
Content sponsored by:
Phileo by Lesaffre
Actisaf® and Safmannan® help reduce microbial contamination in beef cattle carcasses
Published: April 27, 2020
By: Mohamed Mammeri, Global Product Manager, Phileo by Lesaffre

Each year, about one in 10 people worldwide become sick with foodborne illnesses, resulting in 420,000 deaths out of a total infected population of 600 million people. The WHO (World Health Organization) estimates that a total of 33 million years of healthy life are lost worldwide each year due to the consumption of contaminated foods. Diarrheal diseases are responsible for most of this global burden, causing 550 million cases of disease and 230,000 deaths (https://www.who.int/).
Among diarrheal diseases, Listeriosis and colibacillosis are the two most serious foodborne diseases in the world. Thus, reducing the rate of foodborne outbreaks caused by Escherichia coli (E. coli) and Listeria in humans remains a major concern worldwide.
Listeriosis is caused by the bacteria Listeria monocytogenes which can survive and multiply at very low temperatures, even at refrigerator levels.
Colibacillosis, which is due to E. coli infections, is an important cause of septicemia, urinary tract infections and meningitis in humans.
Animals are recognized as a reservoir of E. coli and Listeria, which is present in their intestinal tract, and is therefore a source of contamination for humans. Food, especially contaminated meat, is the main route for contamination passing to humans. The high mortality rates associated with E. coli and Listeria makes them both very important public health problems.
Limiting the prevalence of these bacteria in animals, while reducing as much as possible, the use of antibiotics, is a strategy which responds well to the ‘one health’ principle.
In this context, results of the trial show that supplementing beef cattle with Actisaf® and Safmannan® during the fattening period reduces the microbial load of pathogenic bacteria present in the feces. This is the main conclusion from a study carried out on an Italian beef farm in which the impact of using Actisaf® and Safmannan® as a feed supplement was assessed across 78 Charolais beef cattle.
During this scientific trial, animals were divided into two homogenous groups according to their origin, age and body weight:
In the control group (n=39) animals were fed with a normal diet.
In the supplemented group (n=39) animals were also fed with a normal diet but with an additional supplementation of 5g of Actisaf® and 10 g of Safmannan®, both products given per day and per animal.
In the supplemented group (n=39) animals were also fed with a normal diet but with an additional supplementation of 5g of Actisaf® and 10 g of Safmannan®, both products given per day and per animal.
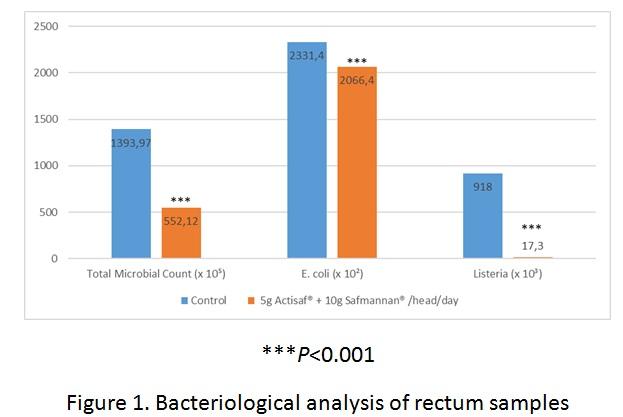
Rectum samples were subsequently collected from these animals at the slaughterhouse, placed in plastic containers and transported to a laboratory at 4°C for bacteriological analysis. The resulting analysis included recording levels of Total Microbial Count (TMC), E. coli and Listeria.
The results from this analysis, as represented in Figure 1, showed TMC, E. coli and Listeria counts being reduced by 60%, 11%, and 98% respectively in the Actisaf®+ Safmannan® group, compared to the control group.
This result could be explained by the pathogen-binding properties of Actisaf® and Safmannan® which limited the colonization of the gastrointestinal tract (Posadas et al., 2017).
It is therefore shown that supplementation with Actisaf® and Safmannan® reduces the bacterial load in the cecum samples from the slaughterhouse. This, in turn, reduces the bacterial contamination of the carcass by pathogenic bacteria, as is related to the potential transmission to humans through the consumption of meat. Therefore, Actisaf® and Safmannan® ensure better quality microbiological meat and provides better food safety.
Posadas, G.A., Broadway, P.R., Thornton, J.A., Carroll, J.A., Lawrence, A., Corley, J.R., Thompson, A., Donaldson, J.R., 2017. Yeast Pro- and Paraprobiotics Have the Capability to Bind Pathogenic Bacteria Associated with Animal Disease. Transl. Anim. Sci. 1, 60–68. https://doi.org/10.2527/tas2016.0007
Related topics:
Authors:

Influencers who recommended :
KUHN GeraldineRecommend
Comment
Share

Would you like to discuss another topic? Create a new post to engage with experts in the community.


