1. Introduction
The process of industrialization of the poultry sector has achieved a high degree of automation, however, such progress does not translate into an improvement in the quality of the meat, rather, they contribute to increase the microbial load of the poultry carcasses gaining importance today Campylobacter spp, among other microorganisms involved in food-(ETAs) transmitted diseases [1]. Infections among species of the family or Campylobacteraceae have campylobacteriosis Campylobacter enteritis, considered the most important inpublic health, its main agents are C. jejuni and C. coli (can also cause systemic infections and complications after infection; Agents Guillain Barre GBS) the impact of public health campylobacteriosis is increasing [2].
The genus Campylobacter, is dated and comprising gram negative bacilli curved (gullwing), with polar Flagellation, microaerophilic, do not use sugar, but energy of amino acids, are thermotolerant species 42ºC, zoonotic, birds are an important reservoir, are the causative agents of diarrhea in humans (first cause in industrialized countries and second or third cause in Latin America). They have been isolated 25 species and 9 subspecies [2].
Campylobacter spp, requires optimal growth conditions(5% O2, CO2 3-15% and 85% N), mentioned three species of thermophilic Campylobacter causing significant healthproblems in humans (C. jejuni, C. coli, C. laridis) and outnumber cases of enteritis caused by Salmonella sp. and Shiguellas sp. [3]. According Seminar INFAL 2015 [4],following up on time pathogens Campylobacterspp, it was reported steadily increasing in England and Wales between 1997 and 2002 beating Rotavirus and Salmonella; even as [5], at the Ninth International Congress of Tropical Medicine and Health held in Sweden indicated that cases of campylobacteriosis were more than doubled between 1988 and 2013, i.e. 3127 cases in 7499, an issue that has worsened since 1995. The center for Disease Control and Prevention (CDC) [6], in its surveillance program of Campylobacter spp said that for this year experienced an increase of 14% over the years 2006-2008; noting that for every case of campylobacteriosis reported, there are 30 undiagnosed cases
Domestic and wild animals serve as host to the bacteria, causing pollution 90% of chicken carcasses during processing ([7]. Studies conducted in Venezuela, specifically in the Aragua state by [8] reported Campylobacter spp in samples of whole, breasts, thighs and wings 75%, 95.83%, 83.33% and 70.83% respectively chickens, found in the first three samples indicated conditions≥500 units colony forming (CFU) per ml. Other researchers [9], [10] found Campylobacter spp in a 70.83% in liver and 48.95% inchicken gizzards; likewise [11] in Chile reported that poultry liver 95.1% recorded in Brazil isolation and a percentage that goes from 13.5 to 78.7.
Campylobacteriosis is a zoonotic disease caused by eating food contaminated with bacteria of the genus Campylobacter as raw milk, seafood, poultry and other animals (cross-contamination), as well as in the untreated water; It occurs most often in children and young people, where it was reported 24.08 and 10.54% respectively in 2012 and in adults between 20 and 64 years and over reported 14.54 and 15.26. It is characterized by diarrhea, cramping, abdominal pain, fever, nausea and vomiting, some sequels neurological in which the syndrome Gillian Barre Syndrome (GBS) and Miller Fisher syndrome (MFS) [12].
The aim of the study was to analyze the prevalence of Campylobacter spp in whole chickens packed and viscerathree lots traded in the municipality Girardot Aragua, Venezuela State through a quick test plate, and infer risk to which it is subjected consumer; also alert the population and institutions to exercise an active epidemiological surveillance in terms of ensuring public health.
2. Methodology
2.1. Taking the Sample
In this descriptive and cross-sectional research was acquired by a non-probability sampling four chickens in each of three lots, weekly reach outlets increased demand in the municipality Girardot Aragua state, Venezuela; in June 2013, to give a total sample of 48 samples of whole chickens and viscera; these were transported in ice coolers to the laboratory of Public Health of the Central University of Venezuela, Maracay Estado Aragua for a time did not exceed 30 minutes. Samples were collected and recorded in the collection protocol and processed under strict hygiene materials, equipment and surfaces used.
2.2. Procedures
Simple rapid test used in detecting the presence Campylobacter spp, which is a rapid, commercial, easy to prepare, sensitive and useful test in processing large numbers of samples, was used following the indications for use [13],
[14] as described below. i. Rifampicin Additive
0.25 gg of rifampicin, diluted in 60-80 ml of alcohol, add
distilled water to a final volume of 100ml.
ii. Hemin Additive
Mix 10ml of NaOH in 90 ml water, add 0.4 g of hemin continuously stir to homogenize the mixture.
Phosphate buffered water for dilutions
Buffered water for dilution: 1.25 ml of phosphate buffer stock solution was mixed into one liter of distilled water, sterilized by autoclaving at 121°C for 15 min. It was prepared and distributed in bottles of the required amount of dilution solution to wash the samples and viscera.
All reagents described are preserved under refrigeration.
2.3. Washing of Carcasses and Offal
Chicken or viscera within a sterile bag was introduced, it was added 500 ml of phosphate buffered solution, stirred manually about 2 min, for washing the sample.
2.4. Procedure for Conducting Rapid Test Plate Used for Whole Chicken and Viscera
(1) Nine (9) ml of distilled water to the vial (half Simplate) was added, homogenized and 0.025 ml was added 0.040 ml Rifampicin and Hemin. After washing 1ml of the sample, was added. The vial contents was placed in the center of the plate evenly distribute must endeavor all wells
(2) Seeded plates were placed one upon the other in the grid carrier plates and the jar microaerophilic, microaerophilic embedded envelope (oxoid®), the tab holder on located on the side of the grid plates porta pitcher, was introduced into the jar, quickly covered and incubated in the oven at 42°C for 48 hours.
2.5. Interpretation of Results
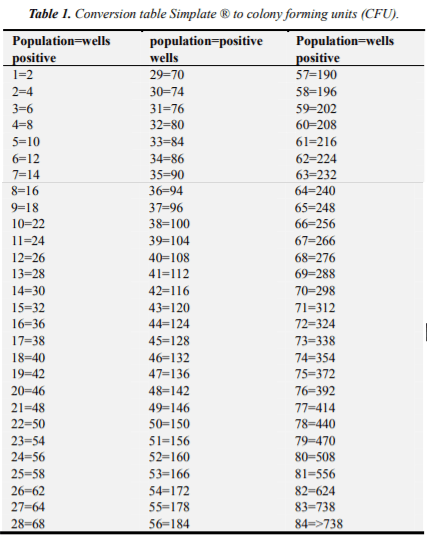
All red little wells were counted, they are presumably positive for Campylobacter spp. Subsequently, he placed in a UV lamp, were counted and the number of wells with fluorescence was recorded and subtracted the number of red wells, obtained in the previous step. The result started Simplate conversion table and the number of colony forming units (CFU) was obtained for that sample. The results descriptive statistical analysis and analysis matching underwent Kendal.
3. Results
Bacteriological study showed the presence of Campylobacter spp, where rapid test detected a 68.75% inthe 48 samples of whole chickens, 81.25% (39/48) of Campylobacter spp positivity and viscera (33/48). The resultsin percentage values of positivity in chickens and viscera, found in different batches of product analyzed, are presented discriminately, as shown:
3.1. Whole Chickens
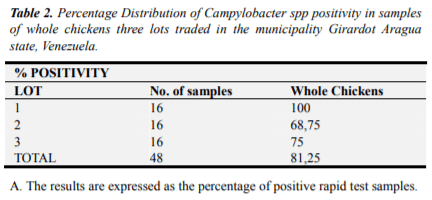
In Table 2, the results of the 48 samples of whole chickens are indicated, the rapid test detected the presence Simplate® 100% (16/16) positive for Campylobacter spp, for lot 1; 68.75% (11/16) lot 2 and 75% (12/16) lot 3.
3.2. Viscera
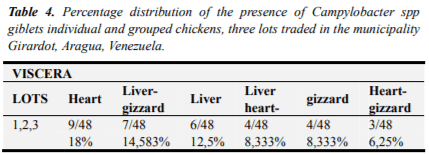
48 viscera of chickens tested positive lot 1 was 100% (16/16) Campylobacter spp, for lot 2, 50% (8/16) and 56.25% (9/16) Lot 3 (Table 3). Regarding the comparison of the percentages of Campylobacterspp detected in three commercial chicken flocks and viscera, a high level of presence of the bacteria found in lot 1, represented by 100% (16/16) detection, both as whole chickens viscera; in descending order lot 3, presented 75% of presence (12/16) in the whole chickens and viscera 56.25% (9/16) and finally batch 2 was located with percentages Campylobacter spp 68, 75% (11/16) in chicken samples and 50% (8/16) for samples of viscera.
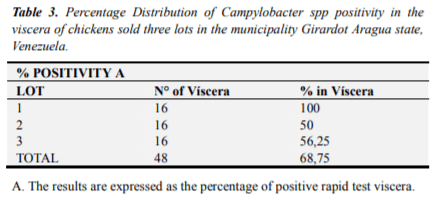
They were distributed individual and grouped different organs analyzed between the three commercial lots, noting that the viscera heart (18%) had the highest percentage of presence of Campylobacter spp, followed by liver-gizzard (14.58%), liver (12,5%), liver-heart (8.33%), gizzard (8.33%) and the lowest percentage stood the group consisting of viscera gizzard heart-values of 6.25%. (Table 4)
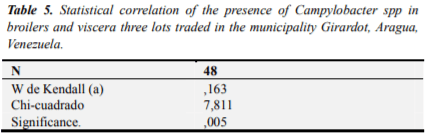
By measuring the degree of matching the extracted data from the study of the UFC variables in chicken and UFC viscera, we can see that the statistic Kendall as concordance coefficient for this study is under 0,163 and significance is (*) P<0.005, which indicated that between the two variables was statistically significant association, this statistical association reflects the correlation between the UFC and UFC variables in whole chickens in viscera (Table 5).
Regarding the means of the colony forming units of Campylobacter spp detected in whole chickens and viscera,obtained in three batches of marketed products submitted to the detection of Campylobacter spp, the values were below the infective dose and found to lot 1 showed higher recovery values Campylobacterspp the infective dose (≥500 CFU) represented by 43.75% (7/16) for chickens and 25% (4/16) to the viscera, in lot 2 only 12.5% (2/16) for each of the samples of meat and offal presented above infective dose values, finally lot 3 with 6.25% (1/16) values greater than 500 UFC he found both in the flesh and viscera.
4. Discussion of Results
It may show that the rapid test detected the highest percentage of positivity of 81.25%, in whole chickens among the three batches of chicken carcasses studied (Table 2). These results can be attributed to the entry of birds to the processor from poultry production units with low management level, where feces of these birds, contaminated feathers, skin and different tanks used for processing plant, as well as the post evisceration washing water leaving the microorganism trapped within the abdominal cavity and the skin as also, noted [2] and [4]. These results are in more than 63% reports obtained from fresh chicken expended in Costa Rica range [3], [5], [11], to 71% reported in England by [2], [5], [14]. In Venezuela they were presented inferior results, 75% presence of campylobacter spp detected in whole chickens or their parts in Aragua state [8].
In relation to the viscera had a percentage lower than the results found in whole chickens in a 68.5% positivity, this may be due to more rigorous control in this area line profit in the different processing plants (Table 3 ). 68.5% These results are in lower proportions to 70.83% found by [8], liver and gizzard samples collected in expense of Aragua, Venezuela and equally low state when compared with those reported by [2], in liver of birds in Chile that reach 95.1% isolation of Campylobacter and Brazil that reach up to 78.7%.
These results reflect like other previous research that the Campylobacter spp is present in the birds that come tobeneficiary plants and can often survive the different stages of processing, as specified [4] and should be taken programs into account in quality assurance
By measuring the degree of matching the extracted data from the study of the UFC, variables in chicken and giblets (Table 5). It is considered a new data, because in the literature reviewed no information related to the correlation of CFU of Campylobacter spp in broilers and viscera with which to conduct comparison found. The correlation evidenced in this study, is indicative of improper handling of carcasses and offal and reflects the degree of cross-contamination at different stages at the level of processing plants or benefit of birds as is argued by [2] and [5].
Regarding the means of the colony forming units of Campylobacter spp detected in whole chickens and viscera,obtained in three batches marketed analyzed; [8] reported 20% of samples chicken carcasses with over 500 U. F. C, inferior results to those found in this research for chicken samples values. This evaluation can deduce that chicken meat as a product of high consumption becomes for those who ingest it in a high-risk food, important in triggering the disease, since the largest number of UFC in relation to the viscera, if the handling and preparation of food at home is inadequate.
5. Conclusions
Detection of Campylobacter spp was 81.25% for chickens and 68.50% for the viscera and found to lot 1 had the highest levels of contamination 100% for both samples, followed by batch 3 75% and 68.75% respectively for broilers and viscera, and the lowest values were found detection in batch 2 with 68.75% (chickens) and 50% (viscera), which is indicative of low hygienic conditions operability of chicken processing plants in the plots studied.
Statistical results indicate a correlation between samples of chickens and viscera of the lots analyzed, indicative of the association between colony forming units found in chickens with those found in the viscera and based on the infective dose of bacteria to some individuals (≥500 CFU) in order to cause disease risk was represented by 43.75% chickens and 25% of the viscera of lot 1, and 12.5% for the samples studied in the lot 2 and finally 6.25% of the samples lot 3.
It is observed that the hygienic conditions of the plants beneficiary chickens brands evaluated, lack of hygiene and control of critical points in the processing of products intended for human consumption.
This article was originally published in International Journal of Microbiology and Biotechnology 2017; 2(1): 7-11. http://www.sciencepublishinggroup.com/j/ijmb. doi: 10.11648/j.ijmb.20170201.12.