1974 was based on cost of the selenium supplements and lack of information on selenomethionine, and it was ironic because the commonly used plant- and animal-based feed ingredients contain selenium almost exclusively as organic compounds such as the naturally occurring selenoamino acids (Burk and Hill, 1976; Levander, 1986; Cai et al., 1995).
Without a doubt, the use of inorganic selenium supplements in feeds has improved the performance of all classes of commercial poultry, but modern high-yielding poultry have higher metabolic rates and different nutritional needs compared with poultry from 35 years ago, thereby signaling a need to reassess the nutrient requirements.
Inorganic selenium has some problems associated with its use. Among those problems are the minimal levels of selenium in meat proteins and the potential for toxicity if too high a dietary level of inorganic selenium is provided to chickens. Sodium selenite has a documented pro-oxidant influence in all animals tested (Spallholz, 1997; Terada et al., 1999). Thus, a need to revisit organic selenium as a feed supplement for poultry is apparent.
After many years of laboratory and field research, a source for natural organic selenium (Sel-Plex®, Alltech, Inc.), was approved for use in the poultry industry by the US FDA (Federal Register, 2000 and 2002). Sel-Plex® provides a cocktail of selenium compounds (Kelly and Power, 1995), but selenomethionine in the selenium-enriched yeast cellular protein component is the primary form of selenium in Sel-Plex®.
The organic selenium profile in Sel-Plex® is similar to the organic selenium profile in plants and grains (Kelly and Power, 1995). The organic selenium in Sel-Plex® is readily available and will be absorbed actively (Mahan, 1995) from the intestine via the Na+-dependent methionine transport system (Spencer and Blau, 1962) while sodium selenite is absorbed passively by diffusion from the intestinal tract (Schrauzer, 2000).
Selenium influence on host:virus interaction
There are more than 13,000 scientific studies demonstrating increased susceptibility to infection in association with malnourishment in both humans and lower vertebrate animals. Most of those studies have focused on poor diets and how the host immune system has been negatively affected. In comparison, little is known about how the malnourished host may affect a virus and how that virus then interacts with the host. There is one ultramicro trace element, selenium, that seems to play a major role in infections due to RNA viruses (Combs, 2001; Field et al., 2002; Lyons et al., 2003; Beck et al., 2004).
Research into the influence of malnutrition on host-viral interactions was initiated by Melinda A. Beck and colleagues at the University of North Carolina at Chapel Hill when she discovered the emergence of new viral variants in a selenium-deficient model (Beck et al., 1994; 1995; 1998; 2003). Beck’s group found that mice deficient in selenium were more susceptible to coxsackievirus B3. Those mice infected with a normally harmless strain of coxsackievirus developed myocarditis, because in the selenium-deficient mice, the avirulent virus had mutated to a virulent form. The re-isolated virus was then shown to be virulent by inducing myocarditis in normal mice.
The viral genome mutation from avirulent to virulent was shown to be mediated by increased oxidative stress due to selenium deficiency. Beck and colleagues also demonstrated that even influenza virus is influenced by host selenium status (Beck et al., 2001; Nelson et al., 2001; Beck et al., 2004). Using the low pathogenic influenza A/ Bangkok/1/79 (H3N2) virus strain that produces a mild pneumatitis in mice, Beck et al. (2001) found much more severe pathology in selenium-deficient mice than in mice fed a selenium-adequate diet. Part of the increased level of pathology was due to increased proinflammatory cytokine production in lungs of selenium-deficient mice compared with selenium-adequate mice.
Following Beck’s pioneering work, numerous other studies have revealed similar relationships involving host selenium deficiency and virulence of RNA viruses. Selenium is very important in the maintenance of health status in HIV-infected patients (Foster, 2003). It has been reported that selenium inhibits HIV replication (Look et al., 1997) and reactivation by hydrogen peroxide (Sappey et al., 1994).
Selenium also plays a protective role in patients with hepatitis B and C infections preventing progression to cirrhosis and liver cancer (Yu et al., 1997; 1999). Measles virus infected children given a 12-month nutritional supplement with selenium had significantly lower incidences of diarrhea, fever, and acute lower respiratory infection (Juyal et al., 2004). Additionally, Broome et al. (2004) reported that patients given a live attenuated polio vaccine along with selenium supplementation had improved immune functions and stopped shedding the polio vaccine virus more quickly, and a higher rate of selenium supplementation (100 μg/day) was better than a lower rate (50 μg/day).
Influence of selenium on avian virus:host interaction
To date there has been little information linking selenium with the responses of poultry species to viral infections. One study with chicks was identified in which supplementation of vitamin E and selenium to diets already adequate in these nutrients was assessed (Panda and Rao, 1994). Compared with unsupplemented groups, birds fed the vitamin E and selenium supplement had enhanced immune function, including higher geometric mean titers in the tube agglutination test against infectious bursal disease virus (IBDV) and higher numbers of rosette–forming cells in the peripheral blood.
Newcastle Disease virus (NDV) infections also appear to be affected by vitamin E and selenium supplementation. Bassiouni et al. (1990) observed improved resistance against NDV, increased antibody titers, and improved weight gain associated with selenium and vitamin E supplementation. Swain et al. (2000) have shown that broiler chicken responses to NDV vaccine were enhanced by selenium and vitamin E supplements greater than NRC requirements.
Additionally, Marin et al. (2003) reported increased antibody titers against IBDV and NDV, increased serum protein levels, and prevention of bursal damage in aflatoxin-B1-exposed chickens when the chickens were given dietary supplements of selenium and vitamin E. Singh et al. (2006) reported a synergistic effect between vitamin E and selenium in chickens given a NDV vaccine. NDV-vaccinated chickens had higher hemagglutination inhibition titers when given the combination of selenium and vitamin E along with higher circulating levels of immunoglobulins and circulatory immune complexes.
Reovirus in chickens
In the poultry world, there are numerous economically important RNA viruses. Among these are the reoviruses, which are species-specific and have a broad range of virulence in chickens (Robertson et al., 1984). Reoviruses can infect joints and tendons, the respiratory tract, and the intestinal tract. Recently, there have been numerous field cases of enteric reovirus infections leading to malabsorption syndrome, moderate to severe weight gain depression, and increased mortality.
Malabsorption syndrome was investigated by Jensen and colleagues (1991) who found that both selenium and vitamin E were effective in reducing mortality. In fact, Swick (1995) has recommended that selenium be supplemented to the highest allowable dietary level to reduce reovirus-related mortality associated with malabsorption syndrome. The higher metabolic rates of the modern high meat-yielding broiler chicken might contribute to the severity of RNA virus infection because the RNA viruses depend on high levels of oxidative stress to facilitate replication.
Thus, it is important to investigate the role selenium might play in RNA virus infection. This work was conducted to examine the influence of dietary selenium on intestinal morphology of broiler chickens challenged with an enteric avian reovirus (ARV).
MATERIAL AND METHODS
Chicks
Cobb broiler chicks were hatched from eggs produced by breeders that had been maintained on isocaloric torula yeast diets that provided 1) no selenium (less than 0.02 ppm), 2) Sel-Plex® as the source of organic selenium (0.3 ppm Se), or 3) sodium selenite (0.3 ppm Se). Hatchling chicks were placed in pens in heated brooder batteries and were given the same diet that had been fed to their parents. Body weights were determined at 14 and 21 days of age, and at those ages tissues were collected for other analyses related to selenium and reovirus infection.
Reovirus challenge
On the day of hatch, 30 chicks in each of the three dietary treatments were placed into either control or ARV-infected groups (total of 180 chicks) in heated metal growing batteries in separate isolation rooms. Chicks in the ARV-infected groups were given an oral gavage of 0.5 mL of the reovirus ARV-CU98 (104.2 plaque-forming units/chick), and control chicks were given the medium only. At 21 days of age, the chicks used in this report were killed by carbon dioxide asphyxiation and intestinal tracts were dissected to determine total and segmental weight and length. Tissues were also collected for histomorphometry.
Histomorphometry
An individual bird was regarded as the experimental unit, and there were five birds per dietary selenium and virus treatment group analyzed. A total of 30 chickens were used in this phase of the overall study. After dissection, the duodenum and ileal segments were collected and fixed in Carnoy’s solution. Tissues were processed for histology and stained with hematoxylin-eosin.
A computerized microscope-based image analyzer (Southern Micro Instruments, Atlanta, GA) was used to determine the histomorphometric parameters for villus height, villus width at its mid-height, villus perimeter length, crypt depth, external muscle layer thickness, and villus height:crypt depth ratio (Fan et al., 1997). The criterion for selection of histological sections for examination was based on the presence of an intact lamina propria, and villi were chosen that were perpendicularly sectioned through the midline axis.
Statistics
All data from this completely randomized experimental design were analyzed using the general linear models procedure of SAS (SAS Institute, 1996). Significant differences were determined by analysis of variance and were separated by Least Significant Difference at P≤0.05. For the histomorphometric calculations, an average of ten measurements for each parameter from each bird were statistically analyzed as a one-way analysis of variance.
Fisher’s Least Significant Difference was used to test differences between means only when the analysis of variance indicated significance at P≤0.05 (Motulsky, 2005).
RESULTS
Body weight was depressed significantly by reovirus infection at both 14 days (Control = 514 g; Infected = 362 g) and 21 days (Control = 962 g; Infected = 724 g) postchallenge, but there were no differences in body weights for the three selenium treatments within control and infected groups.
It was of interest however, that weight gain from 14 to 21 days in organic selenium-fed chicks was somewhat greater than that in the no selenium and sodium selenite groups in both control (no selenium = 466 g, Sel-Plex® = 476 g; selenite = 460 g) and infected (no selenium = 355 g, Sel-Plex® = 388 g, selenite = 342 g) groups. The concentration of plasma protein was reduced significantly by reovirus infection at 14 days post-challenge, but Sel-Plex® supplementation had maintained plasma protein at a higher concentration compared with no selenium and selenite feeding at both 14 and 21 days post-challenge.
Reovirus infection increased duodenal and jejunal segment relative wet weight, but reovirus infection did not affect ileum relative wet weight and decreased large intestine wet weight (Table 1).
A selenium effect was seen in the relative weights of the duodenum with organic selenium decreasing relative wet weight in control birds and increasing the relative weight in the virus-challenged birds and the selenite-fed diet, causing a decrease in relative weight in the infected birds. This caused a significant diet × virus interaction for duodenum relative weight compared with the whole of the GIT. On a live weight basis (Table 2), the whole GIT, duodenum, jejunum, and ileum showed increases in relative weight due to infection. The large intestine relative weights were not affected by virus but Sel-Plex® supplementation caused a consistent increase in relative weight.
The heart also showed an increase in relative weight due to selenium treatment. Overall, reovirus-infected birds exhibited longer and heavier gastrointestinal tracts. Reovirus infection did not affect length of the intestines (data not shown). However, there were statistically significant diet effects on average gastrointestinal lengths, with torula yeast increasing length by almost 9% compared with the two selenium source diets (P<0.05).
Table 1. Selenium and reovirus influence on duodenum, jejunum, ileum and large intestine relative weights (%).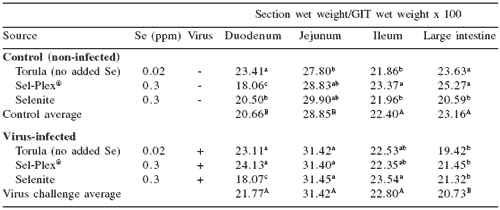
abcMeans in a column differ, P=0.05
ABMeans in a column differ, P=0.05
Table 2. Selenium and reovirus influence on gastrointestinal tract (GIT): duodenum, jejunum, ileum, and large intestine relative weights (%).1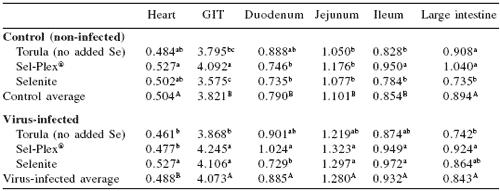
abcMeans in a column differ, P=0.05
ABMeans in a column differ, P=0.05
1Organ net weight/live BW × 100
Typical fields of view of the ileum in control and reovirus-infected chickens are shown in Figure 1. It can be seen that there are differences among the control and infected chickens and that there are selenium source effects in both the control and infected groups. Similar effects were also evident in the duodenum (not shown).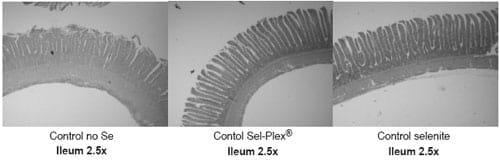
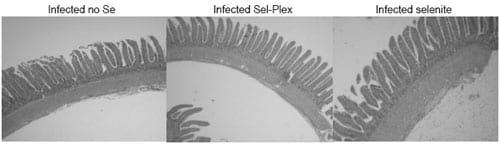
Figure 1. Typical fields of view in control and infected sections of the ileum from no selenium, Sel-Plex® and sodium selenite-fed chickens at three weeks of age following reovirus challenge at day of hatch.
Intestinal morphometric analysis of the duodenum (Table 3) revealed both diet and reovirus infection effects on the different parameters. The perimeter of the villus significantly increased in the Sel-Plex® control and infected groups compared with the perimeters of the no selenium and selenite groups. There was no difference between control and selenite for villus perimeter.
Villus height was significantly greater for Sel- Plex® compared with no selenium and selenite in both control and reovirus-infected birds. Villus width was greater in selenite-fed birds in the control group compared with no selenium and Sel-Plex®. However, in the infected birds, those given no selenium had the greatest villus width followed by selenite and Sel-Plex®, respectively. There was a significant virus-related increase in crypt depth, and there was a significant increase in crypt depth in the no selenium- and selenite-fed birds compared with Sel-Plex® feeding.
The villus height:crypt depth ratio for controls and for infected groups was increased compared with Sel-Plex®-fed birds in both control and infected groups. Muscularis externa thickness was increased in virus-challenged chickens and was of the greatest thickness in Sel-Plex®-fed and no selenium fed birds. In the control groups, selenite-fed birds had the greatest muscle thickness followed by those give no selenium and those given Sel-Plex®, respectively.
The morphometric analysis results for the ileum are shown in Table 4. There was a significant depressive effect of virus infection on villus length in all dietary treatments.
In control groups the torula yeast-fed (no selenium) treatment resulted in the longest perimeter followed by Sel-Plex® and selenite, respectively, but in the infected group, the no selenium treatment resulted in the shortest perimeter followed by the selenium treatments, which did not differ from each other. These responses resulted in a significant diet × virus interaction.
Villus height was also negatively impacted by virus infection in all selenium treatments. In the control groups the no selenium and Sel-Plex®-fed fed birds had longer villi than selenite-fed birds, and in the infected groups, Sel-Plex®-fed birds had longer villi than either no selenium or selenite-fed birds.
Villus width was increased in the infected birds given either no selenium or sodium selenite treatments, but villus width in the Sel-Plex®-fed birds was not different from controls. This resulted in a significant diet × virus interaction for villus width. The crypt depth was less in Sel- Plex®-fed birds in both the control and virus infected groups. No selenium and sodium selenite treatments resulted in increased crypt depth in both control and infected groups.
There were significant selenium and virus effects for height:crypt depth ratios. Sel- Plex®-fed birds had the largest ratios in both control and infected groups, and no selenium and sodium selenite fed birds had significantly smaller H:D ratios. Diet and virus effects were evident for muscularis externa muscle thickness in the ileum, which showed that both selenium treatments resulted in increased muscle thickness after infection. Birds fed no selenium did not alter muscle thickness due to infection.
Table 3. Selenium and reovirus influence on morphology of villus perimeter, height, width, crypt depth, villus height:crypt depth [H:D] ratio, and muscularis externa [muscle] thickness) of duodenum at 21 days.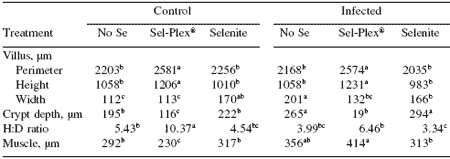
abcdIn a row, means differ, P<0.05
Table 4. Selenium and reovirus influence on morphology of villus perimeter, height, width, crypt depth, villus height:crypt depth [H:D] ratio, and muscularis externa [muscle] thickness) of ileum at 21 days of age.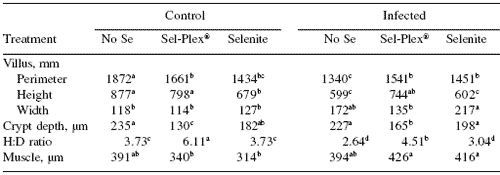
abcdIn a row, means differ, P=0.05.
DISCUSSION
There is no doubt that reovirus infection causes decreased weight gain in chickens. Part of the cause for this decreased weight gain after infection is attributed to the fact that the virus hijacks the protein synthesis mechanism in the cells early in the infection. This observation was supported by the results from this study and others (Guy, 1998). Rebel et al. (2004) report that by 14 days post-challenge, chickens exposed to an enteric reovirus begin to recover from the initial infection as evidenced by reduced severity of lesions in the villus and crypt regions of the intestine.
An external indication of the reovirus infection can be observed as poor feathering, but as the infection begins to resolve, the protein synthesis mechanism regains its efficiency. In this study, weight gain was suppressed by reovirus infection and was evident through 21 days of age.
However, there was an interesting observation, which showed that Sel-Plex®-fed chickens were gaining weight at a slightly faster rate at 21 days than were chickens fed no selenium or selenite in both control and infected groups. This suggested that the improved antioxidant status found in chickens fed Sel-Plex® had facilitated a faster recovery after reovirus infection.
Mahmoud and Edens (2003) have demonstrated that organic selenium as Sel-Plex® has the ability to induce higher selenium-dependent antioxidant enzyme activities than do sodium selenite or no selenium feeding regimes. The improved redox status then can be directly related to improved performance of broiler chickens fed Sel- Plex® (Edens and Gowdy, 2004).
Surai (2006) reviewed extensively the health effects of dietary selenium supplementation.
He pointed out that selenium does not prevent virus-induced disease, but selenium supplementation appears to play a pivotal role in reducing the severity of the infection. That observation can be stated regarding the results of this study with reovirus infection in chickens.
The improved health status imparted by the feeding of selenium is attributed to enhanced immune responses and increased activity of antioxidant enzymes such as glutathione peroxidase (Surai, 2006; Mahmoud and Edens, 2003) and thioredoxin reductase (Edens and Gowdy, 2004). Even though there is improved redox status and enhanced immunity associated with feeding Sel-Plex®, there are other physiological manifestations of the influence of selenium on the overall health status of the chicken.
Earlier studies have suggested that selenium has a role in regulating virus replication.
Balansky and Argirova (1981) concluded that selenium depressed RNA-dependent RNA polymerase in an oncovirus as an indicator of reverse transcriptase activity. Balansky and Argirova (1981) further hypothesized that selenium prevents integration of the virus genome with the host’s DNA.
Similar observations were made earlier by both Billard and Peets (1974) and Oxford (1973) who reported that selenocystine and selenocystamin both inhibited RNA-dependent RNA polymerase activity in influenza A1, A2 and B viruses.
Schrauzer and Sacher (1994) also reported that selenium causes symptomatic improvements and possibly slows the course of the HIV disease possibly through the inhibition of reverse transcriptase activity in the HIV-virus-infected patients. Schrauzer and Sacher (1994) concluded that it is possible that selenium might also prevent the replication of HIV and retard the development of AIDS in newly HIVinfected subjects. The depressive influence of selenium on reverse transcriptase activity in RNA viruses is very important and suggests that selenium is needed by the host to resist expression and possibly transmission of viruses.
There is evidence in this report to show that selenium increased the relative weight of the heart, and this is important due to the known influence of congestive heart disease on the induction of ascites in modern high yielding broilers. Sel-Plex® and sodium selenite increased heart weight in the control and reovirus-infected birds compared with birds given no selenium.
Relative weights of the whole gastrointestinal tract were apparently elevated by Sel-Plex® feeding compared with the other dietary treatments, and this was followed by sodium selenite and by no selenium-fed birds. When the morphometry of the intestinal segments was examined, it was apparent that the muscle thickness of the ileum and duodenum, very large segments of the GIT, were not affected consistently by either of the dietary selenium treatments. Selenium generally increased muscle thickness in the infected chickens. Whether this was a response leading to recovery from the virus infection or for some other reason was not determined in this study.
One of the most impressive and consistent observations made in the morphometry study was that Sel-Plex® generally maintained longer and more slender villi than either sodium selenite or no selenium feeding. Histomorphometric analysis indicated that increased villus height and perimeter, as well as muscle thickness and crypt depth in the ileum and duodenum were associated with Sel-Plex® feeding compared with either sodium selenite or no selenium (Tables 3 and 4). Muscle thickness generally was greater in the ileum than in the duodenum in both control and infected chickens. Villus height and villus height/crypt depth ratio is directly correlated with increased enterocyte turnover (Fan et al., 1997).
In the present study, morphometry parameters strongly indicated that Sel-Plex® feeding had resulted in the maintenance of the integrity of the intestinal tract as indicated by taller villi and an increased height/crypt depth ratio. Lower villus height:crypt depth ratios in both control and infected birds suggests that the increases in villus height and perimeter associated with Sel-Plex® treatment are not associated with enterocyte turnover rates but maintenance of the enterocyte population covering the villus. The reason for this is attributed to improved redox status in the GIT, and this resulted in less oxidative stress during normal and infective periods. An extension of this observation is that with an improved redox status, the reovirus probably had a more difficult task associated with replication within the enterocytes of Sel-Plex®-fed chickens.
Rebel et al. (2004) have described pathological changes associated with malabsorption syndrome in chickens as being characterized by degenerate epithelial cells in the crypt, villus atrophy, and low villus height:crypt depth ratio similar to the condition associated with intestinal segments from no selenium and sodium selenite-fed chickens in this study. Thus, the improved weight gain after the peak infective state in the chickens fed Sel-Plex® might be related to 1) greater surface area:volume ratio in the villi, 2) increased ability to assimilate nutrients compared with selenite and no selenium-fed birds after recovery was apparent, 3) decreased enterocyte cell death due to oxidative stress, and 4) most likely enhanced immunity during the infection.
| Conclusions In conclusion, it can be stated that dietary selenium supplementation has improved performance of broiler chickens ever since it was introduced as an approved feed ingredient in 1974. Nevertheless, the modern broiler, which is a faster growing and significantly larger bird than the broiler of 1974, is at risk for distress from increased oxidative stress due to its higher rate of muscle growth and metabolism. It has been demonstrated that it is possible to improve the performance of the modern broiler with a simple replacement of inorganic selenium with organic selenium (Sel-Plex®), which allows for development of an improved redox status compared with sodium selenite (Edens, 2001). This study demonstrated that the use of Sel-Plex® also has a major influence on intestinal morphology that facilitates the potential for improved nutrient assimilation for digested food. This is especially important for chickens that might have been exposed to enteric viruses, such as the enteric reovirus that has a limited time of residence but long lasting effects on the performance of the bird. As the bird begins to recover from the viral infection in the gastrointestinal tract around 14 days after virus challenge (Rebel et al., 2004), it is important to resume rapid and efficient assimilation of nutrients. These results suggest that Sel-Plex®, more so than sodium selenite, has that potential at least in reovirus-infected chickens. These observations suggest that the improvements in performance of broiler chickens fed Sel-Plex® can be partially explained by improved integrity of the intestinal tract and potentially an improvement in the immune status as well. |
References
Authors: F.W. EDENS1, S. BURGOS1, J. READ-SNYDER1, A. CANTOR2 and A. PESCATORE2Balansky, R.M. and R.M. Argirova. 1981. Sodium selenite inhibition of the reproduction of some oncogenic RNA-viruses. Experientia 37:1194-1195.
Bassiouni, A.A., M.M. Zaki and M.M. Hady. 1990. Effect of vitamin E and selenium on the immune response of chickens against living Newcastle disease vaccine. Vet. Med. J. Giza 38:145-155.
Beck, M.A., P.C. Kolbeck, L.H. Rohr, Q. Shi, V.C. Morris and O.A. Levander. 1994. Benign human enterovirus becomes virulent in selenium-deficient mice. J. Med. Virol. 43:166-170.
Beck, M.A., Q. Shi, V.C. Morris and O.A. Levander. 1995. Rapid genomic evolution of a non-virulent Coxsackievirus B3 in selenium-deficient mice results in selection of identical virulent isolates. Nature Med. 1:433-436.
Beck, M.A., R.S. Ellsworthy, Y.S. Ho and F.F. Chu. 1998. Glutathione peroxidase protects mice from viral-induced myocarditis. FASEB 12:1143-1149.
Beck, M.A, H.K. Nelson, Q. Shi, P. Van Dael, E.J. Schiffrin, S. Blum, D. Barclay and O.A. Levander. 2001. Selenium deficiency increases the pathology of an influenza virus infection. FASEB 15:1481-1483.
Beck, M.A., O.A. Levander and J. Handy. 2003. Selenium deficiency and viral infection. J. Nutri. 133:1463S-1467S.
Beck, M.A., J. Handy and O.A. Levander. 2004. Host nutritional status: The neglected virulence factor. TRENDS Microbiol. 12:417-423.
Billard, W. and E. Peet. 1974. Sulfhydryl reactivity: mechanism of action of several antiviral compounds—selenocystine, 4-(2-propinyloxy)-ß-nitrostyrene, and acetylaranotin. Antimicrob. Agents Chemother. 5:19- 24.
Broome, C.S., F. McArdle, J.A. M. Kyle, F. Andrews, N.M. Lowe, C.A. Hart, J.A. Arthur and M.J. Jackson. 2004. An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. Am. J. Clin. Nutri. 80:154- 162.
Burk, R.F. and K.E. Hill. 1999. Orphan selenoproteins. BioEssays 21(3):231-237.
Cai, X., E. Block, P.C. Uden, X. Zhang, B.D. Quimby and J.J. Sullivan. 1995. Allium Chemistry: Identification of selenoamino acids in ordinary and selenium-enriched garlic, onion, and broccoli using gas chromatography with atomic emission detection. J. Agric. Food Chem. 43:1754-1757.
Combs, Jr., G.F. 2001. Selenium in global food systems. Brit. J. Nutr. 85:517-547.
Edens, F.W. 2001. Involvement of Sel-Plex® in physiological stability and performance of broiler chickens. In: Science and Technology in the Feed Industry, Proceedings of Alltech’s 17th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). Nottingham University Press, UK, pp. 349-376.
Edens, F.W. and K.M. Gowdy. 2004. Selenium sources and selenoproteins in practical poultry production. In: Nutritional Biotechnology in the Feed and Food Industries, Proceedings of Alltech’s 20th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). Nottingham University Press, UK, pp. 35-55.
Fan, Y., J. Croom, V. Christensen, B. Black, A. Bird, L. Daniel, B. McBride and E. Eisen. 1997. Jejunal glucose uptake and oxygen consumption in turkey poults selected for rapid growth. Poult. Sci. 76:1738-1745.
Federal Register. 2000. Food additive permitted in feed and drinking water: Selenium yeast. Fed. Reg. 65(109):35823-35824. June 6.
Federal Register, July 17. 2002. Food additive permitted in feed and drinking water: Selenium yeast. Fed. Reg. 67(137):46850-46851.
Field, C.J., I.R. Johnson and P.D. Schley. 2002. Nutrients and their role in host resistance to infection. J. Leuk. Biol. 71:16-32.
Foster, H.D. 2003. Why HIV-1 has diffused so much more rapidly in Sub-Saharan Africa than in North America. Med. Hypoth. 60:611-614.
Guy, J. 1998. Virus infections of the gastrointestinal tract of poultry. Poult. Sci. 77:1166- 1175.
Jensen, L.S. 1991. Interaction of vitamin E with diseases in poultry. In: Vitamin E in Animal Nutrition and Management: A BASF Reference Manual (M.B. Coelho, ed). BASF Corp. Parsippan, NJ, USA, pp. 205-213.
Juyal, R., M. Osmamy, R.E. Black, U. Dhingra, A. Sarkar, P. Dhingra, P. Verma, D. Marwah, R. Saxena, V.P. Menon and S. Sazawal. 2004. Efficacy of micronutrient fortification of milk on morbidity in pre-school children and growth – a double blind randomized controlled trial. Asia Pac. J. Clin. Nutri. 13:44S.
Kelly, M.P. and R.F. Power. 1995. Fractionation and identification of the major selenium compounds in selenized yeast. J. Dairy Sci. 78(Suppl. 1):237.
Leeson, S. and J.D. Summers. 1991. Commercial Poultry Nutrition. University Books, Guelph, Ontario, Canada.
Levander, O. 1986. Selenium. In: Trace Elements in Human and Animal Nutrition (W. Mertz, ed). Academic Press, Orlando, FL, USA, pp. 209-280.
Look, M.P., J.K. Rockstroh, G.S. Rao, K.A. Kreuzer, U. Spengler and T. Sauerbruch. 1997. Serum selenium versus lymphocyte subsets and markers of disease progression and inflammatory response in human immunodeficiency virus-infection. Biol. Trace Elem. Res. 56:31-41.
Lyons, G., J. Stangoulis and R. Graham. 2003. Nutriprevention of disease with high selenium wheat. J. Australasian Coll. Nutri. Environ. Med. 22:3-9.
Mahan, D.C. 1995. Selenium metabolism in animals: What role does selenium yeast have? In: Biotechnology in the Feed Industry, Proceedings of Alltech’s 11th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). Nottingham University Press, UK, pp. 257-267.
Mahmoud, K.Z. and F.W. Edens. 2003. Influence of selenium sources on age-related and mild heat stress-related changes of blood and liver glutathione redox cycle in broiler chickens (Gallus domesticus). Comp. Biochem. Physiol. B 136:921-934.
Marin, F.P., S. Rivera, G. Finol and Y. Mavarez. 2003. Aflatoxin ß1, selenium, and Saccharomyces cerevisiae in the immune response of broiler chickens in Zulia state, Venezuela. Revis. Cient. Facultad Ciencias Vet. 13:360-370.
Motulsky, H. 2005. Intuitive Biostatistics. Oxford University Press, New York, NY, USA.
Nelson, H.K., Q. Shi, P. Van Dael, E.J. Schiffrin, S. Blum, D. Barclay, O.A. Levander and M.A. Beck. 2001. Host nutritional selenium status as a driving force for influenza virus mutations. FASEB 15:1846-1848.
Oxford, J.S. 1973. An inhibitor of the particle-associated RNA-dependent RNA polymerase of influenza A and B viruses. J. Gen. Virol. 18:11-19.
Panda, S.K. and A.T. Rao. 1994. Effect of vitamin E-selenium combination on chickens infected with infectious bursal disease virus. Vet. Rec. 134:242-243.
Rebel, J.M.J, J.T.P. van Dam, B. Zekarías, F.R.M. Balk, J. Post, A. Flores Minambres and A.A.H.M. ter Huurne. 2004. Vitamin and mineral content in feed of breeders and their progeny: effects on growth, feed conversion, and severity of malabsorption syndrome of broilers. Brit. Poult. Sci. 45:201-209.
Robertson, Md., G.E. Wilcox and F.S.B. Kibenge. 1984. Prevalence of reoviruses in commercial chickens. Aust. Vet. J. 61:319-322.
Sappey, C., S. Legrand-Poels, M. Best-Belpomme, A. Favier, B. Rentier and J. Piette. 1994. Stimulation of glutathione peroxidase activity decreases HIV type 1 activation after oxidative stress. AIDS Res. Human Retrovir. 10:1451-1461.
SAS® Institute. 1996. A User’s Guide to the Statistical Analysis System. SAS® Institute, Cary, NC, USA.
Schrauzer, G.N. 2000. Nutritional selenium supplements: Product types, quality, and safety. J. Am. Coll. Nutr. 20:1-4.
Schrauzer, G.N. and J. Sacher. 1994. Selenium in the maintenance and therapy of HIVinfected patients. Chem.-Biol. Interact. 91:199-205.
Singh, H., S. Sodhi and R. Kaur. 2006. Effects of dietary supplements of selenium, vitamin E, or combinations of the two on antibody responses of broilers. Brit. Poult. Sci. 47:714- 719.
Spallholz, J.E. 1997. Free radical generation by selenium compounds and their pro-oxidant toxicity. Biomed. Environ. Sci. 10:260-270.
Spencer, R.P. and M. Blau. 1962. Intestinal transport of selenium 75selenomethionine. Science 163:155-156.
Surai, P.F. 2006. Selenium and immunity. In: Selenium in Nutrition and Health. Nottingham University Press, Nottingham, UK, pp. 213-278.
Swain, B.K., T.S. Johri and S. Majumdar. 2000. Effect of supplementation of vitamin E, selenium, and their different combinations on the performance and immune response of broilers. Brit. Poult. Sci. 41:287-292.
Swick, R.A. 1995. Importance of nutrition and health status in poultry. American Soybean Association Publication MITA(P) No. 083/12/94 (vol. P015.1995) http://www.asasea.com.
Terada, A., M. Yoshida, Y. Seko, T. Kobayashi, K. Yoshida, M. Nakada, K. Nakada, H. Echizen, H. Ogata and T. Rikihisa. 1999. Active oxygen species generation and cellular damage by additives of parenteral preparations: Selenium and sulfhydryl compounds. Nutrition 15:651-655.
Yu, M.W., I.S. Horng, Y.C. Chiang, Y.F. Liaw and C.J. Chen. 1999. Plasma selenium levels and the risk of hepatocellular carcinoma among men with chronic hepatitis virus infection. Am. J. Epidemiol. 150:367-374.
Yu, S.Y., Y.J. Zhu and W.G. Li. 1997. Protective role of selenium against hepatitis B virus and primary liver cancer in Qidong. Biol. Trace Elem. Res. 56:117-124.
1 North Carolina State University, Raleigh, North Carolina, USA
2 University of Kentucky, Lexington, Kentucky, USA









.jpg&w=3840&q=75)