INTRODUCTION
Mycotoxins are structurally diverse compounds produced by filamentous fungi. Contamination of foods and feeds with mycotoxins is a significant problem worldwide (Hussein and Brasel, 2001). The major Fusarium mycotoxins occurring in cereal grains, animal feeds, and forages are the trichothecenes, zearalenone (ZEN), and fumonisins. Other important Fusarium mycotoxins include moniliformin and fusaric acid (D’Mello et al., 1999). More than 180 trichothecenes have been isolated and characterized (Pestka and Smolinski, 2005). The toxic effects of Fusarium mycotoxins in animals and poultry include reduced growth, feed refusal and vomiting, immunosuppression, gastrointestinal lesions, and neurological and reproductive disorders (Rocha et al., 2005).
Effects of feeding Fusarium mycotoxins from naturally contaminated grains on performance and health have been documented in broiler chickens (Swamy et al., 2002, 2004; Danicke et al., 2003), laying hens (Danicke et al., 2002; Chowdhury et al., 2005b), ducks (Danicke et al., 2004; Chowdhury et al., 2005a), and broiler breeders (Yegani et al., 2006). There are very few reports of the effects of feeding of grains naturally contaminated with Fusarium mycotoxins to turkeys. Chowdhury (2005) observed reductions in weight gain, plasma total protein, albumin, globulin, Ca, bilirubin, and uric acid concentrations. It was also observed that such treatments caused alterations in hematology, increased levels of biliary IgA, and decreased the response to dinitrochlorobenzene (Chowdhury et al., 2005c). Feeding turkey poults pure T-2 toxin or diacetoxyscirpenol (DAS) at levels up to 1 mg/kg of feed for 32 d adversely influenced small intestinal morphology but did not affect growth or antibody production (Sklan et al., 2003). The feeding of both T-2 toxin and DAS, however, resulted in severe oral lesions. It has been reported that the feeding of fusarial culture materials increased the redness of turkey breast meat (Wu et al., 1994). McMillan et al. (1986) found turkeys to be resistant to feedborne semipurified deoxynivalenol (DON) at up to 15 mg/kg of feed. Similarly, when turkey poults were fed diets containing 4.4 mg of DON/kg of feed in naturally contaminated corn for 3 wk, there were no adverse effects on feed consumption or BW gain (Manley et al., 1988). It has also been shown that there were no adverse effects of feeding 4.6 mg of DON/kg of feed from naturally contaminated wheat for 5 wk (Hamilton et al., 1985).
These discrepancies may be attributable to the source of mycotoxins, dose, and duration of exposure, and mycotoxins from naturally contaminated sources may be more toxic than an equivalent amount of purified compound (Harvey et al., 1991). This is probably due to the presence of the unidentified mycotoxins and precursors in naturally contaminated grains resulting in additive or synergistic effects among mycotoxins (Smith et al., 1997). There is a dearth of knowledge regarding the effects feeding of low concentrations of naturally produced Fusarium mycotoxins on production, health, and immunocompetence to turkeys.
A polymeric glucomannan mycotoxin adsorbent (GMA) derived from the cell wall of yeast has been shown to prevent some of the deleterious effects of Fusarium mycotoxins on performance and metabolism of poultry (Swamy et al., 2002, 2004; Chowdhury et al., 2005a,b,c). The current experiment was conducted, therefore, to study the effects of feeding blends of grains naturally contaminated with Fusarium mycotoxins on performance, metabolism, hematology, and immunological parameters of turkeys and to determine the efficacy of GMA in preventing these effects.
MATERIALS AND METHODS
Experimental Birds and Diets
Three hundred 1-d-old Hybrid turkey poults (Hybrid Turkeys, Kitchener, Ontario, Canada) were individually weighed, wing-banded, and distributed randomly into groups of 20 poults per floor pen at the Arkell Poultry Research Station of the University of Guelph. Five pens were randomly assigned to each of the 3 diets with each diet fed to 100 poults. Poults were initially maintained at 32°C, and the temperature was gradually reduced by 3°C per week to reach a temperature of 21°C by the end of wk 4. This temperature was maintained for the duration of the experiment. Turkey poults were fed wheat, corn, and soybean meal-based starter (0 to 3 wk), grower (4 to 6 wk), developer (7 to 9 wk), and finisher (10 to 12 wk) diets formulated with control grains, contaminated grains, or contaminated grains + 0.2% GMA. The control diet was formulated to meet or exceed the minimum nutrient requirements of turkeys according to the NRC (1994). Mycotoxin-contaminated diets were prepared by replacing 10 and 35% of the control corn and wheat with contaminated corn and wheat naturally contaminated with Fusarium mycotoxins. The levels of replacement of control grains with the contaminated grains were the same in all growth phases to achieve as close as possible a constant mycotoxin challenge. The GMA-supplemented diets were prepared by substituting control corn in the contaminated diet with 0.2% GMA. Feed and water were provided ad libitum. Representative feed samples were taken at the beginning of each phase for proximate and mycotoxin analyses. Dietary contents of protein, DM, and ash were determined according to the AOAC (1980). The experimental procedures were approved by the University of Guelph Animal Care Committee following the guidelines of the Canadian Council on Animal Care.
Quantification of Dietary Mycotoxin Concentrations
Dietary concentrations of DON, 3-acetyl DON, 15-acetyl DON, T-2 toxin, iso T-2 toxin, acetyl T-2 toxin, HT-2 toxin, T-2 triol, T-2 tetraol, fusarenon-X, DAS, scirpentriol, 15-acetyl scirpenol, neosolaniol, ZEN, and zearalenol were analyzed at Veterinary Diagnostic Laboratory, North Dakota State University, Fargo, using a combination of gas chromatography and mass spectrometry as described by Groves et al. (1999) and modified by Raymond et al. (2003). Aflatoxins were determined using a Shimadzu HPLC (Shimadzu VP system with a LC-10A pump, Shimadzu Corp., Tokyo, Japan), and fumonisin analysis was performed on an Agilent HPLC (1100 series, Agilent Technologies Inc., Palo Alto, CA; Leung et al., 2007). The detection limits for aflatoxins and fumonisins were 0.02 and 2 mg/kg, respectively. Fusaric acid was estimated by the HPLC method of Matsui and Watanabe (1988) as modified by Smith and Sousadias (1993) and confirmed by Porter et al. (1995).
Experimental Parameters Measured
BW, Feed Consumption, and Organ Weights. Poults were weighed individually, and feed consumption for each pen was measured at the end of each growth phase during the 12-wk experiment. Weight gain, feed consumption, and cumulative gain:feed ratios were calculated at the end of each growth phase. Feed consumption and gain:feed were adjusted for mortalities when necessary. At the end of the experiment, 16 birds from each pen were killed by instantaneous cervical dislocation. Liver, kidney, spleen, pancreas, heart, gizzard, proventriculus, and bursa of Fabricius were excised and weighed. Organ weights were expressed on a relative BW basis.
Hematology. At the end of wk 3, 6, 9, and 12, blood samples were collected from the jugular vein of 2 birds per pen (10 birds/diet) for hematological and plasma chemistry analyses. Hemoglobin and hematocrit were measured, and mean corpuscular hemoglobin concentrations (MCHC) were calculated as MCHC = hemoglobin/ hematocrit. Total white blood cell and differential leukocyte counts (heterophils, lymphocytes, monocytes, and basophils) were also determined (Veterinary Diagnostic Laboratory, University of Guelph, Guelph, Ontario, Canada).
Plasma Chemistry. Plasma concentrations of Ca, P, total protein, albumin, globulins, glucose, cholesterol, bilirubin, and uric acid and activities of amylase, lipase, aspartate aminotransferase, γ-glutamyltransferase, lactate dehydrogenase, glutamate dehydrogenase, and creatine kinase were also determined after 3, 6, 9, and 12 wk of feeding (Veterinary Diagnostic Laboratory, University of Guelph).
Phenotyping of Peripheral Blood Lymphocytes. Peripheral blood mononuclear cells were separated from blood after 3, 6, 9, and 12 wk of feeding using density gradient medium (Histopaque-1077, Sigma Chemical Co.) for phenotyping and lymphoblastogensis assays. Fifty microliters of cell suspension in PBS containing 1 million peripheral blood mononuclear cells was transferred to a 96-well round-bottom plate (Sarstedt Inc., Newton, NC) and resuspended with the appropriate antibody (Southern Biotechnology Associates, Birmingham, AL). Cells were double-stained with mouse antichicken CD4 antibody (CT-4 clone, mouse IgG1K conjugated to fluorescein isothyocyanate, 1μg/106 cells) and mouse antichicken CD8 antibody (3-298 clone, mouse IgG2bK conjugated to R-phycoerythrin, 0.2 μg/106 cells) to identify CD4+ and CD8+ T cells, respectively. The B lymphocytes were bound with mouse antichicken IgM (μ heavy chain specific) unlabeled primary antibody (M-4 clone, mouse IgMK, 1 μg/ 106 cells) and labeled with R-phycoerythrin-conjugated goat antimouse IgM (μ heavy chain specific, 0.1 μg/106 cells) secondary antibody. Appropriate isotype controls (Southern Biotechnology Associates, Birmingham, AL) were included in the analysis and were subtracted from the sample values. The isotype control was used to determine nonspecific binding of the lymphocyte subset-specific antibodies and to set the cut-off between fluorescence-negative and fluorescence-positive staining. Ten thousand cells were acquired and analyzed using a Becton-Dickinson FACScan flow cytometer and Cell Quest software of Becton-Dickinson Immunocytometry System (San Jose, CA). For data analysis, a size (forward scatter) vs. internal complexity (side scatter) dot plot was generated, and a region was drawn around the small live cell population containing the lymphocyte population. This region, however, may include variable amounts of thrombocytes and red blood cells. The cell population data obtained from the quadrant statistics (2-color staining) were standardized for the proportion of lymphocytes using the sum of CD4+, CD8+, and IgM+ cells within this region (e.g., CD4− CD8− γg-δ were not accounted). Lymphocyte subset data were then adjusted by calculation to yield an estimate of the percentage of CD4+ lymphocytes, CD8+ lymphocytes, and B lymphocytes.
Lymphocyte Blastogenesis Assay. A 3-4, 5-dimethyl-thiazol 2, 5-diphenyl tetrazolium bromide (MTT; Sigma Chemical Co., St. Louis, MO) assay was used to assess the proliferation of lymphocytes in response to 2 mitogens, concanavalin A (Con A) and pokeweed mitogen (PWM), and was purchased from Sigma Chemical Co. After washing in PBS, blood lymphocytes were adjusted to 2 × 106 viable cells in RPMI-1640 cell culture media (Sigma Chemical Co.) with L-Gln (2 mM), penicillin (100 U/mL), and streptomycin (100 μg/mL; Sigma Chemical Co.). One hundred microliters of cell suspension (2 × 106 cells) was added to a 96-well, flat-bottomed tissue culture plate (Nunclon, Roskilde, Denmark), and cells were incubated with either Con A (10 μg/mL) or PWM (8 μg/mL). Plates were incubated at 37°C in a CO2 incubator at 5% CO2 tension. After 72 h, 10 μL of MTT in RPMI1640 culture media (5 mg/mL) was added, and the plates were incubated for 4 h. The MTT formazan was extracted using 150 μL of dimethylsulfoxide (Sigma Chemical Co.) per well, and the optical density (OD) was measured at 510 nm and with a reference of 650 nm. A stimulation index was calculated using the formula OD stimulated/ OD of unstimulated control.
Analysis of Ig in Serum and Bile. Two milliliters of blood was collected from the jugular vein of 2 birds per replicate (10 birds/diet) after 3, 6, 9, and 12 wk of feeding. Birds were euthanized after 12 wk of feeding, and bile was collected. Serum and bile samples were frozen at −80°C until further analysis. The IgA was analyzed in serum and bile samples, whereas other Ig including IgM and IgG were analyzed in serum samples. Immunoglobulins were analyzed using a sandwich ELISA technique (Bethyl Laboratories, Montgomery, TX). The concentrations of affinity purified polyclonal goat antichicken IgA (α-chain specific), goat antichicken IgM (μ-chain specific), and goat antichicken IgG (fragment crystallizable region fragment specific) coating antibodies were 0.01 mg/mL. The same polyclonal antibodies, conjugated with horseradish peroxidase (HRP), were used as the detection antibodies at concentrations each of 0.02 μg/mL for IgA and IgM and 0.025 μg/mL for IgG. Sample concentrations were determined from standard curves using graded concentrations of chicken reference sera (15.625 to 1,000 ng/ mL for IgA, 3.9 to 250 ng/mL for IgM, and 3.12 to 1,000 ng/mL for IgG).
Antibody Response to SRBC. During the eighth week of feeding, 2 birds per replicate (10 birds per diet) were injected intrajugularly with 1 mL of 5% SRBC (Cleveland Scientific, Bath, OH) in PBS (Fisher Scientific Ltd., Nepean, Ontario, Canada) immediately after the collection of preimmune sera. At wk 9 and 10, serum from jugular vein blood was collected to determine the primary immune response. The secondary immune response serum was collected at wk 11 and 12 after readministration of the SRBC at wk 10 as described above. Serum samples were frozen at −80°C until analysis. Serum IgM and IgG antibody titers specific to SRBC were measured according to Temple et al. (1995). Detection antibodies used in the assay were HRP conjugated goat antichicken IgM (μ- chain specific, 2 ng/mL; Bethyl laboratories) or HRP conjugated goat antichicken IgG (fragment crystallizable region-fragment specific, 2 ng/mL; Bethyl Laboratories). The antibody titer was defined as the highest dilution of the test serum whose absorbance was greater than the average absorbance plus 3 SD of 8 wells without serum.
Contact Hypersensitivity Response to Dinitrochlorobenzene. The procedure of Prescott et al. (1982) was followed with minor modifications to measure the response to dinitrochlorobenzene (DNCB). During wk 9, fifty microliters of 90% dimethylsulfoxide in deionized water was applied to an area of wing skin previously cleaned with 70% ethyl alcohol in deionized water (10 birds/diet). Ten minutes later, birds were sensitized by topical application of 50 μL of 2.5% DNCB (Sigma Chemical Co.) dissolved in acetone. The same procedure was subsequently conducted every 48 h for 6 d. Two weeks after the first sensitization, the right webfoot of the sensitized birds as well as that of 2 unsensitized birds per replicate (10 birds/diet) were challenged by topical application of 50 μL of 1% DNCB dissolved in a 4:1 mixture of acetone and olive oil. Fifty microliters of acetone:olive oil was applied to the left webfoot as an internal control for each bird. The thickness of the webfoot was measured before the challenge as well as 24, 48, and 72 h after the challenge using a constant-tension dial micrometer (Mitutoya Corp., Tokyo, Japan). Three measurements were taken from each webfoot, and the averages of 3 measurements were calculated. The percentage of thickness increase was calculated as follows: percentage of thickness increase = [(skin thickness after challenge − skin thickness before challenge)/skin thickness before challenge] × 100. The contact hypersensitivity (CHS) response resulting from challenge was calculated as the percentage of thickness increase in the right webfoot minus the percentage of thickness increase in the left webfoot. In turn, CHS response due to sensitization was calculated by subtracting the response to challenge exhibited by unsensitized birds from the response exhibited by sensitized birds.
Statistical Analyses
Data were analyzed by analysis of covariance (production parameters and relative organ weights) or variance using a PROC MIXED model of SAS based on a randomized complete block design with subsampling (Kuehl, 2000; SAS Institute, 2000). Pens were treated as individual experimental units, and rooms were treated as blocks. Multiple comparisons among the treatment least square means were made using Tukey’s test. Day-old BW was used as a covariate for analysis of BW, and 12-wk BW was used as covariate for organ weights analysis. Data representing percentages or ratios were transformed before statistical analysis. Statements of statistical significance were based on P ≤ 0.05.
RESULTS
Dietary Mycotoxin Concentrations
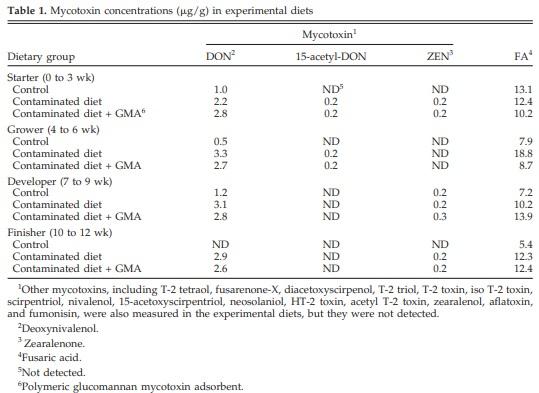
Dietary concentrations of DON, 15-acetyl-DON, and ZEN are given in Table 1. Other mycotoxins were not detected at or above the detection limit, which was 0.02 mg/kg for aflatoxin, 2 mg/kg for fumonisins, and 0.2 mg/kg for the remaining mycotoxins analyzed.
BW and Feed Consumption
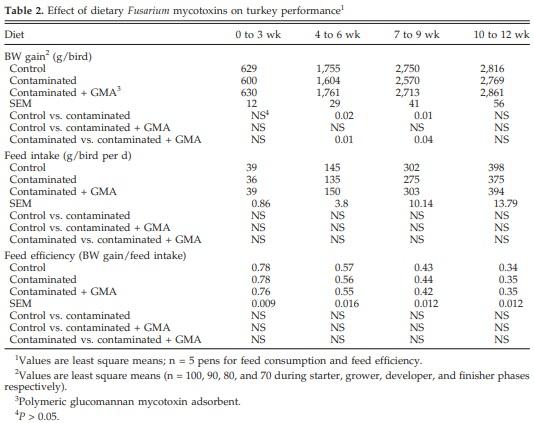
Feeding contaminated grains significantly (P < 0.05) decreased BW gains during the grower and developer phases, and GMA supplementation prevented these effects (Table 2). There was no effect of diet, however, on feed intake or feed efficiency.
Organ Weights
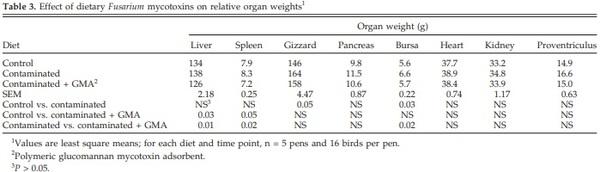
There was a significant (P < 0.05) increase in the relative weights of gizzard and bursa of Fabricius after feeding contaminated grains for 12 wk (Table 3). Dietary supplementation with GMA to contaminated diet prevented the effect on the bursa of Fabricius. Reductions in weights of liver and spleen were observed, however, in birds fed the GMA-supplemented diet (Table 3) compared with controls and birds fed the contaminated diet.
Hematology and Plasma Chemistry
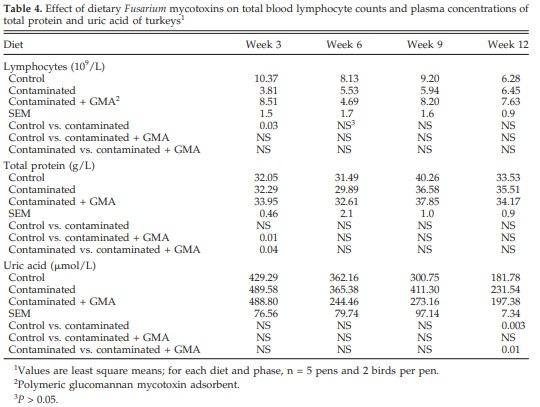
Hemoglobin concentrations, hematocrit, and MCHC were not significantly affected by the diet (data not shown). The feeding of contaminated grains reduced (P < 0.05) total lymphocyte counts at wk 3 (Table 4). There was no change, however, in total white cell and other differential leukocyte counts (data not shown). Supplementation with GMA increased plasma total protein concentrations compared with controls and birds fed the contaminated diet. Plasma uric acid concentrations in birds fed contaminated grains were increased at the end of the experiment compared with controls, and the feeding of GMA prevented this effect (Table 4). No significant effect of diet was observed for other plasma constituents including activities of enzymes (data not shown).
Phenotyping of Peripheral Blood Lymphocytes
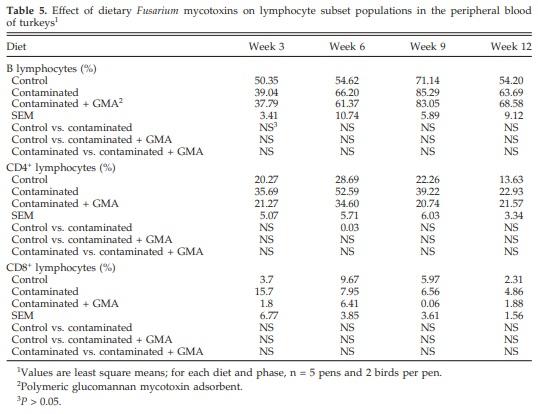
There was a significant (P < 0.05) increase in the percentage of CD4+ lymphocyte populations during wk 6 in birds fed contaminated grains. There was no change, however, in the percentage of CD8+ and B-lymphocyte populations (Table 5).
Lymphocyte Blastogenesis Assay
Mitogenic responsiveness of peripheral lymphocytes to Con A and PWM was not affected (P > 0.05) by diet (data not shown).
Serum and Biliary Ig
There was no significant effect of diet on serum IgA, IgM and IgG, and biliary IgA concentrations (data not shown).
Immune Responses to Antigens
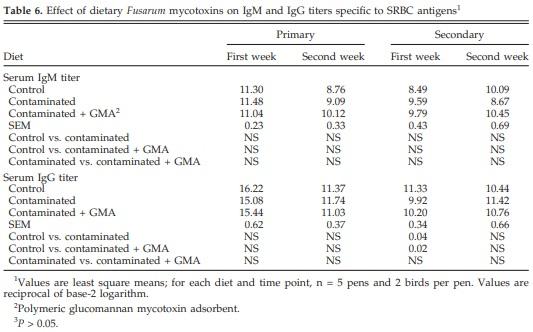
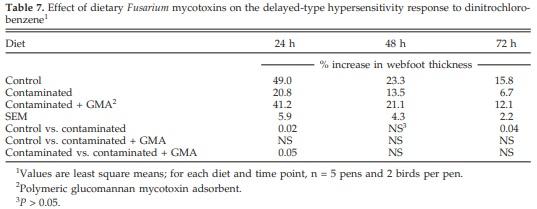
There was no significant effect of diet on IgM primary and secondary antibody titers to SRBC. Secondary IgG titer against SRBC antigens, however, was significantly decreased after feeding contaminated grains compared with controls (Table 6). The CHS response was significantly (P < 0.05) decreased after 24 and 72 h by feedborne mycotoxins compared with controls. Supplementation of the contaminated diet with GMA prevented the decrease in CHS response after 24 h (Table 7).
DISCUSSION
Dietary Mycotoxin Concentrations
Diets were formulated to achieve similar concentrations of mycotoxins at all growth phases by incorporating the same levels and sources of contaminated corn and wheat. Deoxynivalenol was the major contaminant in all contaminated diets, and concentrations were consistent at all growth phases (Table 1); however, it was not the same. It was found that 15-acetyl-DON and ZEN were present as minor contaminants. Toxicological interaction between DON and ZEN, however, has not been observed in swine (Cote et al., 1985) or mice (Forsell et al., 1986). The cytotoxicity of 15-acetyl-DON is similar to that of DON (Eriksen et al., 2004), and the toxicity of 15-acetylDON in the present study may be additive to that of DON. It has been reported that the same level of inclusion of contaminated grains resulted in 1.9 mg of DON/kg of feed in 1 experiment and 4.4 mg of DON/kg of feed in another experiment (Smith et al., 1997). Failure to achieve the same concentrations of DON in the present experimental diets may be attributable to lack of uniformity in occurrence of mycotoxins in contaminated corn and wheat (Hamilton, 1978) and problems associated with sampling, surveying, post-collection handling, and analysis of mycotoxin-contaminated grains (Davis et al., 1980).
It has also recently been shown that some of the Fusarium mycotoxins including DON and ZEN form conjugates with glucose, thereby escaping routine analytical detection procedures (Schneweis et al., 2002; Berthiller et al., 2005).
Deoxynivalenol concentrations of up to 1.2 mg/kg of feed (starter, grower, and developer) and 0.2 mg of ZEN/ kg during the developer phase were detected in the control diets, thereby indicating that control corn and wheat contained, nevertheless, detectable amounts of mycotoxins. Fusarium mycotoxin contamination of Ontario-grown grains is widespread, and it was not possible to find grains devoid of contamination. There is no evidence for DON toxicity in turkeys at the concentrations detected in control diets of the present study.
Production Parameters
Chowdhury (2005) observed reductions in BW gains in all the phases. There were no significant changes, however, in feed intake, but the feeding of grains contaminated with Fusarium mycotoxins reduced the efficiency of feed utilization in the grower and developer phases. Findings of the current study contrast with those of Chowdhury (2005) in that significant reduction in BW gains were seen only in the grower and developer phases. This may be attributable to the lower levels of mycotoxins in the contaminated diets in the current study. Chowdhury (2005) fed diets containing 6.8 to 13.6 mg of DON/ kg of feed, 0.6 to 1.3 mg of 15-acetyl-DON/kg of feed, and 0.4 to 0.7 mg of ZEN/kg of feed. The corresponding concentrations in the current study were 2.2 to 3.3 mg of DON/kg of feed, 0.2 mg of 15-acetyl-DON/kg of feed, and 0.2 to 0.3 mg of ZEN/kg of feed. In contrast to the present study, it was previously reported that turkeys were resistant to feeding 15 mg of semipurified DON/ kg of feed (McMillan et al., 1986) or 4.4 mg of DON/kg of feed containing contaminated corn (Manley et al., 1988) or 4.6 mg of DON/kg of feed containing contaminated wheat (Hamilton et al., 1985). Deoxynivalenol has been reported to cause adverse effects in poultry when fed in combination with other mycotoxins (Morris et al., 1999). The possible reasons for adverse effects on performance observed in the present study may be due to interaction between the Fusarium mycotoxins and the possible presence of unidentified mycotoxins, mycotoxin precursors, and metabolites in naturally contaminated grains.
Organ Weights
Previously, reports of feeding grains naturally contaminated with Fusarium mycotoxins to turkeys indicated that diet did not affect relative liver, spleen, kidney, or bursal weights (Chowdhury, 2005). Feeding contaminated grains in the present study, however, increased relative gizzard and reduced the bursa of Fabricius weights at wk 12. Increased weight of bursa of Fabricius after feeding Fusarium mycotoxins to broilers has been previously reported (Kubena et al., 1989). An increase in gizzard weight in the present study may be associated with irritation caused by Fusarium mycotoxins leading to inflammation and thickening of the mucosal layer (Kubena et al., 1997).
The feeding of contaminated diets did not significantly affect relative weights of liver, spleen, kidney, and testes of broiler breeders (Yegani et al., 2006). Chowdhury and Smith (2004) reported an increase in the relative weights of kidneys in laying hens fed contaminated grains and perhaps caused by elevation in blood uric acid concentrations. There was no effect of 5 mg of DON/kg of feed in broiler diets on absolute and relative weights of gizzard, pancreas, heart, spleen, colon, and cecum. Both absolute and relative weights of small intestine, however, were significantly decreased (Awad et al., 2006). Reports on the effects of Fusarium mycotoxins on organ weights of poultry are contradictory, and, hence, organ weights might not be good indicator of toxicity of some of the Fusarium mycotoxins. The nature of the mycotoxin challenge varies in these reports. Duration of toxin exposure may also be a significant factor, because the organ initially swells with toxin exposure followed by shrinkage (Swamy et al., 2004).
Hematology and Plasma Chemistry
Hematopoietic progenitor cells are susceptible to trichothecene toxicity (Parent-Massin, 2004). Hematoxicity of trichothecenes is characterized by hematological effects due to abnormal blood cell counts and dysfunction of blood cells of lymphoid and myeloid origins. Ingestion of trichothecenes causes a reduction in circulating blood cells frequently associated with bone marrow failure in horses, cattle, poultry, cats, mice, and guinea pigs (ParentMassin, 2004).
Feeding naturally contaminated grains did not affect (P > 0.05) hemoglobin, hematocrit, or MCHC in the present study. Total lymphocyte counts were significantly (P < 0.05) reduced at wk 3. Total white blood cell counts and other differential leukocyte counts, however, were not significantly affected by diet. Chowdhury et al. (2005c) observed transient changes in hemoglobin, hematocrit, and differential leukocyte counts including basophil and monocyte counts. Dietary effects were also seen in hemoglobin and hematocrit concentrations. It is noteworthy, however, that variation was within the normal range found in turkeys. Discrepancies in the effects of Fusarium mycotoxins on hematology in these 2 studies can be attributed to the different doses of mycotoxins fed. A reduction in total lymphocytes during the early growth phase in the absence of hematological parameters indicates the sensitivity of the immune system to such a challenge. This effect may be attributable to reduced proliferation of lymphocytes or direct lymphocyte cytotoxicity caused by Fusarium mycotoxins (Sharma, 1993). Consumption of low concentrations of mycotoxins may result in impaired immunity (Corrier, 1991).
Decreased T- or B-lymphocyte activity, suppressed antibody production, and impaired macrophage function are the clinical manifestations of mycotoxin-induced immunosuppression (Corrier, 1991). The vulnerability of the highly proliferating and differentiating cells of lymphoid origin that participate in immune-mediated activities indicates the sensitivity of the immune system to mycotoxin-induced immunosuppression (Oswald et al., 2005). Deoxynivalenol is the least myelotoxic of the trichothecenes (Parent-Massin, 2004). Deoxynivalenol was the major contaminant in the present study, and this may have resulted in few or minor dietary effects. Circulating blood cells may be less sensitive, although the hematological changes observed may be due to the myelotoxicity of trichothecenes (Parent-Massin, 2004). The effects of Fusarium mycotoxins on hematological indices, however, have been reported to be limited (Chowdhury et al., 2005c).
A significant increase in plasma uric acid concentrations compared with controls (Table 4) was observed after 12 wk of feeding contaminated grains. Swamy et al. (2002) also observed increases in serum uric acid concentrations in broilers exposed to a similar challenge. Laying hens have also been reported to exhibit elevations in plasma uric concentrations under similar circumstances (Chowdhury and Smith, 2004). It has also been shown that feeding contaminated grains reduced hepatic fractional synthesis rates in laying hens (Chowdhury and Smith, 2005). Inhibition of hepatic protein synthesis might have resulted in the elevation of hepatic free amino acid concentrations. The excess free amino acids could be used for energy utilization, resulting in increased plasma uric acid concentrations. Chowdhury (2005), however, reported that turkeys fed grains contaminated with Fusarium mycotoxins had significantly decreased plasma uric acid concentrations. The reason for this observation is not clearly understood.
Immunological Parameters
Macrophages, T cells, and B cells of the immune system are sensitive to effects of DON, which can be immunostimulatory or immunosuppressive depending on dose, exposure frequency, and timing of the functional immune assay (Pestka et al., 2004). Low exposure to DON upregulates expression of cytokines, chemokines, and inflammatory genes with concurrent immune stimulation. High exposure to DON promotes leukocyte apoptosis associated with immune suppression (Pestka et al., 2004).
Deoxynivalenol exposure at higher levels injures actively dividing cells in the bone marrow, lymph nodes, spleen, thymus, and intestinal mucosa. This results in immunosuppression (Ueno, 1984) as suggested by depression of circulating white blood cells, decreased resistance to pathogens (Listeria monocytogenes and Salmonella), inhibition of antibody responses to antigens, and impaired delayed-type hypersensitivity responses (Bondy and Pestka, 2000).
A significant increase in CD4+ subset populations at the end of wk 6 was observed. Chowdhury et al. (2005c) did not observe significant changes in lymphocyte subset populations. The increase in CD4+ subset populations in the current study, however, may be due the stimulation of these subsets. Pestka et al. (1990) observed an increase in the number of T cells in both Payer’s patches and spleen of mice fed 25 mg of DON/kg of feed for 12 wk. Repeated stimulation of T-cell receptors results in effectors with impaired function (Jelley-Gibbs et al., 2005), which may be associated with loss of functionality of the immune cells (Goldsby et al., 2000). Poapolathep et al. (2004), however, observed significant depletion of CD4+CD8+, CD4+CD8−, and CD4− CD8+ numbers after mouse thymocyte primary cultures were treated with nivalenol (0.25 to 1.0 μg/mL). Similar findings were reported including reduction in thymic and splenic T-lymphocyte subset populations after i.p. injection of the Isaria mycotoxin myriocin (1.0 mg/kg) to mice (Johnson et al., 2004).
Two mitogens were chosen in the present study so that proliferative response of T and B cells could be distinguished. Concanavalin A and PWM were mitogens selected to stimulate T- and B-lymphocyte proliferation, respectively. Blastogenic responses are nonspecific in nature, whereas CHS is an antigen-specific response. Lymphocyte blastogenic responses to mitogens, however, have been routinely used in immunotoxicity assays, because they provide reliable information about the proliferative ability of T and B lymphoctyes, which is an important event for most immune responses (Smialowicz, 1995). Both lymphoblastogenesis and CHS assays were performed in this present study. No significant changes were observed, however, in the stimulation indices of lymphocytes to Con A and PWM. Rotter et al. (1994) observed no significant effect of Fusarium mycotoxins on lymphocyte blastogenic responses in pigs. Harvey et al. (1991) reported a significant reduction in the stimulation response of splenocytes to phytohemagglutinin in 3-wk old broiler chickens fed 50 mg of purified DON/kg of feed. It has been recently shown that T-2 toxin (10 ng/ mL) depressed chicken lymphocyte proliferation in response to Con A and phytohemagglutinin M (Jaradat et al., 2006). This inconsistency of results in lymphoblastogenesis assays may be due to the ability of Fusarium mycotoxins to both upregulate and downregulate the lymphocyte counts and their ability to proliferate. In the current study, a diet containing comparatively low concentrations of Fusarium mycotoxins significantly reduced the CHS response at 24 and 72 h, in agreement with Chowdhury et al. (2005c).
The secondary IgG titers to SRBC antigens were significantly reduced in the present study when contaminated diets were fed. Total serum IgA, IgM, and IgG and biliary IgA, however, were not affected by diet. Specific antibody response to SRBC in pigs (Swamy et al., 2003) and turkeys (Chowdhury et al., 2005c) was not affected in these studies by feeding naturally contaminated grains. The feeding of contaminated grains did not affect concentrations of serum IgA (Swamy et al., 2002; Chowdhury et al. 2005c), biliary IgA (Swamy et al., 2004), or serum IgM and IgG (Swamy et al., 2002, 2004) in poultry. Significant changes in biliary IgA, however, were reported in broiler chickens (Swamy et al., 2002) and turkeys (Chowdhury et al., 2005c) after feeding naturally contaminated grains. In contrast to the apparent resistance of humoral competence to Fusarium mycotoxins in the current study, cell-mediated immune response was affected by consumption of Fusarium mycotoxins.
Anti-SRBC antibody response is mediated by CD4+ T cells (Goldsby et al., 2000), whereas anti-DNCB response is mediated by CD8+ T cells (Bour et al., 1995), and the present study results suggest that the cell-mediated immune response involving CD4+ and CD8+ T cells may be more sensitive to feedborne Fusarium mycotoxins. Reductions in antibody titers for Newcastle disease virus in broiler chickens (50 mg of purified DON/kg of feed; Harvey et al., 1991) and infectious bronchitis virus in broiler breeders (12.6 mg of naturally contaminated DON/kg of feed; Yegani et al., 2006) have been observed, and these are also CD4+ T cell-mediated responses. The present study indicates that cell-mediated immune responses are adversely affected by feedborne Fusarium mycotoxins even at low concentrations.
Efficacy of GMA
Several strategies to prevent mycotoxicoses in animals and poultry including physical, chemical, and biological have been investigated (Diaz and Smith, 2005). Glucomannan mycotoxin adsorbent has been shown to have beneficial effects in preventing adverse effects of Fusarium mycotoxins in turkeys (Chowdhury, 2005; Chowdhury et al., 2005c), broiler chickens (Swamy et al., 2002), laying hens (Chowdhury and Smith, 2004), and broiler breeders (Yegani et al., 2006). In the current study, GMA prevented many of the adverse effects caused by feeding Fusarium mycotoxins to turkeys. Interactions between mycotoxins and adsorbents in the intestinal lumen prevent the absorption of mycotoxins and transfer to target tissues (Ramos et al., 1996).
It can be concluded that turkeys are susceptible to feedborne Fusarium mycotoxins, as can be seen in reduced performance and immunosuppression. The feeding of grains naturally contaminated with Fusarium mycotoxins should be minimized.
ACKNOWLEDGMENTS
Financial support for this study was provided by the Ontario Ministry of Agriculture, Food and Rural Affairs and Alltech Inc. Nicholasville, Kentucky. We gratefully acknowledge the following members of the Department of Animal and Poultry Science of the University of Guelph: Margaret Quinton for the statistical advice and Mojtaba Yegani and Gabriel Diaz for their technical help.
This article was originally published in Poultry Science 87:421–432. doi:10.3382/ps.2007-00181. This is an Open Access article distributed under a Creative Commons license.