Effect of dietary antioxidants on the incidence of pulmonary hypertension syndrome in broilers
Pulmonary hypertension syndrome (PHS) followed by ascites is a major cause of economic loss in the broiler industry. The main mechanism responsible for ascites in broiler chickens is an increase in intravascular hydrostatic pressure occurring secondary to right ventricular failure.
Development of cardiac failure in an apparently healthy young broiler chicken is affected by a range of factors including genetic selection for rapid growth rate, high feed efficiency and a large pectoral muscle mass; all of which increase oxygen demand.
The low ratio between lung volume and body weight in the modern chicken is responsible for the inability of the respiratory system to respond to elevated oxygen needs, which leads to hypoxia and respiratory acidosis. During hypoxia, various mechanisms increase free radical production. The objective of this study was to verify the effects of antioxidants (dietary vitamin E, organic and inorganic selenium (Se)) on the incidence of pulmonary hypertension syndrome (PHS), serum glutathione peroxidase (GSHPx), blood gases, electrolytes and growth performance in cold-stressed broilers.
The experimental design was a 2x3x4 factorial; with a starter feed containing either 50 or 250 IU vitamin E per kg, 0.3 ppm Se (organic selenium yeast, Sel-Plex), and 0.3 ppm or 0.6 ppm Se from sodium selenite. Vitamin E levels were decreased to 30 and 150 IU/kg, and 20 and 100 IU/kg for the grower and finisher diets, respectively. Broilers were fed ad libitum and subjected to cold stress beginning at 21 days of age with temperatures decreasing to 19, 14 and 11.5°C over a two week period. Weekly body weights, daily feed consumption and mortality were recorded and right ventricular/ total ventricular weight ratios (RV/TV), blood gases, electrolytes and GSH-Px at 47 days of age were measured. The combination of high vitamin E and organic selenium (Sel-Plex) reduced mortality associated with PHS in cold stressed broilers from 10 to 0.90%. Protective effects of antioxidants on cellular membranes were demonstrated by higher hemoglobin concentration, increased GSH-Px activity, improved blood gas parameters, bicarbonate, pH and normal acid-base status in birds given the high vitamin E level and Sel-Plex organic selenium. Lower mortality and better feed conversion appeared to be the result of improved antioxidant status in the birds.
Introduction: the pulmonary hypertension syndrome
Pulmonary hypertension syndrome (PHS), also called ascites, is a major source of economic loss in the broiler industry. Olkowski et al. (1996) showed that PHS was responsible for 2% of the death loss and 0.35% of condemnations in Canada. In 1994, economic losses due to ascites were estimated at USD$12 million and USD$100 million for Canadian and American industries, respectively (Odom, 1993; Olkowski et al., 1996). Conservative estimates of yearly economic cost of PHS worldwide would approach USD$500 million.
The main mechanism responsible for ascites in broiler chickens is an increase in intravascular hydrostatic pressure occurring secondary to right ventricular failure. In response to increased pressure, the transudate leaks out of blood vessels and accumulates in the abdominal cavity, hence the ascites. Development of cardiac failure in an apparently healthy young broiler chicken is explained by a series of factors such as genetic selection for a rapid growth rate, high feed efficiency and a large pectoral muscle mass, all requiring high oxygen levels (Chabot, 1992; Boulianne, 1993; Julian, 1993; Julian et al., 1995; Maxwell et al., 1986a; 1986b; Maxwell and Robertson, 1997; 1998).
The modern chicken has small lung volume:body weight ratio, causing an inability of the respiratory system to respond to the broiler’s elevated oxygen needs, which leads to hypoxia and respiratory acidosis (Table 1) (Vidyadaran et al., 1990; Peacock et al., 1990; Reeves et al., 1991; Chabot, 1992; Julian, 1989; 1993).
| Table 1. Blood parameters of broilers with and without PHS syndrome.* |
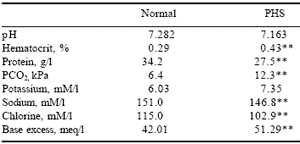 |
| *Chabot, 1992. *P<0.05 **P<0.01 |
Impact of hypoxia on free radical production
During hypoxia, various mechanisms increase free radical production including lipid peroxide, hydrogen peroxide and superoxide (Figure 1). Tissue damage secondary to hypoxia attracts white blood cells, which in turn release more free radicals, causing further damage (McCord, 1985; Halliwell and Gutteridge, 1990; Bottje et al., 1995a). Maxwell et al. (1986a) and Enkvetchakul et al. (1993) have observed inflammatory cell infiltration in various tissues of PHS chickens. Acidosis will also affect cellular membrane integrity and reduce free radical elimination, hence exacerbating the negative effect of free radicals.
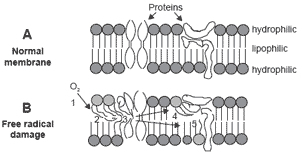 |
| Figure 1. Lipid peroxidation (adapted from Bottje et al., 1995a). |
Higher plasma lipid peroxide values have been reported in PHS broilers (Bottje et al., 1995a). Maxwell et al. (1986a) have hypothesized that autogenous antioxidant levels were lower in PHS broiler chickens. This theory is supported by the findings of Enkvetchakul et al. (1993), who demonstrated lower pulmonary and hepatic tocopherol and glutathione levels in PHS broilers (Figure 2).
The role of these antioxidants is to transform free radicals into harmless compounds, stopping the lipid peroxidation process. Vitamin E (tocopherol) and GSH-Px are excellent antioxidants. First, vitamin E reduces the peroxyl radical to an oxidized lipid. These lipids are then converted by GSH-Px to a lipid alcohol, which contributes to lipid repair (Figure 3) (Maddaiah, 1990; Machlin, 1991; Yu, 1994; Bottje et al., 1995a). Formation of one type of GSH-Px is dependent upon the presence of selenium. This is why selenium and vitamin E work in synergy to protect cellular membranes. Vitamin E in prevention of PHS Subcutaneous vitamin E implants have been shown to reduce mortality in PHS chickens (Bottje et al., 1995b).
In this study control broilers were provided normal ventilation but others maintained under low ventilation conditions to induce PHS were randomly assigned to nonimplanted, placebo, or vitamin E-implanted groups. The vitamin E implant released a total 15 mg "-tocopherol over the first three weeks of age. Tissues and blood samples were obtained at 3 and 5 weeks of age from birds with and without PHS. Cumulative mortality at 5 weeks was lowered by "-tocopherol with mortality rates of 3.6, 4.2, 11.9 and 11.8% for controls (maintained under normal ventilation conditions), vitamin E, non-implanted and placebo treatments, respectively. The PHS birds had lower body weights, higher hematocrit, lower "-tocopherol and glutathione (GSH) concentrations in liver and lung tissue. Oxidative stress was indicated by elevated plasma lipid peroxides and lower oxidized GSH in liver and erythrocytes. These results indicate that the vitamin E implant was effective in reducing PHS mortality in broilers. In contrast, vitamin E supplementation of the diet had no effects on performance and mortality (Bottje et al., 1997). The maximum vitamin E dose used for this study was 87 IU/kg, a dose similar to recommended commercial levels. We hypothesized that this level was too low to have a significant impact and that it would be necessary to test higher vitamin E doses.
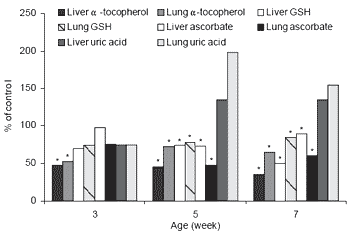 |
| Figure 2. Liver and lung "-tocopherol, GSH, ascorbate and uric acid in PHS broilers. (* means differ, p<0.05; Enkvetchakul et al., 1993). |
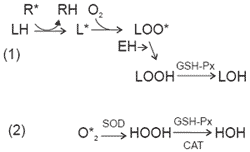 |
| Figure 3. Protection of membranes by antioxidants (Bottje et al., 1995a). |
Selenium in prevention of free radical production
One major role of selenium is as a part of the GSH-Px structure. Inorganic selenium (sodium selenite) is the most common form of selenium added to animal feed. Inorganic selenium is passively absorbed in the intestine then reduced to selenide form in the liver where it undergoes an enzymatic process with cysteine to form selenocysteine. GSH-Px, along with the other selenoproteins, is composed of selenocysteine molecules. This mechanism of selenocysteine formation in liver is rapidly saturated when dietary inorganic selenium is increased above 0.3 ppm (Meyer et al., 1981; NRC, 1983; Pehsron, 1993; Mahan, 1995). Animals can store a small quantity of inorganic selenium in different tissues (muscle, liver, etc.,) as a source of non-functional selenium that may eventually be diverted to form biologically important compounds. Because metabolic mechanisms to convert inorganic selenium into organic compounds are rapidly exhausted and body storage is limited, excess inorganic selenium is eliminated in urine and feces. While inorganic selenium is capable of providing selenium for GSH-Px synthesis, its potential prooxidative effects may be a disadvantage. Several authors have pointed out this disadvantage on noting an increase in lipofuscin pigments in mouse liver tissue after supplementation of the diet with inorganic selenium. Lipofuscin is a metabolic end product of lipid peroxidation (Csallany and Menken, 1986; Pehsron, 1993).
Selenomethionine is the major form of organic selenium in grains and vegetable protein sources. Selenium concentration in plants is dependent on selenium content in soils and on soil conditions. Some soils of North America are deficient in selenium, so plants grown in these areas are also low in selenium. When fed without supplementation, symptoms of selenium deficiency can be observed. It is generally accepted that dietary organic selenium is more effectively retained than inorganic selenium (Mutanen, 1986; Pehrson, 1993; Arai et al., 1994; Mahan, 1995). It is suggested that organic selenium (primarily in selenoamino acid form) is absorbed across the intestinal wall by active transport mechanisms rather than the passive diffusion process used for selenium ions derived from inorganic sources. As mentioned above, the ability of organic and inorganic selenium compounds to supply selenium for GSH-Px synthesis is about the same. However retention of selenium is significantly higher for organic compounds (Perhson, 1993; Mahan, 1995; Edens, 1996; Mahan and Kim, 1996). The magnitude of the difference has been found to be more pronounced with increasing levels of dietary selenium and in certain tissues, particularly brain and muscle (Whanger and Butler, 1988). This higher retention of organic selenium could be used for GSH-Px synthesis when animals are under stress conditions. It could also explain why animals raised under adverse conditions perform better when organic selenium is present in feed (Ku et al., 1973; Mahan, 1995; Edens, 1996).
A new source of selenium, selenium yeast (Sel-Plex, Alltech Inc., Nicholasville, Kentucky), has recently been commercialized. Organic selenium in Sel-Plex is more extensively assimilated and thus dietary levels needed to support animal performance are potentially decreased. Many studies have shown improved animal performance (eg. Table 2), tissue selenium levels (blood, milk, eggs, meat) and reduced selenium excretion in urine and feces in response to organic selenium (Marsh et al., 1981; 1986; Mahan, 1995; Edens, 1996; Mahan and Kim, 1996).
| Table 2. Effect of selenium source on mortality and carcass quality in broilers. |
 |
| Edens, 1996. |
Effects of vitamin E level and selenium level and form on incidence of ascites and blood chemistry of broilers
The hypothesis of the present study was that higher dietary vitamin E in synergy with selenium supplementation might protect membranes against oxidation caused by hypoxia and acidosis and help maintain performance of fast growing broilers while protecting against development of PHS. Consequently the objectives were to determine the effects of higher levels of vitamin E and two forms of selenium, organic and inorganic, on the incidence of PHS, serum GSH-Px, blood gases, electrolytes and growth performance in cold-stressed broiler chickens.
MATERIALS AND METHODS
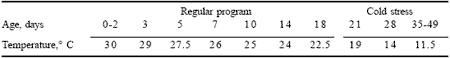
Broiler chickens (1,368) of the Ross 308 x Ross 308 strain were vaccinated at one day of age against Marek’s disease and infectious bronchitis and given ad libitum access to feed and water during a 49 day trial. Birds were wing-tagged at seven days of age and divided into six groups of four replicate pens each. Each pen contained 57 birds to achieve a final floor density of 0.085 m2 per bird. Birds received three types of pelleted feeds: a crumbled starter feed for the first 18 days, a crumbled grower feed for the next 17 days and a pelleted finisher feed during the last two weeks of the grow-out period (Table 3). To predispose birds to PHS, a cold stress temperature protocol modified from Julian et al. (1989) was employed (Table 4).
| Table 3. Formula and composition of basal diets. |
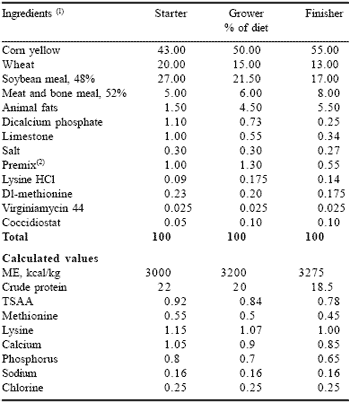 |
| 1 As-fed basis 2 Per kg of diet: vitamin A, 12,000 IU; cholecalciferol, 3,000 IU; menadione, 3 mg; thiamin HCl, 2 mg; riboflavin, 6 mg; pyridoxine HCl, 4 mg; vitamin B12 , 16 mg; niacin, 35 mg; Ca pantothenate, 12 mg; choline Cl, 400 mg; folic acid, 1 mg; D-biotin, 0.1 mg; Mn, 80 mg; Cu, 15 mg; I, 1 mg; Fe, 15 mg; Zn, 60 mg. |
| Table 4. Cold stress temperature protocol. |
 |
Vitamin E levels used in the trial were the recommended commercial level (50 IU/kg) and five times that recommendation (250 IU/kg). The experimental design was a 2x3x4 factorial with starter feed containing either 50 or 250 IU vitamin E per kg fed with either 0.3 ppm Se from organic selenium yeast (Sel-Plex, Alltech Inc), or 0.3 ppm or 0.6 ppm Se from sodium selenite. Vitamin E levels were decreased to 30 and 150 IU/kg, and 20 and 100 IU/kg for the grower and finisher diets, respectively (Table 5).
| Table 5. Antioxidant-supplemented dietary treatments. |
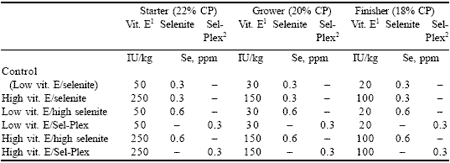 |
| 1 Vitamin E From BASF , Georgetown Ontario Canada Organic selenium yeast from Sel-Plex, Alltech Inc., Nicholasville, Kentucky |
All birds were individually weighed at 7, 18, 35, 40, 47 days of age. Feed consumption and feed conversion were evaluated for each pen and each group at weighing time. Every broiler dying during the study was necropsied to determine the cause of death. Diagnosis of PHS was confirmed when there was presence of fluid in the abdominal cavity, right ventricular dilation, hydropericardium and generalized vascular congestion. At 47 days, venous blood samples were drawn from one third of the birds (16 broilers per pen).
Bicarbonate, pH and hemoglobin were measured with a blood gas apparatus (ABL-300). Hematocrit was determined by centrifugation. Plasma electrolytes (Na, K, Cl) and GSH-Px were measured from frozen samples by HPLC. Birds were slaughtered at 49 days of age. Hearts of bloodsampled birds were collected at the slaughterhouse and dissected to measure the right ventricle/total ventricle (RV/TV) ratio (Julian, 1986; Julian et al., 1989).
Statistical analysis was carried out using General Linear models procedures of SAS® software (SAS Institute, 1966). Growth performance, consumption and feed conversion data, were subjected to repeated measures ANOVA with treatment as fixed effects factors and time as repeated factors. Logarithmic transformation was used to normalize the GSH-Px data. First-order auto-regressive structure to model covariance was performed to provide the best description of covariance structure (Littel et al., 1998). At each time period GLM was used with Tukey’s post-hoc test to determine which treatments were statistically different (P<0.05). Chi-square tests to establish mortality rate were performed to examine association with treatment.
Sequential contrasts were then applied to determine which treatments were different (P<0.05). For blood parameters and RV TV heart ratio analysis, a GLM with Tukey’s post-hoc test was used to determine statistical differences between treatments (P<0.05). Statistical analysis was carried out using General Linear models procedures of SAS (1996).
RESULTS
Ascites mortality
Cumulative mortality rate of the control group was 16.3%, while mortality caused by ascites was 10% (Figure 4). Mortality attributed to ascites decreased in birds given 250 IU vitamin E (4.98%), 0.6 ppm Se from selenite (3.76%), or high vitamin E plus 0.06 ppm Se from selenite (3.24%). The greatest reduction in ascites mortality was observed in birds given 0.3 ppm Se from Sel-Plex with either low vitamin E (2.82%) or high vitamin E (0.90%) (P<0.05).
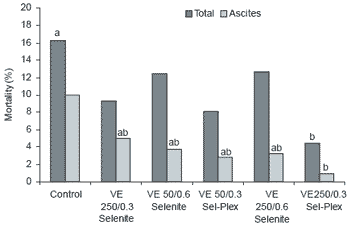 |
| Figure 4. Effect of antioxidant supplementation on mortality in broilers (abMeans differ, p<0.05). |
Bottje et al. (1995b) showed decreased ascites mortality rate in chickens implanted with subcutaneous vitamin E pellets. Boren and Pond (1996) also observed a reduction in total mortality and condemnations due to ascites in commercial flocks supplemented with 260 IU/kg of feed during the first two weeks of age. Similarly, Stanley et al. (1998) showed that chicks exposed to cold stress and aflatoxin-contaminated feed had lower ascites mortality when treated with high levels of vitamin E (500 IU/kg) and Sel- Plex organic selenium (0.1 ppm). However, when Bottje et al. (1997) tested vitamin E feed supplementation (87 IU/kg), no effects on performance and mortality were observed. Results of the present study and those of Stanley et al. (1998) suggest that in order to prevent mortality caused by ascites, higher levels of vitamin E and selenium are required. Furthermore, it appears that organic selenium was more effective than the inorganic source.
Ventricular weight ratio
Addition of any antioxidant in feed improved the RV/TV ratio (P<0.05), decreasing it from 0.30 in the controls to below 0.24 in all other treatment groups (Table 6). Similarly to ascites mortality rate, the smallest ratio was obtained with the combination of high dietary vitamin E and organic selenium (0.215).
| Table 6. Ratio between right ventricular and total ventricular volume.1 |
 |
| 1Values are mean ± standard deviation. abMeans with no common superscripts differ (P<0.05). |
Ventricular weight ratios (RV/TV) above 0.25 have been associated with the clinical appearance of PHS. A positive linear relationship between PHS incidence and RV/TV ratio has previously been demonstrated (Maxwell et al., 1986a; Julian, 1986; Huchzermeyer et al., 1988; Chabot, 1992). The results of the present study are in concordance with these findings.
Blood chemistry responses, GSH-Px
Analysis of blood samples revealed that GSH-Px, hemoglobin (Hb), pH and bicarbonate (HCO3) values were significantly different in the antioxidanttreated groups when compared to controls (Table 7). There were no significant differences in hematocrit values, sodium or potassium levels due to treatment. Serum GSH-Px increased with addition of organic selenium alone (4796 U/l) or when combined with the high vitamin E level (5489 U/l) (Figure 5). Hemoglobin was significantly higher in birds given 250 ppm vitamin E with either 0.6 ppm Se from selenite (10.11 g/dl) or 0.3 ppm Se from Sel-Plex (10.44 g/dl) (Figure 6).
Blood pH in the control group revealed metabolic acidosis with a value of 7.29. All antioxidant treatments prevented acidosis and kept blood pH in the normal range (7.34 to 7.39) (Table 7). Since pH and bicarbonate are positively correlated; a similar effect on blood bicarbonate values was observed with the control group having significantly lower values than those of antioxidant groups.
In the present study, GSH-Px activities in the high vitamin E/high selenite, 0.3 ppm Sel-Plex Se and high vitamin E plus Sel-Plex treatments were 136, 155 and 178% higher, respectively, when compared to controls (P<0.05). A positive correlation between selenium addition to feed and GSH-Px activity has been noted in many species when dietary selenium content was less than 0.3 ppm (Chavez, 1979; Arai et al., 1994; Mahan, 1995; Mahan and Kim, 1996). The difference in GSH-Px activity noted between the two types of selenium in this study is proably due to the nature of the selenium in each form. Since higher GSH-Px levels are desirable in order to better protect various tissues against the deleterious effects of oxidative stress (Bottje et al., 1995b), it is not surprising that lower ascites mortality rates were observed with the more bioavailable organic selenium alone or in combination with higher dietary vitamin E.
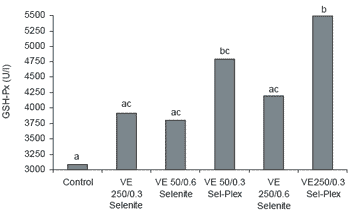 |
| Figure 5. Effect of antioxidant supplementation on plasma GSH-Px in broilers (abcMeans differ, P<0.05). |
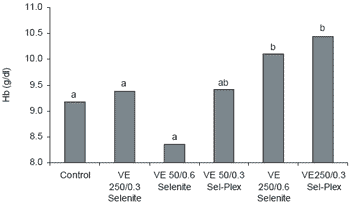 |
| Figure 6. Effect of antioxidant supplementation on hemoglobin levels in broilers (abMeans differ, P<0.05). |
| Table 7. Effect of antioxidant supplementation on blood parameters1. |
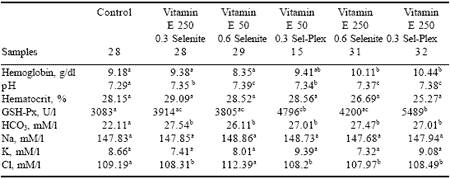 |
| 1 Data are uncorrected for mortality. abcd Means in rows with no common superscript differ (P<0.05). |
The increase in hemoglobin concentrations observed in the high vitamin E/high selenite or Sel-Plex groups likely reflected improved integrity of the erythrocyte cellular membranes and thus potentially better tissue oxygenation. The present results, along with higher hemoglobin concentrations, pH and bicarbonate levels, are compatible with normal acid-base status also reported elsewhere (Chabot, 1992). Body weight and feed efficiency Addition of antioxidants did not affect final body weight. However, feed conversion was significantly improved in birds given 250 IU vitamin E plus 0.3 ppm Se from Sel-Plex diet (Table 8).
Reduction of ascites mortality during the last two weeks of the grow-out period, better feed conversion, improved GSH-Px and blood gas values showed that birds with normal acid-base status both perform better and are better protected against the deleterious effects of oxidative stress. Boren and Bond (1996) were the first to demonstrate the potential of vitamin E to improve broiler performance using 260 IU vitamin E per kg in a starter diet for commercially-reared broilers. Total mortality and condemnation rates were lower in the supplemented group. High levels of vitamin E also proved effective in improving performance under stress conditions. Stanley et al. (1998) submitted broiler chickens to cold stress and fed aflatoxin-contaminated feed supplemented with 500 IU/kg vitamin E and 0.1 ppm Se from Sel-Plex. Incidence of PHS, mortality and hematocrit values were reduced and better hemoglobin values and tissue protection demonstrated. In the present study 250 IU/kg vitamin E and 0.3 ppm Se from Sel-Plex in cold stressed broilers gave similar improvements in performance. Together these three studies demonstrate that higher levels of vitamin E and Sel-Plex could help prevent PHS and maintain performance of broiler chickens.
| Table 8. Influence of antioxidants on body weights and feed conversion of broilers.1 |
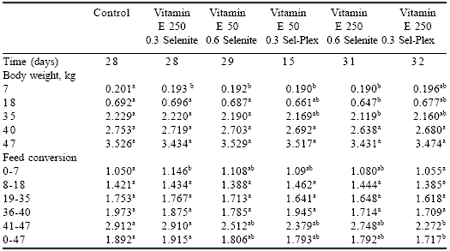 |
| 1 Data are uncorrected for mortality. ab Means in rows with no common superscript differ (P<0.05). |
| Conclusions |
| Results of this study provide evidence that increased vitamin E in the diet and organic selenium (Sel-Plex) reduce mortality associated with PHS in cold stressed broiler chickens. Low mortality and better feed conversion appear to be the result of improved antioxidant status in birds. Protective effects of antioxidants on the erythrocyte cellular membrane were demonstrated by higher hemoglobin concentration, increased GSH-Px activity, improved blood gas parameters, HCO3, pH and normal acid-base status. |
Acknowledgments
We would like to thank Ms. Avila Croisetière and Dr Guy Beauchamps for their technical support in this project. This study was made possible by the financial support of Probiotech Inc., Alltech, Inc., BASF Canada and CORPAQ.
References
Arai, T., M. Sugawara, T. Sako, S. Motoyoshi, T. Shimura, N. Tsutsui and T. Konno. 1994. Glutathione peroxidase activity in tissues of chickens supplemented with dietary selenium. Comp. Biochem. Physiol. 107A (1):245-248.
Boren B. and P. Bond. 1996. Vitamin E and immunocompetence. Broiler Industry, November 1996. pp. 26-33.
Bottje, W., B. Enkvetchakul and R. Wideman. 1995a. Antioxidants, hypoxia and lipid peroxidation involvement in pulmonary hypertension syndrome (Ascite). In: Novus, Nutrition Update 5(2). pp. 1-11.
Bottje, W., B. Enkvetchakul, R. Moore and R. McNew. 1995b. Effect of "-tocopherols on antioxidants, lipid peroxidation and the incidence of pulmonary hypertension syndrome (ascites) in broilers. Poultry Sci. 74:1356- 1369.
Bottje, W., G.F. Erf, T.K. Bersi, D. Wang, D. Barnes and K.W. Beers. 1997. Effect of dietary dl-"-tocopherols on tissue "-and (-tocopherol and pulmonary hypertension syndrome (ascites) in broilers. Poultry Sci. 76:1506-1512.
Boulianne, M. 1993. Cas d’aspergillose précoce suivi d’une forte incidence d’ascite. Le Médecin Vétérinaire du Québec, 23(1):28-31.
Chabot, R. 1992. Le syndrome de l’ascite chez les poulets à chair. Mémoire de Maîtrise, Faculté de Médecine vétérinaire de l’Université de Montréal à St-Hyacinthe. pp. 50.
Chavez, E.R. 1979. Effect of dietary selenium depletion and repletion on plasma glutathione peroxidase activity and selenium concentration in blood and body tissues of growing pigs. Can. J. Anim. Sci. 59:761-771.
Coelho, M.B. and J.L. McNaughton. 1995. Effect of composite vitamin supplementation on broilers. J. Appl. Poultry Res. 4:219.
Csallany, A.S. and B.Z. Menken. 1986. Effect of dietary selenite on hepatic organic solvent-soluble lipofuscin pigments. J. Am. Coll. Toxic 5:79.
Edens, F.W. 1996. Organic selenium: From feathers to muscle integrity to drip loss. Five years onward: No more selenite. In: Biotechnology in the Feed Industry, Proceedings of the 12th Annual Symposium (T.P. Lyons and K.A. Jacques, eds), Nottingham University Press, UK. pp. 165-185.
Enkvetchakul, B., W. Bottje, R. Moore and W. Huff. 1993. Compromised antioxidant status associated with ascites in broilers. Poultry Sci. 72:2272- 2280.
Halliwel, B. and J.M.C. Gutteridge. 1990. Role of free radicals and catalytic metal ion in human disease: An overview. Methods Enzymol. 186:1- 85.
Huchzermeyer, F.W., R.J. Julian and J.K. Barker. 1988. Lesions of right heart failure and ascites in broiler chickens. Avian Dis.32:246-261.
Julian, R.J. 1986. Right ventricular failure as a cause of ascites in broiler and roaster chickens. In: Proceedings IVth International Symposium Veterinary Laboratory Diagnosticians, Amsterdam. pp. 608-611.
Julian, R.J. 1989. Lung volume in meat-type chickens. Avian Dis. 33:174- 176.
Julian, R.J. 1993. Review article; Ascites in poultry. Avian Pathol. 22:419- 454.
Julian, R.J., M. Boulianne and J.P. Vaillancourt. 1995. Prévention de la défaillance ventriculaire droite secondaire à l’hypertension pulmonaire et de l’ascite chez le poulet à griller. Le Médecin Vétérinaire du Québec 25(2):73-76.
Julian, R.J., I. McMillan and M. Quinton. 1989. The effect of cold and dietary energy on right ventricular hypertrophy, right ventricular failure and ascites in meat-type chickens. Avian Pathol. 18:730-732.
Ku, P.K., E.R. Miller, R.C. Wallstram, A.W. Grace, J.P. Hichcock and D.E. Ullrey. 1973. Selenium supplementation of naturally high selenium diets for swine. J. Anim. Sci. 37:501.
Machlin, L. 1991. Vitamin E. In: Handbook of vitamins (L.J. Machlin, ed). Marcel Dekker, Inc. New York, NY. pp. 100-144.
Maddaiah, V.T. 1990. Glutathione correlates with lipid peroxidation in liver mitochondria of triiodothyronine injected hypophysectomized rats. FASEB J. 4:1513-1518
Mahan D.C. 1995. Selenium metabolism in animals: What role does selenium yeast have? In: Biotechnology in the Feed Industry, Proceedings of the 11th Annual Symposium (T.P. Lyons and K.A. Jacques, eds), Nottingham University Press, UK. pp. 257-266.
Mahan, D.C. and Y.Y. Kim. 1996. Effect on inorganic or organic selenium at two levels on reproductive performance and tissue selenium concentrations in first parity gilts and their progeny. J. Anim. Sci. 74:2711-2718.
Marsh, J.A., R.R. Dietert and G.F. Combs Jr. 1981. Influence of dietary selenium and vitamin E on the humoral immune response of chick. Proc. Soc. Exp. Biol. Med. 166:228.
Marsh, J.A., G.F. Combs, Jr., M.E. Whitacre and R.R. Dietert. 1986. Effect of selenium and vitamin E dietary deficiencies on chick lymphoid organ development. Proc. Soc. Exp. Biol. Med. 182:425.
Maxwell, M.H., G.W. Robertson and S. Spence. 1986a. Studies on an ascitic syndrome in young broilers. 1. Haematology and pathology. Avian Pathol. 15:511-524.
Maxwell, M.H., G.W. Robertson and S. Spence. 1986b. Studies on an ascitic syndrome in young broilers. 2. Ultrastructure. Avian Pathol. 15:525- 538.
Maxwell, M.H. and G.W. Robertson. 1997. World broiler ascites survey 1996. Poultry International, April. pp. 16-30.
Maxwell, M.H. and G.W. Robertson. 1998. UK survey of broiler ascites and sudden death syndromes in 1993. Br. Poult. Sci. 39:203-215.
McCord, J.M. 1985. Oxygen-derived free radicals in post-ischemic tissue injury. N. Engl. J. Med. 312:159-163.
Meyer, W.R., D.C. Mahan and A.L. Moxon. 1981. Value of dietary selenium and vitamin E for weanling swine as measured by performance and tissue selenium and glutathione peroxidase activities. J. Anim. Sci. 52:302- 311.
Mutanen, M. 1986. Bioavailability of selenium. Ann. Clin. Res. 18:48.
National Research Council. 1983. Subcommitee on selenium. Selenium in Nutrition. Revised Ed. Nutrient of Poultry 8th rev. Ed. National Academy Press Washington, DC.
Odom, T.W. 1993. Ascites syndrome: Overview and update. Poultry Sci. 50:14-22.
Olkowski, A.A., L. Kumor and L. Classen. 1996. Changing epidemiology of ascites in broiler chickens. Can. J. Anim. Sci. 76:135-140.
Peacock, A.J., C.K. Pickett, K.G. Morris and J.T. Reeves. 1990. Spontaneous hypoxaemia and right ventricular hypertrophy in fast growing broiler chickens reared at sea level. Comp. Biochem. Physiol. 97A:537-541.
Pehrson, B.G. 1993. Selenium in nutrition with special reference to the biopotency of organic and inorganic selenium compounds. In: Biotechnology in the Feed Industry, Proceedings of the 9th Annual Symposium (T.P. Lyons and K.A. Jacques, eds), Alltech Technical Publications, Nicholasville, Ky. pp. 71-89.
Reeves, J.T., G. Ballam, S. Hofmeister, C. Pickett, K. Morris and A. Peacock. 1991. Improved arterial oxygenation with feed restriction in rapidly growing broiler chickens. Comp. Biochem. Physiol. 99A:481-495.
SAS Institute. 1996. SAS/STAT User’s Guide, Version 6, 4th ed., SAS Institute Inc., Cary, NC Stanley G.V., H. Chukwu and D. Thompson. 1998. Singly and combined effects of organic selenium (Se-Yeast) and vitamin E on ascites reduction in broilers. PSSA, 1998 abst: 111.
Vidyadaran, M.K., A.S. King and H. Kassin. 1990. Quantitative comparisons of lung structure of adult domestic fowl and red jungle fowl, with to broiler ascites. Avian Pathol. 19:51-58.
Whanger, P.D. and J.A. Butler. 1988. Effects of various dietary levels of selenium as selenite or selenomethionine on tissue selenium levels and glutathione peroxidase activity in rats. J. Nutr. 118:846.
Yu, B.P. 1994. Cellular defenses against damage from reactive oxygen species. Physiol. Rev. 74:139-162.
Authors: GHISLAINE ROCH, MARTINE BOULIANNE and LASZLO DE ROTH
University of Montreal, St. Hyacinthe, Quebec, Canada









.jpg&w=3840&q=75)