Reaction of Banana Cultivars to The Meloidogyne Javanica X Fusarium Oxysporum F. Sp. Cubense Complex
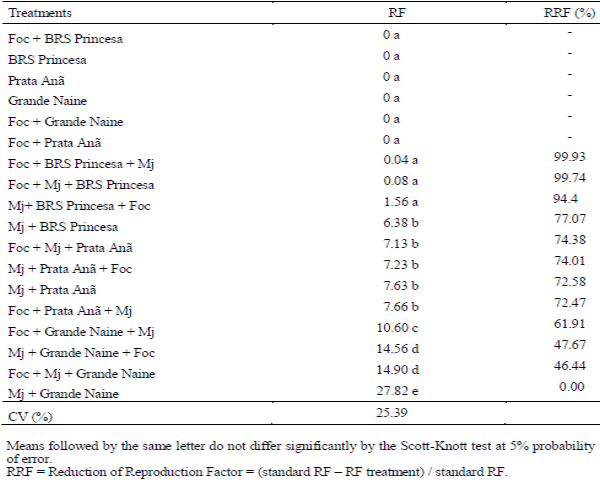
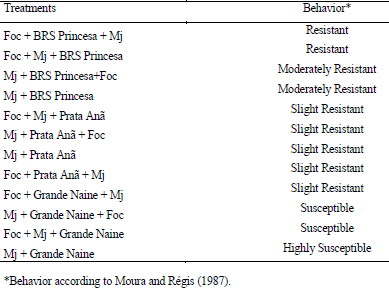
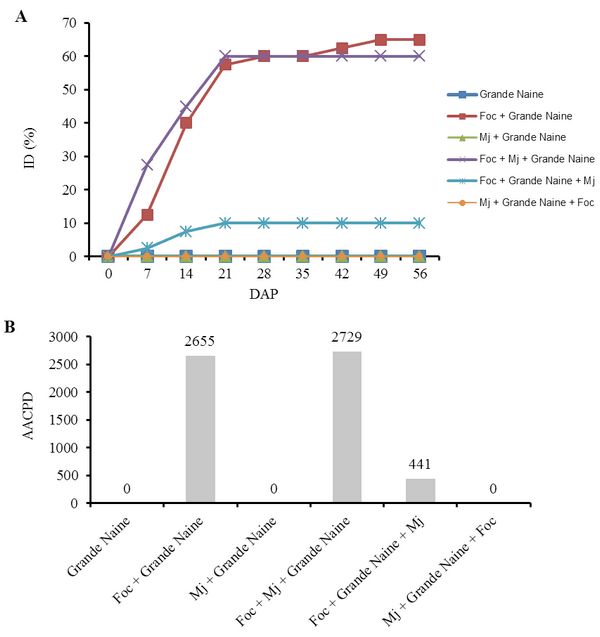
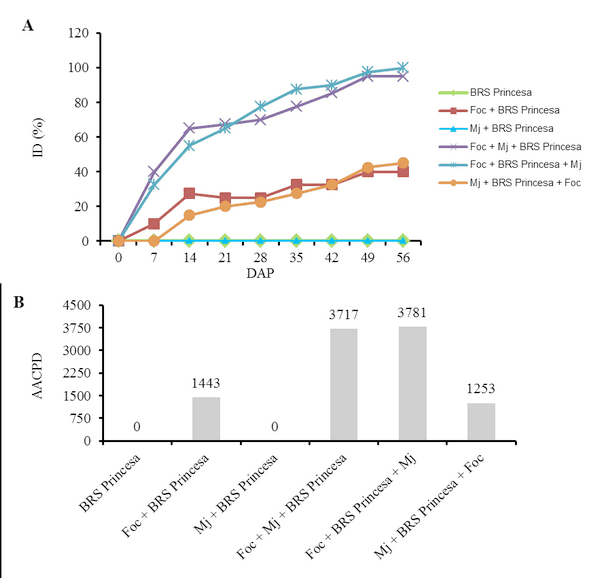
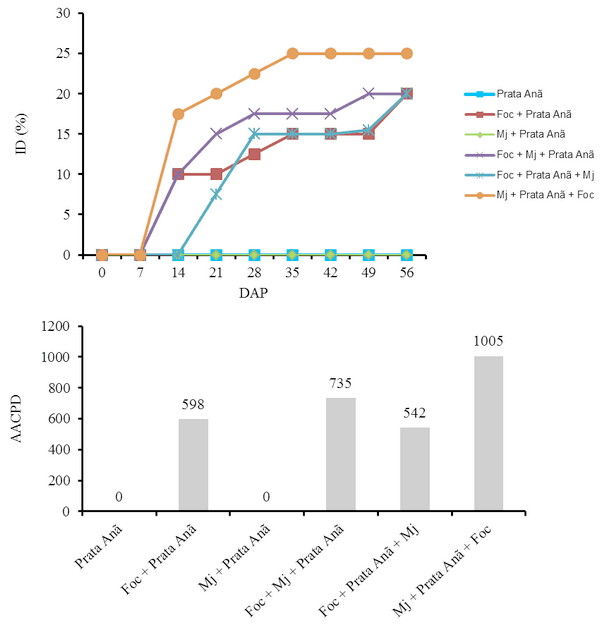
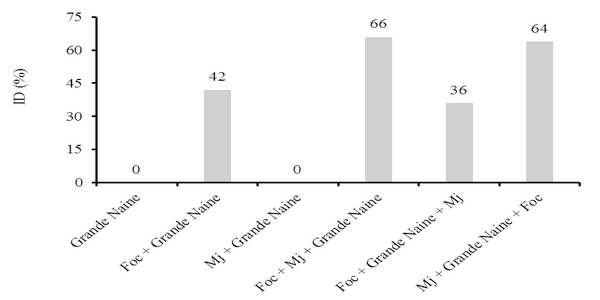
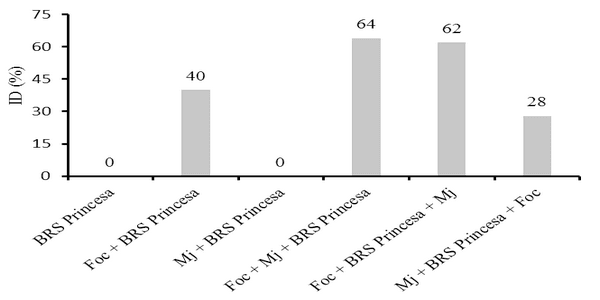
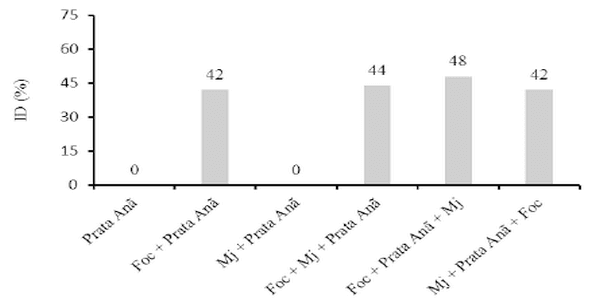
The interaction Fusarium oxysporum f. sp. cubense (Foc) x nematode and the genetic variability of Foc are the main problems with potential to affect the use of resistant varieties in the management of the Panama disease. The objective of this work was to evaluate the interaction between Foc and Meloidogyne javanica on the banana of the Prata Anã, Grande Naine and BRS Princesa cultivars. The experiment was conducted in a randomized block design, with six treatments and ten replications, which differed in the inoculation time of each pathogen. Simultaneous inoculation with Foc and M. javanica, inoculation with Foc one week before inoculation with M. javanica, and inoculation with M. javanica one week before inoculation with Foc. In addition to the controls, Foc isolated, M. javanica isolated, and cultivars without the pathogens. The seedlings were transplanted in 3-liter pots with sterile soil infested with 40 grams of Foc inoculum at the concentration of 1x106CFU/gram and 1000 eggs and J2 of M. javanica. At the end of 56 days, the treatments with 'Grande Naine' presented the highest reproduction factors of M. javanica. In the 'Prata Anã' no interaction was observed between Foc x M. javanica. The treatments with simultaneous inoculation of the two pathogens, and with Foc a week before, in the 'BRS Princesa', presented the highest external disease indices (DI), which promoted the highest AUDPC. The highest internal DI were observed in the treatments Foc+Mj+Grande Naine, Foc+Mj+BRS Princesa, Mj+Grande Naine+Foc and Foc+BRS Princesa+Mj. The simultaneous presence of M. javanica and Foc increases the severity of the Panama disease in Grande Naine and BRS Princesa cultivars.
Keywords: Musa spp.. Fusarium wilt. Root-knot nematodes. Interaction. Panama disease.
ALFENAS, A. C.; PETERS, W.; PASSADOR, G. C. Eletroforese de proteínas e isoenzimas de fungos e essências florestais. 1. ed. Viçosa, MG: UFV, 1991. 242 p.
BONETI, J. I. S.; FERRAZ, S. Modificação do método de Hussey e Barker para extração de ovos de Meloidogyne exigua de cafeeiro. Fitopatologia Brasileira, Brasília, v. 6, n. 3, p. 553, 1981.
BRANDES, E. W. Banana wilt. Phytopathology, Saint Paul, v. 9, s/n., p. 339-389, 1919.
CAMPOS, H. D. et al. Atração e penetração de Meloidogyne javanica e Heterodera glycines em raízes excisadas de soja. Ciencia Rural, Santa Maria, v. 41, n. 9, p. 1496-1502, 2011.
CARNEIRO, M. D. G.; ALMEIDA, M. R. A. Técnica de eletroforese usada no estudo de enzimas dos nematoides de galhas para identificação de espécies. Nematologia Brasileira, Brasília, v. 25, n. 1, p. 35-44, 2001.
COFCEWICZ, E. T. et al. Reação de cultivares de bananeira a diferentes espécies de nematóides das galhas. Nematologia Brasileira, Brasília, v. 28, n. 1, p. 11-22, 2004.
CORDEIRO, Z. J. M.; MATOS, A. P.; HADDAD, F. Doenças Fúngicas e bacterianas. In: FERREIRA, C. F. et al. (Eds.). O agronegócio da banana. Brasília: Embrapa, 2016. v. 1, cap. 16, p. 545-575.
CORDEIRO, Z. J. M. et al. Avaliação de resistência ao mal-do-Panamá em híbridos tetraploides de bananeira. Fitopatologia Brasileira, Brasília, v. 18, n. 4, p. 478-483, 1993.
COSTA, D. C. et al. Nematoides. In: FERREIRA, C. F. et al. (Eds.). O agronogócio da banana. Brasília: Embrapa, 2016. v. 1, cap. 15, p. 505-543.
COSTA, S. N. et al. Genetic structure of Fusarium oxysporum f. sp. cubense in different regions from Brazil. Plant Pathology, London, v. 64, n. 1, p. 137-146, 2015.
DAVIS, E. L.; HUSSEY, R. S.; BAUM, T. J. Getting to the roots of parasitism by nematodesTrends in Parasitology, Cambridge, v. 20, n. 3, p. 134-141, 2004.
DAVIS, E. L. et al. Nematode Parasitism Genes. Annual Review of Phytopathology, Palo Alto, v. 38, s/n., p. 365-396, 2000.
DITA, M. A.; PÉREZ VICENTE, L.; MARTINEZ DE LA PARTE, E. Inoculation of Fusarium oxysporum f. sp. cubense causal agent of fusarium wilt in banana. In: PÉREZ-VICENTE, L.; DITA, M. A.; MARTINEZ-DE LA PARTE, E. (Eds.). Technical Manual: Prevention and diagnostic of Fusarium Wilt of banana caused by Fusarium oxysporum f. sp. cubense Tropical Race 4 (TR4). Trinidad and Tobago: FAO/CARDI, 2014. v. 1, p. 55-58.
EISENBACK, J. D. Diagnostic characters useful in the identification of the four most common species of root-knot nematodes (Meloidogyne spp.). In: SASSER, J. N; CARTER, C. C. (Eds.). An advanced treatise on Meloidogyne. Raleigh: North Carolina State University Graphics, 1985. v. 1, cap. 8, p. 95-112. ,
ESBENSHADE, P. R.; TRIANTAPHYLOU, A. C. Use of enzyme phenotypes for identification of Meloidogyne species. Journal of Nematology. Bethesda, v. 17, n. 1, p. 6-20, 1985.
ESBENSHADE, P. R.; TRIANTAPHYLOU, A. C. Isozyme phenotypes for the identification of Meloidogyne species. Journal of Nematology, Bethesda, v. 22, n. 1, p. 10-15, 1990.
FERREIRA, D. F. Sisvar: a computer statistical analysis system. Ciência e Agrotecnologia, Lavras, v. 35, n. 6, p. 1039-1042, 2011.
FISCHER, I. H. et al. Reação de maracujazeiro-amarelo ao complexo fusariose-nematoide de galha. Acta Scientiarum Agronomy, Maringá, v. 32, n. 2, p. 223-227, 2010.
GRIFFIN, G. D.; THYR, B. D. Interaction of Meloidogyne hapla and Fusarium oxysporum f. sp. medicaginis on alfafa. Phytopathology, Saint Paul, v. 78, n. 4, p. 421-425, 1988.
HIRSCHMANN, H. The genus Meloidogyne and morphological characters differentiating its species. In: BARKER, K. R.; CARTER, C. C.; SASSER, J. N. (Eds.). An advanced treatise on Meloidogyne. Raleigh: North Carolina State University Graphics, 1985. v. 2, cap. 2, p. 79-93.
HUSSEY, R. S.; DAVIS, E. L.; BAUM, T. J. Secrets in secretions: genes that control nematode parasitism of plants. Brazilian Journal of Plant Physiology, Campos dos Goytacazes, v. 14, n. 3, p. 183-194, 2002.
HWANG, S. C.; KO, W. H. Cavendish banana cultivars resistant to Fusarium Wilt acquired through somaclonal variation in Taiwan. Plant Disease, Saint Paul, v. 88, n. 6, p. 580-588, 2004.
INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA - IBGE. Levantamento Sistemático da Produção Agrícola. Disponível em: <http://www.ibge.gov.br/home/>. Acesso em: 08 ago. 2016.
JENKINS, W. R. A rapid centrifugal flotation technique for separating nematodes from soil. Plant Disease Reporter, Beltsville, v. 48, n. 9, p. 692, 1964.
JONES, D. R. Diseases of Banana, Abacá and Enset. Wallingford, UK: CABI Publishing, 2000. 544 p.
KNOGGE, W. Fungal infection of plants. The Plant Cell, Los Angeles, v. 8, n. 10, p. 1711-1722, 1996.
KUNIEDA DE ALONSO, S. et al. Análise de isoenzimas para identificação de espécies de Meloidogyne. Fitopatologia Brasileira, Brasília, v. 20, n. 1, p. 20-23, 1995.
MCKINNEY, R. H. Influence of soil temperature and moisture on infection of wheat seedlings by Helminthosporium sativum. Journal of Agricultural Research, Washington, v. 26, n. 5, p. 195-217, 1923.
MOURA, R. M.; REGIS, E. M. O. Reações de feijoeiro comum (Phaseolus vulgaris) em relação ao parasitismo de Meloidogyne javanica e M. incognita (Nematoda: Heteroderidae). Nematologia Brasileira, Piracicaba, v. 11, s/n., p. 215-225, 1987.
OOSTENBRINK, M. Major characteristics of the relation between nematodes and plants. Mededlingen Landbouwhogeschool, Wageningen, v. 66, s/n., p. 3-46, 1966.
PLOETZ, R. C. Management of Fusarium wilt of banana: A review with special reference to tropical race 4. Crop Protection, Lincoln, p. 1-9, 2015.
PLOETZ, R. C.; PEGG, K. G. Fusarium wilt. In: JONES, D. R. (Eds.). Diseases of Banana, Abacá and Enset. Wallingford, UK: CABI Publishing, 2000. p. 143-159.
RIBEIRO, R. C. F. et al. Rizobactérias no controle de Meloidogyne javanica e mal-do-Panamá em bananeira. Nematropica, Jackson, v. 42, n. 2, p. 218-226, 2012.
SILVA, G. S.; PEREIRA, A. L. Efeito da incorporação de folhas de nim ao solo sobre o complexo Fusarium x Meloidogyne em quiabeiro. Summa Phytopathologica, Botucatu, v. 34, n. 4, p. 368-370, 2008.
SIMÃO, G. et al. Reação de cultivares e linhagens de feijoeiro em relação a Meloidogyne javanica e Fusarium oxysporum f. sp. phaseoli. Ciência Rural, Santa Maria, v. 40, n. 5, p. 1003-1008, 2010.
STOVER, R. H.; BUDDENHAGEN, I. W. Banana breeding: Polyploidy, disease resistance and productivity. Fruits, Leuven, v. 41, n. 3, p. 175-191, 1986.
TENENTE, R. C. et al. Reaction of Different Banana (Musa spp.) Cultivars to Meloidogyne incognita Races 1 and 4. Nematologia Brasileira, Brasília, v. 32, n. 4, p. 285-293, 2008.







.jpg&w=3840&q=75)