Introduction
Necrotic ear syndrome or ear necrosis in pigs has been reported as an increasing health problem in many countries with intensive pig farming.1 It is characterized by large erosive lesions at the margin of the pinna(e) in both sexes. It occurs mainly in weaning pigs and growers/ fatteners with bodyweight ranging approximately from 10 to 40 kg.2 The earliest lesions are normally visible on the ear' tips at 6-7 weeks old pigs, beginning as a superficial vesicular dermatitis associated with superficial auricular trauma, which can bleed, attractive to pen mates, who may then start to bite at the lesion, resulting in swelling and reddening of the ear. Localized lesions slowly healed or sporadically progressed to deep necrotic ulcers, cellulitis, vasculitis, thrombosis, ischemia.3
The etiology of this disease is complex and therefore it is often named as porcine ear necrosis syndrome (PENS). The presumed triggering factors can be divided into infectious and not infectious agents. It is usually the result of a mixed infections causing damage to the skin. Staphylococcus hyicus is the most common isolated agent in lesions of PENS cases, but other pathogens such as Mycoplasma suis, Streptococcus suis and spirochetes are often implicated.4-8 Moreover, non-infectious factors such as intensive pen density and overpopulation, poor air quality with high concentrations of gases (e.g. ammonia), poor hygienic conditions, copper and magnesium deficiency, contamination of feed with mycotoxins and cannibalism, were associated with an increased risk of incidence of PENS.2,9-12
Recently, an important causative role has also been attributed to immunosuppressive agents such as Porcine Circovirus type 2 (PCV2), Porcine Reproductive and Respiratory Syndrome Virus (PRRSV), as well as mycotoxins.7,13 PCV2 is considered to be involved in etiology agents of the development of PNES14 and is present in ear lesions PCV2 infected pigs,15 associated sometimes with co-infections of PRRSV,16,17 Pasteurella multocida, Streptococcus suis types 1 and 2 and other pathogens.18,19
Last years a marked increase of field cases characterized by PNES associated with PCV2 infection has been observed in the USA,20 in Canada21 and Europe.1,9,14,22 In Greece, during the last two years our clinical observations in field conditions suggest the cases of PNES have been increased and when Porcine Circovirus Associated Diseases (PCVAD) is present on a farm, more pigs with ear tip necrosis are observed, accompanied with severe wasting, respiratory clinical signs and significant mortality.23
Case Description
History. The present report describes a case of PCV2 infection associated with lesions of PNES at the weaning stage of a farrow-to-finish pig farm of 200 sows (Large x White x Landrace) in Central Greece. Weaning took place at the age of 25 ± 3 days. All weaning pigs were moved every week into the flat deck unit, grouped in pens of 15 pigs. This farm applied all appropriate facilities of biosecurity of good hygiene.
Clinical observations. Approximately 35% of weaning pigs (1-3 weeks after weaning), through out of different batches, presented clinical symptoms similar to Post-weaning Multisystemic Wasting Syndrome (PMWS), such as fever, anorexia, diarrhea, considerable weight loss cough, dyspnea, paleness of the skin, enlarged lymph nodes and lethargy. About 2-3 weeks after weaning the first lesions of PNES occurred in approximately 20% of pigs, resulting in a significant health problem characterized by poor growth or severe wasting and finally mortality up to 15% in some batches. Moreover, approximately 5% of survived weaning pigs, during growing / finishing stage, presented poor growth and secondary co-infections that lead to death.
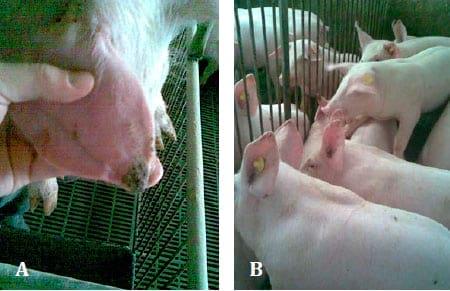
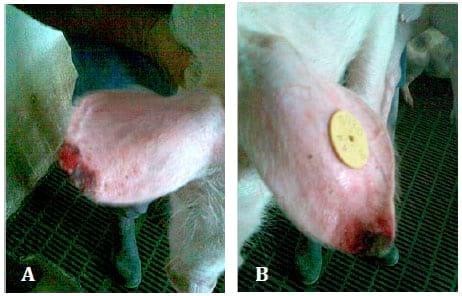
Regarding the lesions of PNES, they initially appeared at the margin of the pinna(e) (on the tip of the ear or at its base) as a superficial vesicular dermatitis associated with superficial auricular trauma (Fig. 1. A and B). These lesions were characterized by necrosis, vasculitis, dry gangrene and inflammation that sporadically progress in exudative o r ulcers (Fig. 2. A and B). In addition, the necrotic lesions were complicated with secondary infections.
Fig. 1. A. Weaning pig with initial lesions of PNES. B. Group of pigs with PNES clinical signs.
Sampling / Diagnosis. Blood samples were collected from pigs at age of 5-6 weeks (group 1), 7-8 weeks (group 2), 9-10 weeks (group 3) and 22-23 weeks (group 4). Serum samples were stored at -20 °C, and analyzed for PCV2 IgM and IgG antibodies by Ig capture ELISA based on method described by van Esch and Wellenberg.24 IgM and IgG specific ELISA tests (Ingezim PCV IgG / IgM®, Spain) were also used. Moreover, five weaning pigs (4-9 weeks of age) were euthanized for necropsy. Samples collected from lung, heart, inguinal lymph node, tonsil, thymus, spleen, small and large intestines, liver, kidney, and pancreas were fixed in 10% (w/v) buffered formaldehyde for 24 h and embedded in paraffin by standard histologic procedures.
Fig. 2 A and B. Weaning pigs with severe lesions of ear necrosis syndrome.
Results
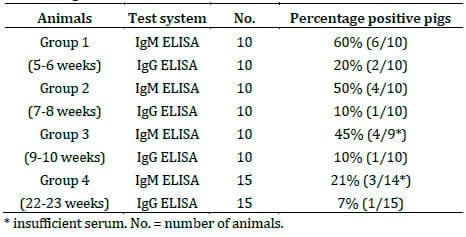
The serological results for PCV2 IgM and IgG responses in sera blood samples are shown in Table 1. Based on the serological results and the proposed interpretation of PCV2 IgM and IgG ELISA results in pigs,24 it is noticed that infected pigs suffered by sub-acute infection.
Table 1. Percentage of PCV2- positive pigs (PCV2 IgM, IgG) in different ages.
Pathological examinations based on microscopic analysis of lymphoid tissue immunohistochemistry and in situ hybridization, indicated abundant amount of PCV2 DNA within observed histopathological PMWS-specific lesions (lymphocyte depletion in lymphoid tissues and interstitial pneumonia).
Treatment protocol / Control strategies. In this case of PCV2-infection associated with PNWS, intramuscular injection of florfenicol (20 mg kg-1 body weight) was suggested for the treatment and control of skin lesions and respiratory signs. Moreover, the following batches of piglets were vaccinated against PCV2 with commercial vaccine Porcilis® PCV (MSD Animal Health Animal Health) administered with 2 × 2 mL dose scheme (first dose at 7th day of age, with the second dose 3 weeks later -day of weaning). This vaccination scheme resulted in a significant reduction of PNES prevalence and mortality rate of weaning pigs in the herd (data not shown). At the same time, for these following batches of piglets a grouping of 10 animals per pen at weaning stage was applied.
Discussion
Based on the clinical signs of infected weaning pigs and serological as well as pathological results, we concluded that weaning pigs suffered by sub-acute PCV2 infection resulting in PMWS associated with PNES. The lesions of PNES were initially observed at the same period (4-8 weeks of age) the higher seroprevalence of PCV2 infection, as is shown in Table 1.
The results of the present study indicate that PNES co-existing with PCV2 infection in many cases. The prevention and control of PCVAD are based on proper immunization (vaccination) and management practices.19 Nowadays, at least four commercial vaccines are available against PCVAD in piglets and sows. The vaccines have succeeded in reducing losses caused by PCV2 in Europe, Canada and the USA.25 The findings of our study agree with the results of previous studies,1,14,22 with the difference that we managed to reduce the losses due to PCV2 infection associated with PNES, using an inactivated vaccine in 2 doses scheme (early at 7th day of age and at day of weaning).
In conclusion, sub-acute PCV2 infection could be included in triggering factors PNES in weaners. The mass vaccination against PCV2 of infected piglets might be effective in reduction of clinical signs and losses of PNES in cases of PCV2 infection associated with PNES.
References
1. Petersen HH, Nielsen EO, Hassing AG, et al. Prevalence of clinical signs of disease in Danish finisher pigs. Vet Rec 2008; 162(12): 377-382.
2. Cameron RDA. Diseases of the Ski. In: Straw BE, Zimmerman JJ, D'Allaire SD, et al., Eds. Diseases of Swine, 9th ed. Iowa, Ames: Iowa State Press 2006: 179-198.
3. Richardson A, Morter RL, Rebar AH, et al. Lesions of porcine necrotic ear syndrome. Vet Pathol 1984; 21 (2): 152-157.
4. Fraser CM, Bergeron JA, Mays A, et al. The Merck Veterinary Manual, 7th ed. Rahway, New Jersey: Merck & Co. 1991: 308.
5. Mirt D. Lesions of so-called flank biting and necrotic ear syndrome in pigs. Vet Rec 1999; 144: 92-96.
6. Tanabe T, Hisaaki S, Hideki S, et al. Correlation between occurrence of exudative epidermitis and exfoliative toxin-producing ability of Staphyulococcus hyicus. Vet Microbiol 1996; 48: 9-17.
7. Molnár T, Glávits R, Szeredi L, et al. Occurrence of porcine dermatitis and nephropathy syndrome in Hungary. Acta Vet Hung 2002; 50: 5-16.
8. Thacker EL. Mycoplasmal Diseases. In: Straw BE, Zimmerman JJ, D'Allaire SD, et al., Eds. Diseases of Swine, 9th ed. Iowa, Ames: Iowa State Press 2006: 701-717.
9. Busch ME, Wachmann H, Nielsen EO, et al. Risk factors for each necrosis and tail lesions in weaners. In Proceedings of 20th International Pig Veterinary Society Congress, Durban, South Africa 2008: OR.12.12.
10. Osweiler, GD. Occurence of Mycotoxins in Grains and Feed. In: Straw BE, Zimmerman JJ, D'Allaire SD, et al. Eds. Diseases of Swine, 9th ed. Iowa, Ames: Iowa State Press 2006: 915-929.
11. Schroder-Petersen DL, Simonsen HB. Tail biting in pigs. Vet J 2001: 196-210.
12. Truszczynski M, Pejsak Z. Mycoplasma suis and porcine eperythrozoonosis, including achievements of the last years, Medycyna Wet 2009; 65: 223-227.
13. Allan GM, Ellis JA. Porcine circovirus: a review. J Vet Diagn Invest 2000; 12(1): 3-14.
14.Madec F, Rose N. Post-weaning multisystemic wasting syndrome (PMWS), in pigs: An up-date with still questions. In Proceedings of International Society for Animal Hygiene, Warsaw Poland, 2005: 367.
15. Zlotowsk, P, Correa AMR, Barcellos DESN, et al. Presence of PCV2 in ear lesions in the course of PCVAD in growing pigs. In Proceedings of 20th International Pig Veterinary Society Congress, Durban, South Africa 2008; 555.
16. Choi C, Chae C. Colocalization of porcine reproductive and respiratory syndrome virus and porcine circovirus 2 in porcine dermatitis and nephropathy syndrome by double- labeling technique. Vet Pathol 2001; 38: 436-441.
17. Thibault S, Drolet R, Germain MC, et al. Cutaneous and systemic necrotizing vasculitis in swine. Vet Pathol 1998; 35: 108-116.
18. Lainson FA, Aitchison KD, Donachie W, et al. Typing of Pasteurella multocida isolated from pigs with and without porcine dermatitis and nephropathy syndrome. J Clin Microbiol 2002; 40: 588-593.
19. Thomson JR, Higgins RJ, Smith WJ, et al. Porcine dermatitis and nephropathy syndrome: clinical and pathological features of cases in the United Kingdom (1993-1998). J Vet Med A Physiol Pathol Clin Med, 2002; 49: 430-437.
20. Henry SC, Tokach LM. Porcine circovirus-associated disease in Kansas. In Proceedings of Allen D. Leman Swine Conference, St. Paul – Minnesota – USA 2006: 110-111.
21. Desrosiers R. Overview of PCVD - The Disease in Eastern Canada & US vs. Europe. Advances in Pork Production 2007; 18: 35.
22. Pejsak Z, Markowska-Daniela I, Pomorska-Móla M, et al. Ear necrosis reduction in pigs after vaccination against PCV2. Res Vet Sci 2011; 91(1): 125-128.
23. Papatsiros VG. Exploration of the connection between porcine necrotic ear syndrome and PCV2 infection. J Anim Vet Adv 2011; 10(2): 185-187.
24. Van Esch E, Wellenberg GJ. IgM and IgG antibody responses in sera from pigs experimentally infected with PCV2. In Proceedings of 5th International Symposium on Emerging and Re-emerging Pig Diseases, 2007: 68.
25. Opriessnig T, Meng XJ, Halbur PG. Porcine circovirus type 2 associated disease -Update on current terminology, clinical manifestations, pathogenesis, diagnosis, and intervention strategies. Vet Diagn Invest 2007; 19: 591-615.