Forms of dietary selenium and drivers of efficacy: From genes to biological effects
Published: April 6, 2020
By: Dr Michele De Marco and Dr Mickael Briens, Adisseo

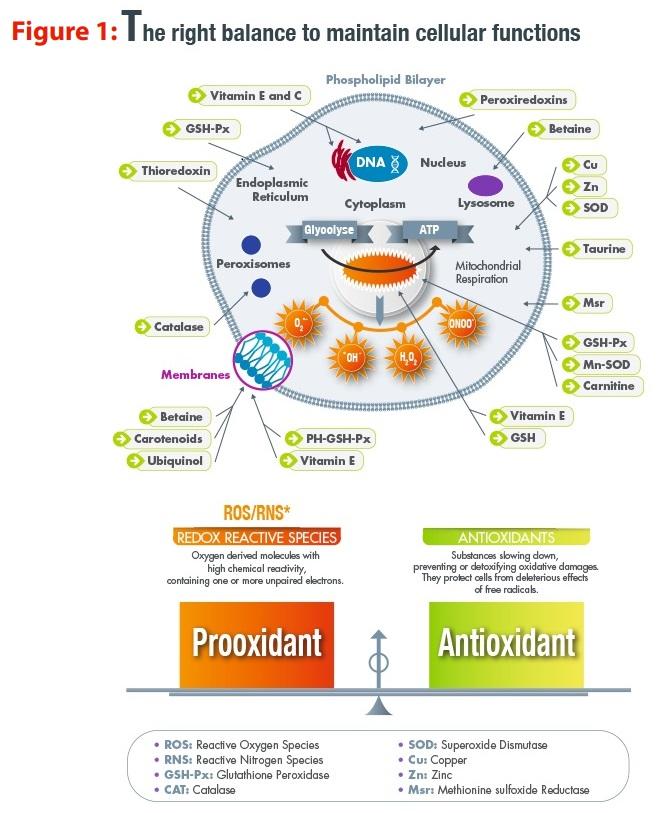
A variety of oxidative reactions occur continuously within our cells in a dynamic environment. One common type of reaction – reduction-oxidation or redox reaction – plays a vital role in maintaining cellular functions. During these reactions, one couple loses electrons (becomes oxidised) and another gains the same electrons (becomes reduced). As a result, our cells have to maintain a delicate redox balance between the various macromolecules that comprise them.
These pro-oxidants, which are referred to as reactive oxygen species (ROS) and reactive nitrogen species (RNS), mainly originate from the oxygen metabolism in the mitochondria. They are signalling molecules that can trigger various systems, but can cause damage to the cell if produced in excess.
On the other hand, antioxidant systems are represented by exogenous molecules (vitamins, polyphenols) that have ROS scavenging activities and endogenous systems (mainly enzymes such as superoxide dismutase, catalase, glutathione peroxidase and thioredoxin reductase). Selenium (Se) is part of this second group as it is a constituent of a small group of enzymes (Figure 1).
From selenium to selenocysteine
Selenium is a unique essential trace element in biology as it is the core constituent of two amino acids, whereas other trace elements are added to proteins later as cofactors. These two amino acids are selenomethionine (SeMet) and selenocysteine (SeCys). SeMet is homologous to methionine with the only, but important, difference that Se replaces sulphur. SeMet is considered a safe storage form of Se and does not induce structural or functional modifications of proteins. Cells are not able to distinguish between methionine and SeMet during protein synthesis.
SeCys is the true biologically active form of Se. This amino acid is incorporated, under gene control, into the so-called selenoproteins through specific and dedicated translation machinery. The particular biological functions of this amino acid and gene encoding incorporation validate its classification as an amino acid; it is considered the 21st.
Functions of selenoproteins
Most higher animals, such as mammals and birds, have 25 selenoproteins, under the control of 25 genes. Selenoproteins have all the features of proteins, such as gene encoding, but also have more specific ones, such as the presence of SeCys amino acid, which is located at the catalytic site of the enzyme. These enzymes have well-described antioxidant functions and play important roles in cell signalling as well as in ROS and RNS detoxification.
Glutathione peroxidases and thioredoxin reductases are well-characterised selenoproteins that play similar roles in detoxifying ROS as H2 O2 produced during oxidative stress. The knowledge regarding other selenoproteins is more recent; they are still being studied and this could explain the many claimed attributes of Se, such as its effect on fertility and immunity.
Selenium for animal nutrition Selenium can be added to livestock diets in either inorganic forms (sodium selenite, SS), organic forms such as Se-yeast (yeast enriched in SeMet), or pure chemically synthesised Se forms such as hydroxyselenomethionine (HMSeBA), L-SeMet or ZnL-SeMet. Recent advances in Se biochemistry have provided a deeper understanding of the principal differences in absorption and metabolism between these forms.
The advantage of feeding animals organic Se over inorganic sources is that SeMet is metabolised as a constituent of the methionine pool. This characteristic leads to creating a storage depot of Se inside the animals as SeMet that can be released later. In fact, selenite or different dietary forms of SeCys cannot constitute Se storage in the body, and any excess Se in these forms are therefore immediately excreted to prevent toxicity from occurring.
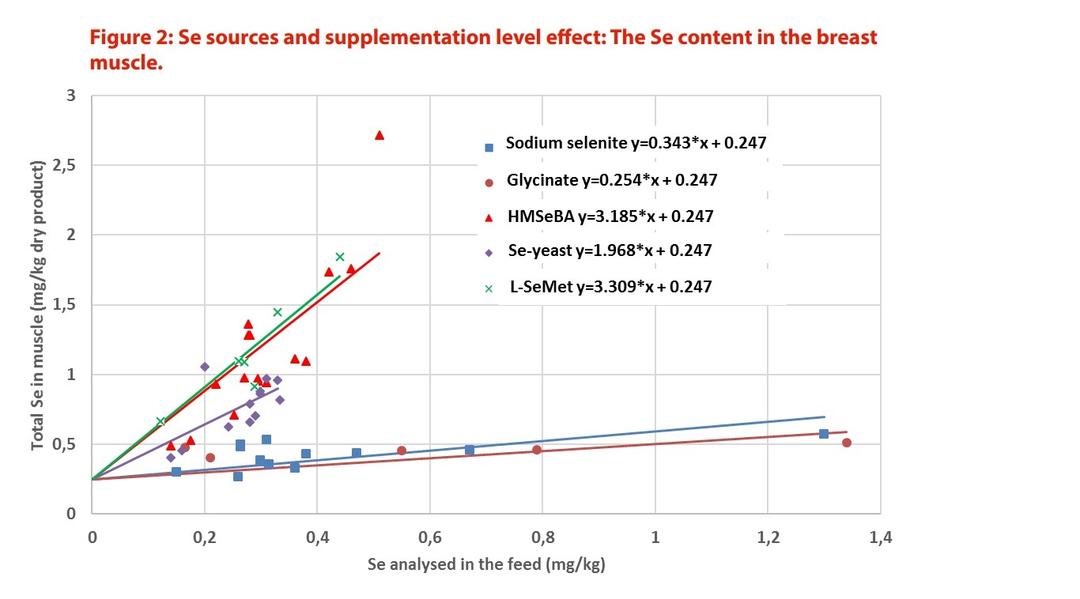
For these reasons, the scientific community recognises that the bio-efficacy of different Se additives may be assessed by considering Se deposition in different animal tissues. Muscles, in particular, are reliable biomarkers. It has been shown, in several publications and species, that the proportion of Se in the form of SeMet is the driver of efficacy of Se additives. Simon et al. (2013) concluded that the difference in efficiency of Se sources can be related to their different SeMet contents.
Furthermore, a meta-analysis that integrated the results of 13 controlled randomised broiler chicken studies, where different Se supplements were evaluated (none, SS, Se-glycinate, Se-yeasts, HMSeBA, SeMet), concluded, on the basis of Se muscle deposition, that pure HMSeBA and SeMet were more efficient in increasing the muscle Se concentration than other Se source products (Jachacz et al., 2017 [Figure 2]).
Regulation of selenogenome
Thanks to gene sequencing, recent technologies now enable a clearer understanding of the biological efficacy of Se. The gene expression of all the selenoproteins in different tissues is becoming an innovative and more intriguing way of evaluating the bio-efficacy of Se supplements. This implies that Se is a key element in proteasome activity control. There might be a feedback recognition mechanism of SeMet as a source of Se for selenoprotein synthesis.
Some amino acids inside muscle proteins can be oxidised under stress conditions.
This triggers an increase in proteasome activity to degrade such proteins (Pajares et al., 2015) and releases SeMet, which becomes available as an additional source of Se for selenoprotein synthesis.
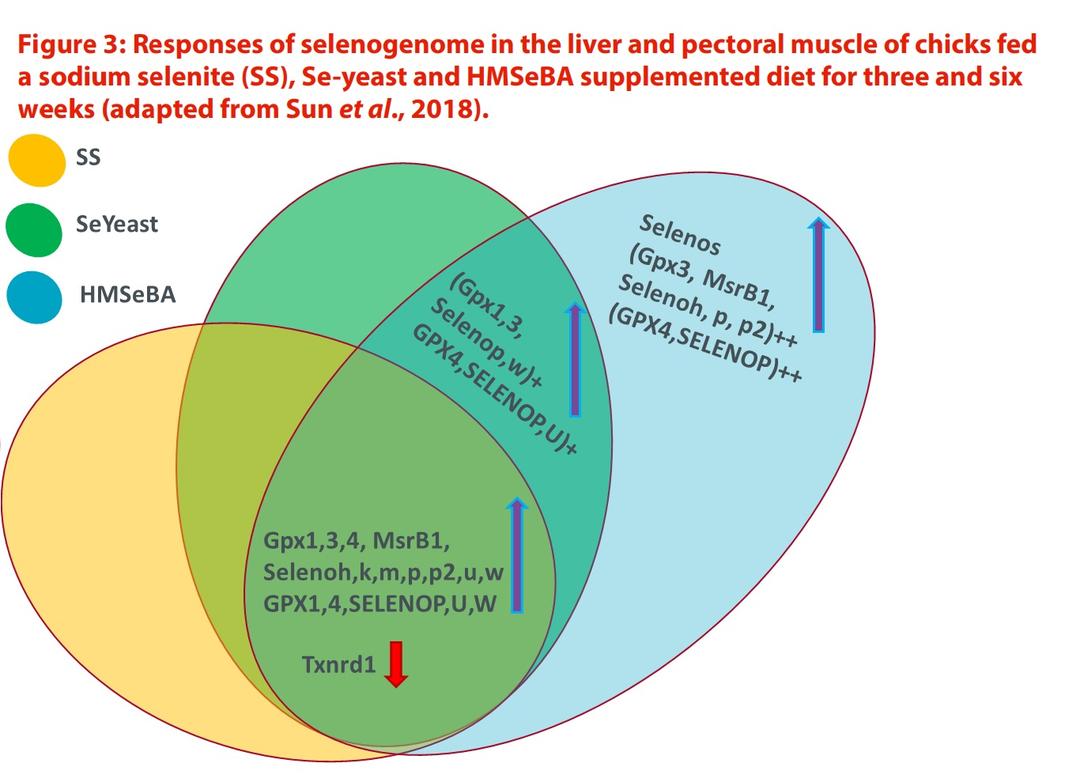
Sun et al. (2018) showed that the dietary supplementation of SS, Se-yeast or HMSeBA upregulated GPX1, GPX3, GPX4, MsrB1, SELENOH, SELENOK, SELENOM, SELENON, SELENOP, SELENOP2, SELENOU and SELENOW mRNA, and downregulated TXNRD1 mRNA in the tissues (liver and breast muscle) of chicks. Their effects on the assayed GPX1, GPX4, SELENOP, SELENOU and SELENOW production and GPX activity were consistent with their respective mRNA changes.
Compared with SS, Se-yeast and HMSeBA both elevated GPX1, GPX3, SELENOP and SELENOW mRNA, the production of GPX4, SELENOP and SELENOU, and GPX activity in the two tissues. However, only HMSeBA upregulated SELENOS mRNA and TXNRD activity in a Se deficiency diet, and elevated GPX3, MsrB1, SELENOH, SELENOP and SELENOP2 mRNA, and the production of GPX4 and SELENOP, compared with SS and Se-yeast supplementations (Figure 3).
In conclusion
The role of Se in antioxidant functions is well known. Mineral forms are also known to be less bioavailable than organic Se forms. There is now much evidence that the SeMet proportions of Se are the main drivers of efficacy in organic form. This has been confirmed through the traditional way of assessing efficacy, i.e. tissue Se enrichment, as well as through the measurement of the activation of Se biological systems, i.e. selenoprotein gene expression.
As animals are not able to synthesise SeMet, its provision through a diet is a key strategy to help fight commercially relevant stress. Indeed, under oxidative stress conditions, when an increased selenoprotein expression requires additional Se, the body’s Se reserves help maintain an effective antioxidant defence. Selenium, via selenoprotein expression, is involved in the protection against oxidative stress and the regulation of various cell functions. Therefore, increased muscle reserves of Se can enhance livestock resistance to stress and diseases.
Related topics
Authors:
Adisseo
Adisseo
Influencers who recommended :
Damien P. Prévéraud, Marcio CeccantiniRecommend
Comment
Share

Would you like to discuss another topic? Create a new post to engage with experts in the community.









.jpg&w=3840&q=75)