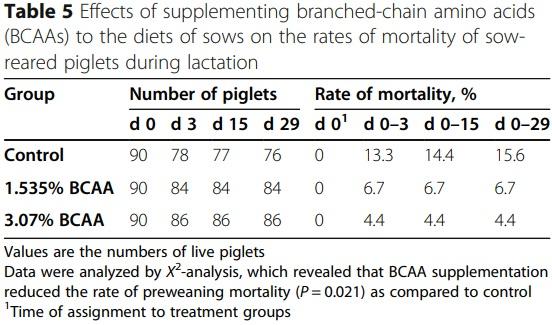
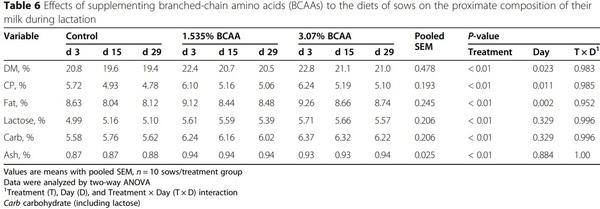
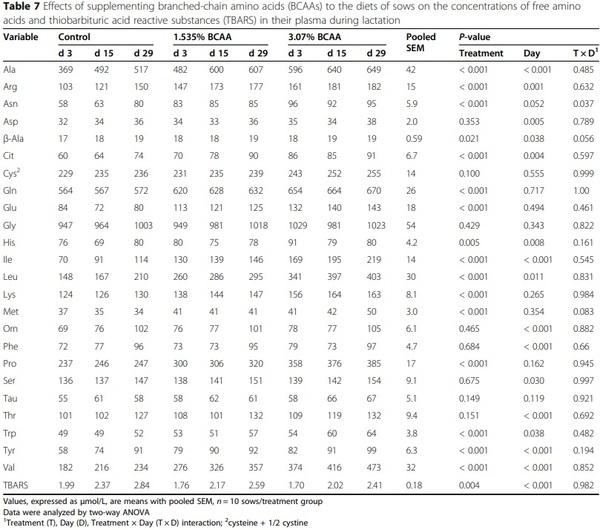
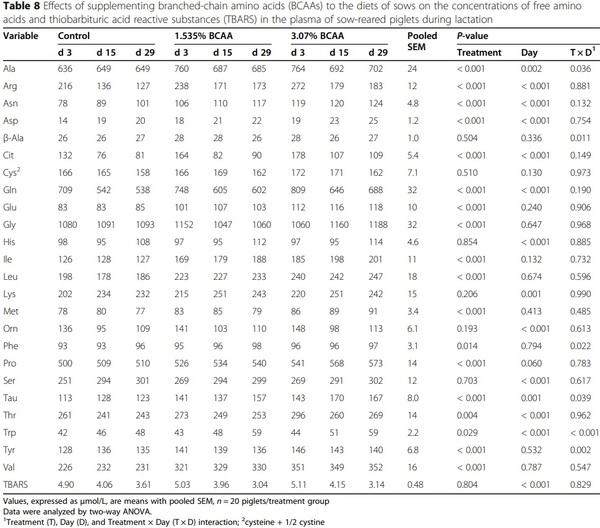
Dietary supplementation with branched-chain amino acids enhances milk production by lactating sows and the growth of suckling piglets
1. Boyd DR, Kensinger RS, Harrell RJ, Bauman DE. Nutrient uptake and endocrine regulation of milk synthesis by mammary tissue of lactating sows. J Anim Sci. 1995;73(suppl_2):36–56. https://doi.org/10.2527/1995.73 suppl_236x.
2. Quiniou N, Dagorn J, Gaudre D. Variation of piglets’ birth weight and consequences on subsequent performance. Livest Prod Sci. 2002;78(1):63–
70. https://doi.org/10.1016/S0301-6226(02)00181-1.
3. Fix JS, Cassady JP, Holl JW, Herring WO, Culbertson MS, See MT. Effect of piglet birth weight on survival and quality of commercial market swine.
Livest Sci. 2010;123(1-3):98–106. https://doi.org/10.1016/j.livsci.2010.05.007.
4. Ward SA, Kirkwood RN, Plush KJ. Are larger litters a concern for piglet survival or an effectively manageable trait? Animals. 2020;10(2):309. https:// doi.org/10.3390/ani10020309.
5. Rezaei R, Knabe DA, Li XL, Feng S, Wu G. Enhanced efficiency of milk utilization for growth in surviving low-birth-weight piglets. J Anim Sci
Biotech. 2011;2:73–83.
6. Ji Y, Wu ZL, Dai ZL, Wang XL, Li J, Wang BG, et al. Fetal and neonatal programming of postnatal growth and feed efficiency in swine. J Anim Sci
Biotechnol. 2016;8(1):42. https://doi.org/10.1186/s40104-017-0173-5.
7. Wu G, Bazer FW, Wallace JM, Spencer TE. Intrauterine growth retardation: implications for the animal sciences. J Anim Sci. 2006;84(9):2316–37. https:// doi.org/10.2527/jas.2006-156.
8. USDA Animal and Plant Health Inspection Service. Preweaning morbidity and mortality. National Swine Survey. U.S. Fort Collins, Colorado:
Department of Agriculture; 1992.
9. USA 2020 Summary, PigCHAMP Benchmarking. https://www.pigchamp. com/Portals/0/Documents/Benchmarking%20Summaries/2020-benchmarksummaries-usa.pdf. .
10. Wolter BF, Ellis M, Corrigan BP, Dedecker JM. The effect of birth weight and feeding supplemental milk replacer to piglets during lactation on preweaning and postweaning growth performance and carcass characteristics. J Anim Sci. 2002;80(2):301–8. https://doi.org/10.2527/2002.
802301x.
11. Manjarin R, Bequette BJ, Wu G, Trottier NL. Linking our understanding of mammary gland metabolism to amino acid nutrition. Amino Acids. 2014;
46(11):2447–62. https://doi.org/10.1007/s00726-014-1818-8.
12. Rezaei R, Wu ZL, Hou YQ, Bazer FW, Wu G. Amino acids and mammary gland development: nutritional implications for neonatal growth. J Anim Sci
Biotechnol. 2016;7(1):20. https://doi.org/10.1186/s40104-016-0078-8.
13. Dunshea FR, Bauman DE, Nugent EA, Kerton DJ, King RH, McCauley I.
Hyperinsulinaemia, supplemental protein and branched-chain amino acids when combined can increase milk protein yield in lactating sows. Br J Nutr.
2005;93(3):325–32. https://doi.org/10.1079/BJN20041366.
14. Richert BT, Tokach MD, Goodband RD, Nelssen JL, Pettigrew JE, Walker RD, et al. Valine requirement of the high-producing lactating sow. J Anim Sci.
1996;74(6):1307–13. https://doi.org/10.2527/1996.7461307x.
15. Richert BT, Goodband RD, Tokach MD, Nelssen JL. Increasing valine, isoluecine, and total brancded-chain amino acids for lactating sows. J Anim
Sci. 1997;75(8):2117–28. https://doi.org/10.2527/1997.7582117x.
16. Moser SA, Tokach MD, Dritz SS, Goodband RD, Nelssen JL, Loughmiller JA.
The effects of branched-chain amino acids on sow and litter performance. J
Anim Sci. 2000;78(3):658–67. https://doi.org/10.2527/2000.783658x.
17. Paulicks BR, Ott H, Roth-Maier DA. Performance of lactating sows in response to the dietary valine supply. J Anim Physiol Anim Nutr. 2003;87(11-
12):389–96. https://doi.org/10.1046/j.1439-0396.2003.00449.x.
18. Richert BT, Tokach MD, Goodband RD, Nelssen JL, Campbell RG, Kershaw S.
The effect of dietary lysine and valine fed during lactation on sow and litter performance. J Anim Sci. 1997;75(7):1853–60. https://doi.org/10.2527/1997.
7571853x.
19. Davis TA, Nguyen H, Garcia-Bravo R, Fiorotto M, Jackson E, Lewis D, et al.
Amino acid composition in human milk. J Nutr. 1994;124(7):1126–32. https://doi.org/10.1093/jn/124.7.1126.
20. Wu G, Knabe DA. Free and protein-bound amino acids in sow's colostrum and milk. J Nutr. 1994;124(3):415–24. https://doi.org/10.1093/jn/124.3.415.
21. Wu G. Principles of Animal nutrition. Boca Raton: CRC Press; 2018.
22. Li P, Knabe DA, Kim SW, Lynch C, Hutson S, Wu G. Lactating porcine mammary tissue catabolized branched-chain amino acids for glutamine and asparate synthesis. J Nutr. 2009;139(8):1502–9. https://doi.org/10.3945/jn.1
09.105957.
23. Wu G, Bazer FW, Dai ZL, Li DF, Wang JJ, Wu ZL. Amino acid nutrition in animals: protein synthesis and beyond. Annu Rev Anim Biosci.
2014;2(1):387–417. https://doi.org/10.1146/annurev-animal-022
513-114113.
24. Strathe AV, Bruun TS, Zerrahn JE, Tauson AH, Hansen CF. The effect of increasing the dietary valine-to-lysine ratio on sow metabolism, milk production, and litter growth. J Anim Sci. 2016;94:155–64. https://doi.org/1
0.2527/jas.2015-9267.
25. Dennis MD, Baum JI, Kimball SR, Jefferson LS. Mechanisms involved in the coordinate regulation of mTORC1 by insulin and amino acids. J Biol Chem.
2011;286(10):8287–96. https://doi.org/10.1074/jbc.M110.209171.
26. Suryawan A, Nguyen HV, Almonaci RD, Davis TA. Differential regulation of protein synthesis in skeletal muscle and liver of neonatal pigs by leucine through an mTORC1-dependent pathway. J Anim Sci Biotechnol. 2012;3(1):
3. https://doi.org/10.1186/2049-1891-3-3.
27. Lei J, Feng D, Zhang Y, Zhao FQ, Wu Z, San Gabriel A, et al. Nutritional and regulatory role of branched-chain amino acids in lactation. Front Biosci (Landmark edition). 2012;17:2725–39.
28. Zhang JM, He WL, Yi D, Zhao D, Song Z, Hou YQ, et al. Regulation of protein synthesis in porcine mammary epithelial cells by L-valine. Amino
Acids. 2019;51(4):717–26. https://doi.org/10.1007/s00726-019-02709-2.
29. Li XL, Bazer FW, Johnson GA, Burghardt RC, Erikson DW, Frank JW, et al.
Dietary supplementation with 0.8% L-arginine between days 0 and 25 of gestation reduces litter size in gilts. J Nutr. 2010;140(6):1111–6. https://doi. org/10.3945/jn.110.121350.
30. Rezaei R. Nutritional and regulatory roles for branched-chain amino acids in milk production by lactating sows. College Station: PhD Dissertation. Texas
A&M University; 2015.
31. Kim SW, Wu G. Regulatory role for amino acids in mammary gland growth and milk synthesis. Amino Acids. 2009;37(1):89–95. https://doi.org/10.1007/ s00726-008-0151-5.
32. AOAC. Official method of analysis, 15th ed. Washington DC: Association of
Official Analytical Chemists; 1990.
33. Marier JR, Boulet M. Direct analysis of lactose in milk and serum. J Dairy Sci.
1959;42(8):1390–1. https://doi.org/10.3168/jds.S0022-0302(59)90747-7.
34. Dole VP. A relation between non-esterified fatty acids in plasma and the metabolism of glucose. J Clin Invest. 1956;35(2):150–4. https://doi.org/10.11
72/JCI103259.
35. Wu G, Davis PK, Flynn NE, Knabe DA, Davidson JT. Endogenous synthesis of arginine plays an important role in maintaining arginine homeostasis in postweaning growing pigs. J Nutr. 1997;127(12):2342–9. https://doi.org/10.1
093/jn/127.12.2342.
36. Dai ZL, Wu ZL, Jia SC, Wu G. Analysis of amino acid composition in proteins of animal tissues and foods as pre-column o-phthaldialdehyde derivatives by HPLC with fluorescence detection. J Chromatogr B. 2014;964:116–27. https://doi.org/10.1016/j.jchromb.2014.03.025.
37. Lassala A, Bazer FW, Cudd TA, Datta S, Keisler DH, Satterfield MC, et al.
Parenteral administration of L-arginine enhances fetal survival and growth in sheep carrying multiple pregnancies. J Nutr. 2011;141(5):849–55. https:// doi.org/10.3945/jn.111.138172.
38. Tekwe CD, Lei J, Yao K, Rezaei R, Li XL, Dahanayaka S, et al. Oral administration of interferon tau enhances oxidation of energy substrates and reduces adiposity in Zucker diabetic fatty rats. Bio Factors. 2013;39(5):
552–63. https://doi.org/10.1002/biof.1113.
39. Draper HH, Squires EJ, Mahmoodi H, Wu J, Agarwal S, Hadley M. A comparative evaluation of thiobarbituric acid methods for the determination of malondialdehyde in biological materials. Free Radic Biol
Med. 1993;15(4):353–63. https://doi.org/10.1016/0891-5849(93)90035-S.
40. Assaad H, Hou YQ, Zhou L, Carroll RJ, Wu G. Rapid publication-ready MSword tables for two-way ANOVA. Springer Plus. 2015;4(1):33. https://doi. org/10.1186/s40064-015-0795-z.
41. Steel RGD, Torrie JH. Principles and procedures of statistics. New York:
McGraw-Hill; 1980.
42. Kensinger RS, Collier RJ, Bazer FW, Ducsay CA, Becker HN. Nucleic acid, metabolic and histological changes in gilt mammary tissue during pregnancy and lactogenesis. J Anim Sci. 1982;54(6):1297–308. https://doi. org/10.2527/jas1982.5461297x.
43. Shennan D, Peaker M. Transport of milk constituents by the mammary gland. Physiol Rev. 2000;80(3):925–51. https://doi.org/10.1152/physrev.2000.
80.3.925.
44. Guan X, Bequette BJ, Ku PK, Tempelman RJ, Trottier NL. The amino acid need for milk synthesis is defined by the maximal uptake of plasma amino acids by porcine mammary glands. J Nutr. 2004;134(9):2182–90. https://doi. org/10.1093/jn/134.9.2182.
45. Kim SW, Hurley WL, Han IK, Easter RA. Growth of nursing pigs related to the characteristics of nursed mammary glands. J Anim Sci. 2000;78(5):1313–8. https://doi.org/10.2527/2000.7851313x.
46. Hughes PE, Varley MA. Reproduction in the pig. London: Butterworth; 1980.
47. Wholt JE, Clark JH, Derrig RG, Davis CL. Valine, leucine, and isoleucine metabolism by lactating bovine mammary tissue. J Dairy Sci. 1977;60(12):
1875–82. https://doi.org/10.3168/jds.S0022-0302(77)84118-0.
48. Viña JR, Williamson DH. Effects of lactation on L-leucine metabolism in the rat. Biochem J. 1981;194(3):941–7. https://doi.org/10.1042/bj194
0941.
49. Wu G. Dietary requirements of synthesizable amino acids by animals: a paradigm shift in protein nutrition. J Anim Sci Biotechnol. 2014;5(1):34. https://doi.org/10.1186/2049-1891-5-34.
50. Rulquin H, Pisulewski P. Effects of graded levels of duodenal infusions of leucine on mammary uptake and output in lactating dairy cows. J Dairy
Res. 2006;73(3):328–39. https://doi.org/10.1017/S0022029906001841.
51. Wu G. Amino acids: biochemistry and nutrition. 2nd ed. Boca Raton: CRC
Press; 2022.
52. Kim SW, Hurley WL, Wu G, Ji F. Ideal amino acid balance for sows during gestation and lactation. J Anim Sci. 2009;87(suppl_14):E123–32. https://doi. org/10.2527/jas.2008-1452.
53. Dosako S, Taneya S, Kimura T, Ohmori T, Daikoku H, Suzuki N, et al. Milk of northern fur seal: composition, especially carbohydrate and protein. J Dairy
Sci. 1983;66(10):2076–83. https://doi.org/10.3168/jds.S0022-0302(83)82053-0.
54. Escobar J, Frank JW, Suryawan A, Nguyen HV, Kimball SR, Jefferson LS, et al.
Regulation of cardiac and skeletal muscle protein synthesis by individual branched-chain amino acids in neonatal pigs. Am J Phys. 2006;290(4):E612–
21. https://doi.org/10.1152/ajpendo.00402.2005.
55. Manjarin R, Zamora V, Wu G, Steibel JP, Kirkwood RN, Taylor NP, et al.
Effect of amino acids supply in reduced crude protein diets on performance, efficiency of mammary uptake, and transporter gene expression in lactating sows. J Anim Sci. 2012;90(9):3088–100. https:// doi.org/10.2527/jas.2011-4338.
56. Trottier NL, Shipley CF, Easter RA. Plasma amino acid uptake by the mammary gland of the lactating sow. J Anim Sci. 1997;75(5):1266–78. https://doi.org/10.2527/1997.7551266x.
57. Nielsen TT, Trottier NL, Stein HH, Bellaver C, Easter RA. The effect of litter size and day of lactation on amino acid uptake by the porcine mammary glands. J Anim Sci. 2002;80(9):2402–11. https://doi.org/10.252
7/2002.8092402x.
58. Conway ME, Hutson SM. Mammalian branched-chain aminotransferases.
Methods Enzymol. 2000;324:355–65. https://doi.org/10.1016/S0076-
6879(00)24245-6.
59. Matsumoto T, Nakamura E, Nakamura H, Hirota M, San Gabriel A, Nakamura
KI, et al. The production of free glutamate in milk requires the leucine transporter LAT1. Am J Phys. 2013;305(6):C623
–31. https://doi.org/10.1152/a jpcell.00291.2012
.
60. Nissen S, Faidley TD, Zimmerman DR, Izard R, Fisher CT. Colostral milk fat percentage and pig performance are enhanced by feeding the leucine metabolite
β-hydroxyβ-methyl butyrate to sow. J Anim Sci. 1994;72(9):
2331
–7. https://doi.org/10.2527/1994.7292331x
.
61. Wang WW, Dai ZL, Wu ZL, Lin G, Jia SC, Hu SD, et al. Glycine is a nutritionally essential amino acid for maximal growth of milk-fed young pigs. Amino Acids. 2014;46(8):2037
–45. https://doi.org/10.1007/s00726-014-1
758-3
.
62. Wang WW, Wu ZL, Lin G, Hu SD, Wang B, Dai ZL, et al. Glycine stimulates protein synthesis and inhibits oxidative stress in pig small-intestinal epithelial cells. J Nutr. 2014;144(10):1540
–8. https://doi.org/10.3945/jn.114.1
94001
.
63. Columbus DA, Fiorotto ML, Davis TA. Leucine is a major regulator of muscle protein synthesis in neonates. Amino Acids. 2015;47(2):259
–70. https://doi. org/10.1007/s00726-014-1866-0
.
64. Appuhamy JA, Knoebel NA, Nayananjalie WD, Escobar J, Hanigan M.
Isoleucine and leucine independently regulate mTOR signaling and protein synthesis in MAC-T cells and bovine mammary tissue slices. J Nutr. 2012;
142(3):484
–91. https://doi.org/10.3945/jn.111.152595
.
65. Yin YL, Yao K, Liu Z, Gong M, Ruan Z, Deng D, et al. Supplementing Lleucine to a low-protein diet increases tissue protein synthesis in weanling pigs. Amino Acids. 2010;39(5):1477
–86. https://doi.org/10.1007/s00726-010-
0612-5
.
66. Wang WW, Wu ZL, Dai ZL, Yang Y, Wang JJ, Wu G. Glycine metabolism in animals and humans: implications for nutrition and health. Amino Acids.
2013;45(3):463
–77. https://doi.org/10.1007/s00726-013-1493-1
.
67. Reeds PJ, Burrin DG. Glutamate and the bowel. J Nutr. 2001;131(9):2505S
–8S. https://doi.org/10.1093/jn/131.9.2505S
.
68. Wu G, Bazer FW, Johnson GA, Knabe DA, Burghardt RC, Spencer TE, et al.
Important roles for L-glutamine in swine nutrition and production. J Anim
Sci. 2011;89(7):2017
–30. https://doi.org/10.2527/jas.2010-3614
.
69. Rezaei R, Wang WW, Wu ZL, Dai ZL, Wang JJ, Wu G. Biochemical and physiological bases for utilization of dietary amino acids by young pigs. J
Anim Sci Biotechnol. 2013;4(1):7. https://doi.org/10.1186/2049-1891-4-7
.
70. Yao K, Yin YL, Chu WY, Liu ZQ, Deng D, Li TJ, et al. Dietary arginine supplementation increases mTOR signaling activity in skeletal muscle of neonatal pigs. J Nutr. 2008;138(5):867
–72. https://doi.org/10.1093/jn/138.5.
867
.
71. Xi PB, Jiang Z, Zheng C, Lin Y, Wu G. Regulation of protein metabolism by glutamine: implications for nutrition and health. Front Biosci (Landmark edition). 2011;16(1):578
–97. https://doi.org/10.2741/3707
.
72. Li XL, Johnson GA, Zhou HJ, Burghardt RC, Bazer FW, Wu G. Microarray analysis reveals an important role for dietary L-arginine in regulating global gene expression in porcine placentae during early gestation. Front Biosci (Landmark edition). 2022;27(1):033.
73. Iwasa M, Kobayashi Y, Mifuji-Moroka R, Hara N, Miyachi H, Sugimoto R, et al.
Branched-chain amino acid supplementation reduces oxidative stress and prolongs survival in rats with advanced liver cirrhosis. PLoS One. 2013;8(7): e70309. https://doi.org/10.1371/journal.pone.0070309
.
74. Weber MG, Dias SS, de Angelis TR, Fernandes EV, Bernardes AG, Milanez VF, et al. The use of BCAA to decrease delayed-onset muscle soreness after a single bout of exercise: a systematic review and meta-analysis. Amino Acids.
2021;53(11):1663
–78. https://doi.org/10.1007/s00726-021-03089-2
.
75. Wu G. Functional amino acids in nutrition and health. Amino Acids. 2013;
45(3):407
–11. https://doi.org/10.1007/s00726-013-1500-6
.
76. Wu G, Li L. The
“ideal protein
” concept is not ideal in animal nutrition. Exp
Biol Med. 2022. https://doi.org/10.1177/15353702221082658
.
77. Rezaei R, San Gabriel A, Wu G. Dietary supplementation with monosodium glutamate enhances milk production by lactating sows and the growth of suckling piglets. Amino Acids. 2022. https://doi.org/10.1007/s00726-022-0314
7-3
.
78. He WL, Wu G. Oxidation of amino acids, glucose, and fatty acids as metabolic fuels in enterocytes of developing pigs. Amino Acids. 2022. https://doi.org/10.1007/s00726-022-03151-7







