Breeding
In the U.S., vaccines are the most commonly used approach to eliminating Salmonella in breeder chickens. In the E.U., Denmark, Finland, The Netherlands, and Sweden have taken the approach that if a breeder flock is positive for Salmonella, they slaughter the flock. This is cost preclusive in the U.S. due to the scale of the industry. The E. U. in general use vaccination and/or competitive exclusion as a means of reducing Salmonella in breeders. In the U.S. undefined competitive exclusion cultures cannot be used. Defined cultures do not work well. Thus, no CE is regularly used in the U.S. The E.U. uses these cultures with good results.
Hatching
To reduce the prevalence of Salmonella on poultry carcasses during processing, a number of approaches have proven useful; however, to be truly successful at controlling Salmonella, intervention strategies should be implemented during the breeding, hatching, growout, and transportation phases of poultry production as well. Salmonella spp. may be found in the nest box of breeder chickens, cold egg-storage rooms at the farm, on the hatchery truck, or in the hatchery environment (Cox et al., 2000). These bacteria may then be spread to fertilized hatching eggs on the shell, or in some cases may penetrate the shell and reside just beneath the surface of the eggshell.
Research has demonstrated that contamination of raw poultry products with Salmonella spp. may be attributable to cross-contamination in the hatchery from Salmonella infected eggs or surfaces to uninfected baby chicks during the hatching process. Cox et al. (1990 and 1991) reported that broiler and breeder hatcheries were highly contaminated with Salmonella spp. Within the broiler hatchery, 71% of eggshell fragments, 80% of chick conveyor belts swabs, and 74% of pad samples placed under newly hatched chicks contained Salmonella spp. (Cox et al., 1990).
Cason et al. (1994) reported that, although fertile hatching eggs were contaminated with high levels of Salmonella typhimurium, they were still able to hatch. The authors stated that paratyphoid salmonellae do not cause adverse health affects to the developing and hatching chick. During the hatching process, Salmonella spp. is readily spread throughout the hatching cabinet due to rapid air movement by circulation fans. When eggs were inoculated with a marker strain of Salmonella during hatching, greater than 80% of the chicks in the trays above and below the inoculated eggs were contaminated (Cason et al., 1994). In an earlier study, Cason et al. (1993) demonstrated that salmonellae on the exterior of eggs or in eggshell membranes could be transmitted to baby chicks during pipping.
Salmonella may persist in hatchery environments for long periods of time. When chick fluff contaminated with Salmonella was held for 4 years at room temperature, up to 1,000,000 Salmonella cells per gram could be recovered from these samples (Muira et al., 1964).
Researchers have demonstrated a link between cross-contamination in the hatchery and contaminated carcasses during processing. Goren et al. (1988) isolated salmonellae from three different commercial hatcheries in Europe and reported that the same serotypes found in the hatcheries could be found on processed broiler chicken carcass skin. Thus, proper disinfection of the hatchery environment and fertile hatching eggs is essential for reducing Salmonella on ready-to-cook carcasses. Suggestions for elimination of Salmonella in the hatchery include:
1. Install a disinfectant fogging system or electrostatic spraying system in the hatchery plenum (Figure 1), setters, and hatchers (Figure 2) that are linked to a timer system.
2. Spray disinfectant every 30 minutes during setting and hatching to prevent cross-contamination.
3. Thoroughly clean and sanitize setters and hatchers regularly using documented sanitation standard operating procedures (SSOP´s).
4. Regularly monitor eggshell fragments, chick paper pads, and chick dander from the bottom of the hatching cabinet for Salmonella.
Photo 1: Electrostatic spraying system in the plenum of a hatchery.
Photo 2: Electrostatic spray application of sanitizer in a hatching cabinet.
Growout
The modern broiler chicken has been bred over the years to be a veritable "eating machine." During growout, broiler chickens eat approximately every four hours. Frequent eating is advantageous because birds that eat this frequently gain weight and put on edible muscle rapidly. This attribute may be considered a disadvantage for maintaining the sanitary quality of the bird during processing. At the end of the growout period, prior to catching the birds and cooping them for transportation to the processing plant, the feed is removed from the birds for a period of approximately 3 to 7 hours. During this time, birds become hungry and begin to search for food. Because there is no food available to them in the feeders, they begin to search for feed on the floor, which may be contaminated. This activity has been demonstrated to significantly contribute to the level of Salmonella on processed carcasses (Byrd et al., 2001). Studies have shown that many birds entering the processing plant have high levels of Salmonella
in their crops as a result of this litter pecking (Byrd et al., 2001) (Photo 3).
Photo 3: Fecal material in crop of chicken upon arrival at back dock of processing plant.
Salmonella in the crops of chickens that have consumed litter may be spread from carcass
to carcass during the crop removal process (Hargis et al., 1995, and Barnhart et al., 1999). During cropping, the cropper piston is ed into the vent area of the carcass and continues through the entire carcass, spinning as it goes (Photo 4).
Photo 4. Cropper
The piston has sharp grooves on the end of it that pick up the crop and wraps the crop around the end of the cropper piston (Photo 5). As the piston moves through the neck opening, the cropper piston comes in contact with a brush that removes the crop from the piston (Photo 6). Then the piston, while spinning, goes back through the entire carcass.
Photo 5: Cropper piston removing crop. Photo 6. Crop removal brush.
If the crop breaks during this removal process, the contents leak onto the cropper piston and are transferred to the interior and exterior of the carcass, possibly spreading Salmonella.
Studies have been conducted by Dr. Allen Byrd of the USDA - Agricultural Research Service (ARS) in which the crops of live birds were filled with fluorescein dye. After thirty minutes, the birds were processed. By examining the carcasses at different stages of processing under a black light, crop contents that were transferred to the inside or outside of the carcass could be clearly visualized. These studies have shown that commercial croppers result in a large amount of contamination of the inside and outside of the carcasses. Thus, efforts should be made to control Salmonella in the crop prior to the crop removal process.
Some companies have been successful at controlling Salmonella in the crop by acidifying the birds drinking water during the feed withdrawal process. Acetic, citric and lactic acids have all been used at low concentrations (0.3 to 0.5 %) to acidify the crop to the extent that Salmonella are unable to survive. Byrd et al. (2001) found that lactic acid was most effective and that 0.44% lactic acid in the waterers of broilers during the feed withdrawal period reduced Salmonella contaminated crops by 80 %. This effect carried over to the pre-chill carcasses on which the prevalence of Salmonella was reduced by 52.4 % (Byrd et al., 2001). When acidifying drinking water using lactic acid, it is best to gradually expose the birds to higher and higher levels of acid in the water the week before birds are to be caught. The key is to make the lactic acid concentration as high as possible while insuring that the birds continue drinking the water. Suggestions for elimination of Salmonella in the crop prior to processing are as follows:
1. Apply lactic acid to drinking water of the chickens before the feed withdrawal period.
2. Begin by applying small amounts and gradually increase levels until they reach 0.5 % (0.64 oz. of lactic acid/gallon of water).
3. Occasionally have the QA employees check the pH of the crops of birds at the plant to insure that they are being acidified.
Coop disinfection
Conventional cage-dump systems used in the poultry industry are very difficult to clean and sanitize (Photo 7).
Photo 7: Dirty cage-dump system.
If transportation coops are to be cleaned, they must be thoroughly washed and sanitized. Dry excreta should be removed before washing if possible. Some companies have implemented rinsing systems that do not thoroughly clean excreta off coops. This rehydrates the excreta, allowing Salmonella to proliferate. All of the environmental conditions (nutrients, pH, moisture, and temperature) that Salmonella require to multiply are available if the company simply rinses the coops. Thus, if disinfection systems are used, it is best to ensure that they are capable of thoroughly removing excreta prior to sanitizing the coops.
Sanitation
All poultry companies in the U.S. are required to operate using guidelines spelled out in their SSOP manual. These SSOP´s describe exactly how each piece of equipment and processing area (walls and floors) should be cleaned and sanitized. Prior to initiation of production, processing equipment surfaces should be evaluated to ensure that they have been properly sanitized. Equipment surfaces should be clean and free of processing residue. There is a tremendous amount of variation with regard to how different companies consider sanitation. Recently a company claimed that they "do not have time for sanitation". With the U.S. being a country of extreme litigation in cases of food-borne illness where negligence has been established, it is untenable that some companies take this approach.
Many poultry companies use old, outdated, and ineffective methods for determining sanitary status of equipment. Traditional swabbing methods are slow (48 hours), expensive, inaccurate (many of the bacteria on surfaces are psychrotrophic and are not detected using a traditional plate count), and are ineffective because inadequately cleaned surfaces cannot be detected prior to production. Equipment surfaces should be evaluated using a "real-time" contamination or bacterial detection system to determine their microbiological condition. Adenosine triphosphate (ATP) bioluminescence is a "real-time" monitoring procedure because an incubation period is not required and cleanliness can be assessed in one or two minutes. Many poultry companies are interested in this new technology because they can determine the effectiveness of their sanitation procedure immediately and make corrections such as re-cleaning and sanitizing before production begins, instead of to discard product that has been produced on inadequately sanitized processing lines. The ATP method has been made simple and inexpensive such that quality control or sanitation personnel can use it daily.
Moreover, the ATP technique fits into a Hazard Analysis and Critical Control Point (HACCP) scheme. Proper cleaning and sanitation of equipment can be considered a control point that is critical to the safety of food products. Because all HACCP programs require that the processor be able to control these critical control points, it would be impossible to use traditional microbiological methods for assessing the efficiency of sanitation, since 24 to 48 hours is not an acceptable time frame for identifying improperly cleaned equipment. The need for an immediate assessment of sanitation efficiency has increased the popularity of ATP bioluminescence.
One consideration with ATP bioluminescent assays is that, when sampling food contact surfaces in a poultry processing facility, the surfaces have often just been exposed to sanitizers. Sanitizers and cleansers are formulated to kill microorganisms and/or remove processing residues from food processing equipment or food contact surfaces. Many sanitizers and cleansers contain components that breakdown or destroy organic material such as fats, proteins, and biological membranes. Green et al. (1998 and 1999) demonstrated that exposing ATP bioluminescence reaction components directly to sanitizers and cleansers may significantly reduce ATP readings. Of the nine chemicals tested in the study, quaternary ammonia increased ATP readings (Green et al., 1999). Sodium hypochlorite (bleach) had no effect on ATP readings. Lactic acid at concentrations of 0.5 % and higher reduced readings by 75 % (Green et al., 1998). ATP measurements were significantly (P< 0.05) reduced by approximately 60 % when levels of trisodium phosphate exceeded 1 %. Hydrogen peroxide, at 1 %, significantly (P< 0.05) decreased ATP measurements by approximately 60 %. These results indicate that commercial sanitizers may negatively affect ATP readings if the sanitizer is allowed to come into direct contact with the ATP bioluminescence reagents (Green et al., 1998).
Water
Process water should be frequently evaluated to determine its characteristics. Incoming water should be evaluated for the following:
1. pH
2. Ammonia
3. Contaminants
4. Hardness
If the pH of the water is too high (>8.0), then bleach added to chlorinated rinse waters or chiller water will be ineffective, as the bleach will drive the pH up even further. At pH's above 8.0, chlorine is not found in its active form (hypochlorous acid) in high quantities and is ineffective for killing bacteria. If lye is used in the water reservoir that supplies the plant as a means of reducing the effects of acid rain, and the pipe that feeds the plant picks up this lye, then the pH of the water may be driven up to 10 or greater, causing problems with the chlorine as previously discussed. If ammonia has been added by the city to the incoming water, there is a greater likelihood that when chlorine is added to the water, some of the chlorine will form trichloramines. Trichloramines form noxious odors and even though the plant may be using very low levels of chlorine (< 20 ppm), the inspectors and employees may complain about the odor, causing the plant to have to reduce chlorine levels, resulting in inadequate bacterial reduction. Some companies using well water have complained about decreased shelf-life of their product. These companies should evaluate their well water to determine if bacterial levels are excessive. If the wells are contaminated, they need to be shock-treated with chlorine. Well and city water should be evaluated to be sure that no chemical or biological contaminant is present. Water hardness should be controlled because salts contained in hard water can make some disinfectants ineffective.
Ice
The ice supply in the plant should be evaluated. In one processing facility, water from the processing floor was allowed to run freely into the fresh ice supply, to be placed back on finished ready-to-cook carcasses. In another case, a fresh ice bin was placed under the corner of a drip pan that was collecting exudate under raw chickens on the processing line. The contaminated water from the drip pan was draining into the fresh ice to be used on product. In another instance, augers used to transport ice around the plant were located on the roof of the plant and were uncovered. Birds were able to roost on the edge of the augers and defecate in the ice. Fresh ice to be used on product should be carefully stored and transported to ensure that it does not become contaminated prior to being used on product. Hence, it is important to evaluate the following with regard to fresh ice:
1. Ice is transported using covered augers.
2. Ice containers are protected from contamination by covering them with plastic.
3. Ice house is protected from contamination by employees or other sources.
3. Ice chutes are evaluated occasionally for microbial contamination using psychrotrophic plate counts.
Air Supply
The air supply for the plant should be thoroughly evaluated to determine its source and whether there is positive or negative pressure inside the plant. In one processing facility, all of the air within the plant was flowing through the inside of a trash compactor. In another instance, the air being blown onto the live birds outside to keep them cool was coming directly into the plant and onto finished ready-to-cook carcasses. In another plant, the wastewater facility was only 30 yards from the door of the processing room and the plant had negative air pressure. All of these conditions may greatly contribute to contamination. Abu-Ruwaida et al. (1994) demonstrated that Salmonella spp. were frequently isolated from air samples where live birds carrying the organisms were handled.
It is important to go to each plant exit doorway, crack it slightly, and try to determine if the air is going into or out of the processing plant. It should be going out. If it is flowing into the plant, then the plant has negative pressure and it needs to have more clean make-up air pumped into the plant. Otherwise, all contaminants outside will be pulled into the plant. Some strategies for ensuring that the air supply is clean are:
1. Evaluate the environmental air make-up to ensure that it is clean.
2. Determine if each room has positive or negative air pressure and strive for positive pressure.
3. Evaluate the air supply for compressors that feed air to areas such as the chiller for bacterial contaminants.
4. Evaluate total air volume coming into the plant and make sure the temperature of the air is controlled, even during the summer months.
Employee hygiene
Employee hygiene is also an important part of keeping contamination down in the plant. Some poultry processing plants employ individuals that have cattle farms. These employees have cattle in their yards at home and are allowed to wear their street clothes and boots into the plant. Any Salmonella on their clothes or boots from the cows may be transmitted to the product. All plants should be equipped with mandatory handwash/sanitizing stations that are refreshed frequently. There should be no access to restrooms directly from the processing floor. Each employee should begin their shift with a clean, long smock, hairnet, and gloves (Photo 8).
Employees should be examined daily for illness. Visibly sick employees should be sent home. Foreign employees returning from their home country should be interviewed briefly to determine if they have had any food-borne illnesses. They may be asymptomatic carriers similar to Typhoid Mary who was infected with typhoid fever in 1900. She worked as a cook and spread the disease to 22 people between 1900 and 1907, causing 1 death. Later, she became a cook at a hospital and 25 more people became infected and two people died. Thus, these individuals may be carrying Salmonella or Shigella and not be aware of the risk they pose to the consumer. Only 10 cells of Shigella are required to cause a severe, life-threatening food-borne infection.
Photo 8: Employees wearing proper clothing.
Strategies for controlling the hygiene of personnel are:
1. Personnel flow should be controlled (i.e. they should not go from the kill room to the deboning area or from a cattle farm into the processing plant).
2. Personal hygiene should be controlled using written standard operating procedures that are spelled out for each area and monitored by managers daily.
3. Managers should evaluate employee health daily.
4. Managers should question employees returning from foreign countries about food-borne illness.
5. Employees must be held accountable for their failures to use hygienic practices.
6. Managers should train employees regularly concerning proper hygiene.
Scalder
As the weather begins to warm, a number of poultry companies will begin to experience problems keeping the birds cool in the growout house. As a result, some growers will use foggers to wet the birds and keep them cool. In some cases, these fogging systems wet the birds, allowing litter to attach to feathers. This may result in a serious problem once the birds arrive at the processing plant, because the litter on their feathers and skin is loosened during scalding and comes off of the birds, resulting in excessive organic material in the scald water. Scalder water containing high concentrations of fecal material is a problem because it comes in contact with the external surface of the birds and, during picking, bacteria contained in this dirty water may be massaged into the skin and open feather follicles.
To reduce this problem, some companies have installed a bird brush and washer prior to scalding (Photos 9 and 10). Larger brushes and chlorinated water physically remove the feces from the feathers and skin of the birds. One company using this technique decreased the amount of fecal material going into the scalder by approximately 90%. This decreases the amount of organic material on the surface of carcasses as they go into the chiller.
Photo 9. Pre-scald bird brush.
Photo 10. Pre-scald bird brush.
The scalder is one of the most important areas in the processing plant in which cross-contamination with Salmonella occurs (Okrend et al., 1986). The water in most scalders in the U.S. does not move against the carcasses, going from the exit of the scalder toward the entrance (counter-current) and contains high levels of excreta (Photos 11, 12, and 13). This opposing water flow is essential to wash the birds and remove contamination from the birds as they travel through the scalder. Counter-current flow may be accomplished by adding a steel barrier between the lines of chickens going in either direction. By separating these chickens, bacteria that are washed off of the external surface of the chickens entering the scalder are not transferred to chickens that are exiting the scalder. The rate of water flow should be high, so as to dilute the concentration of foreign material and bacteria in the scalder. There is a common adage that goes “dilution is the solution to pollution” and it applies in this case. Plants that are not equipped with multi-stage scalders should attempt to make their scalders multi-stage.
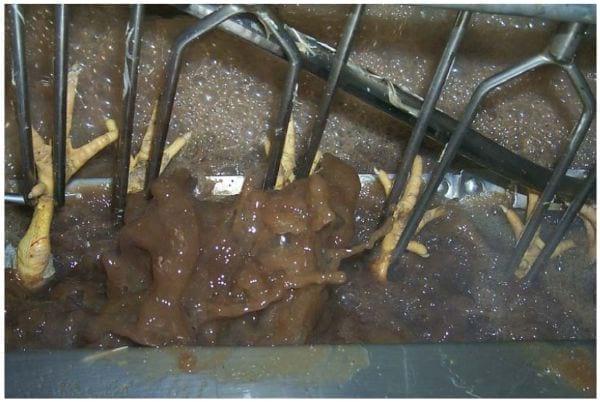
Photo 11: Scald water containing a high concentration of excreta.
Photo 12. Scald water that is not flowing and acts as a bath.
Photo 13. Scald water and lets of water at exit are clean and low in organic material.
Some companies have found that their scalders are not long enough to thoroughly rinse caked material from the outside of the birds. These companies have added sections to their scalders to increase length. In addition, the temperature of the scalder should be maintained as high as possible without causing visible defects to finished carcasses, such as breast striping. The water where the birds exit the scalder should be fairly clean. One company installed a water recycling system that takes all rinse water from equipment and carcass rinsers in the plant and recycles the water using diatomaceous earth filters. The water is then ozonated, heated and returned to the scalder. In one processing plant, this system has had an enormous impact on Salmonella prevalence on finished product. Suggestions to ensure that scalders are operating optimally with regard to decreasing cross-contamination include:
1. Make sure scald water flows in the opposite direction as the birds (counter-current).
2. Put as much fresh water into the scalder as possible to dilute Salmonella concentrations (Photo 11).
3. Keep scalder water temperature as high as possible without causing breast striping.
Feed withdrawal
Prior to evisceration, carcasses should be evaluated to determine if the birds have undergone proper feed withdrawal. By examining the abdominal cavity to see if it is concave (small amount of feces in the intestines) or convex (large amount of feces in the intestines), it is possible to determine if the birds have been withdrawn from feed long enough.
Moreover, the intestinal tracts on the intestines hanging from the birds after evisceration should be flat (Photo 14) and not full of digesta or bloated with gas (Northcutt et al., 1997). In the processing plant, birds held off feed for extended periods may exhibit a higher incidence of contamination with pathogens as the result of cross-contamination from bird to bird during transport due to loose pings. These birds may have intestines that are distended with gas which, if nicked during evisceration, may explode and disperse contents onto the carcass, other carcasses, or processing machinery. Extended periods of feed withdrawal also cause the tensile strength of the intestines to become weak (Northcutt et al., 1997). Weakness increases the propensity for them to be torn during evisceration.
Photo 14: Flat intestines indicate proper feed withdrawal time.
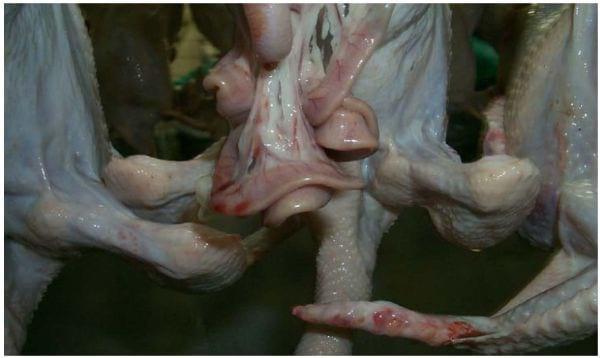
If birds are not held off feed long enough (< 8 hours), the intestines will be full of digesta (Northcutt et al., 1997). If full intestines are nicked during evisceration, it is likely that contents will be spread to the inside or outside of the carcass, to other carcasses, and to processing equipment. Also, if pressure is applied to the outside of a bird with full intestines, the contents may come out of the vent and spread onto the carcass. Immediately after venting, if the colons are full of material, then the contents will leak onto the carcass, especially if any line jerking or swinging occurs (Photo 15). Insufficient feed withdrawal time is perhaps the most important factor in meeting the zero tolerance standard for contamination on carcasses entering the chiller. Reprocessing levels as high as 75% and line speeds as low as 20 birds/minute have been reported in plants due to excessive contamination as a result of insufficient feed withdrawal times.
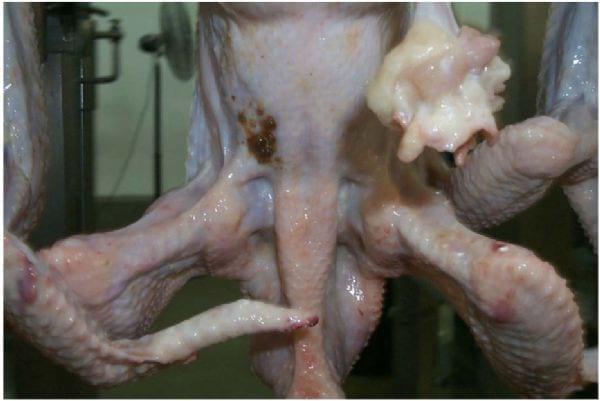
Photo 15: Fecal contamination due to feces leaking from colon. Line swinging caused feces to be spread to the back of the carcass.
Excessive contamination on incoming birds causes the water in the scalder to become dirty. This water may then be driven into the feather follicle during picking (Photo 16).
Photo 16: Broiler picking system.
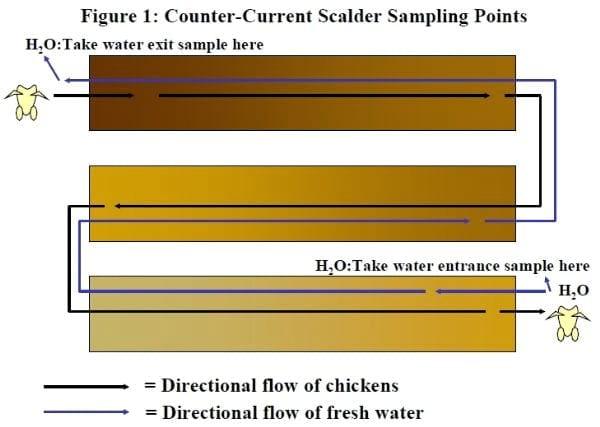
An ideal scalder set-up is depicted in Figure 1.
By introducing plenty of fresh water into the scalder (at the exit end), a significant portion of the organic material can be removed from the surfaces of the carcass. If this material is allowed to remain on the carcass, it will be transferred into the chiller. If the chiller contains high levels of organic material, then oxidative sanitizers, such as chlorine, will have little effect on bacterial concentrations. Thus, maintaining proper flow direction and water flow rate should increase the efficacy of chlorine as it is used later on in the process to kill bacteria.
Controlling Salmonella cross-contamination in the scalder
Scalding is one of the most important processing steps with regard to controlling the prevalence of Salmonella on processed ready-to-cook carcasses. The scalder is the first area in the plant where pathogenic bacteria associated with the surface of one bird can be washed free from that bird and be spread to the surfaces of other birds. This situation may lead to increases in overall prevalence of Salmonella on carcasses. Interestingly, the actual number of Salmonella on positive carcasses decrease as bacteria encased in excreta are washed free from the surface of the bird. However, because Salmonella negative birds may then become contaminated, the overall prevalence increases.
Some companies that use multi-stage scalders have begun to decrease the temperature in the first scalder tank from the normal temperature range of 53.3 to 55.6°C to 37.8-43.3°C. This should not be done because excreta, as it comes off of the surface of the birds, will sink to the bottom of the scalder tank. As the shift progresses, more and more excreta may build up in the bottom of the scalder. If the temperature of the scalder is 37.8-43.3°C, and there are any Salmonella in the excreta at the bottom of the tank, the Salmonella will begin to multiply rapidly. Essentially, at 37.8°C, the processor is operating the world''''s most expensive Salmonella incubator. In the scalder tank at low temperature, the Salmonella cells have all of the components they need to multiply rapidly. They have the optimum growth temperature, moisture, nutrients, pH, etc. Thus, the processor will essentially be inoculating bird after bird with Salmonella as they go through the scalder.
If the surface of the carcass is contaminated with Salmonella in the scalder as a result of the bacteria being transferred from bird to bird, another problem may occur in the next processing step. In the picker, feathers are removed and the bacteria in the contaminated water from the scalder may be transferred from bird to bird.
Testing the scalder efficacy
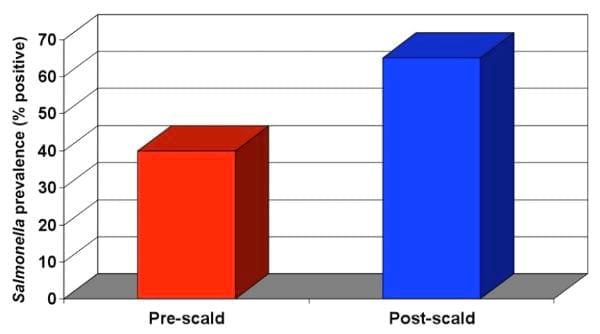
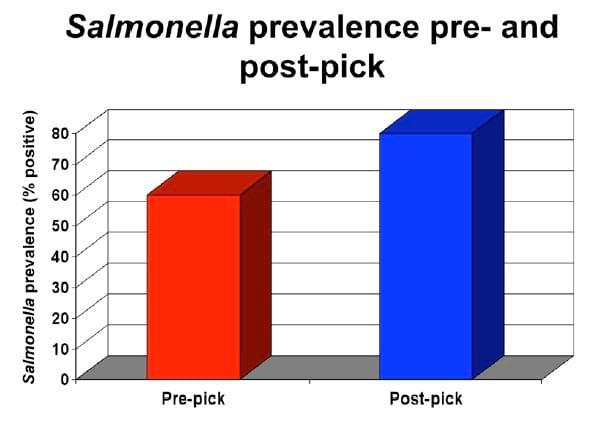
By evaluating the microbiological counts or Salmonella prevalence pre- and post-scald, it is possible to determine if the scalder is operating appropriately. Figure 3 below is an excellent example of an improperly operating scalder. Reasons for this problem may be the following: 1) the temperature of the scalder is far too low to prevent Salmonella from growing, 2) not enough fresh make-up water is being added to the scalder, or 3) the water is flowing in the same direction as the carcasses as opposed to a counter-current flow.
Figure 2. Salmonella prevalence increase as a result of scalding.
Some suggestions for correction of this problem are as follows:
1. Balance the scalders in terms of fresh water makeup and flow direction (counter-current)
2. Make sure that the temperature is above 50.6ºC in all scald tanks
3. Add a post-bleed brush system to remove any clumps of fecal material from the carcasses prior to scalding
4. Consider introduction of an approved chemical into the scald tanks
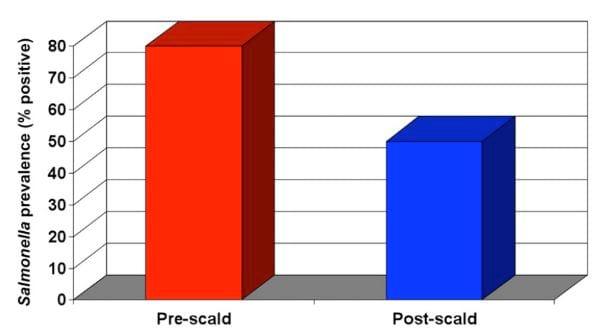
Once these corrections have been made, conduct additional microbiological evaluations. Figure 3 shows a more realistic scenario in a plant that is running the scalder correctly.
Figure 3. Scalder is operating correctly and reducing Salmonella prevalence.
The scalder should be considered a positive intervention step in controlling Salmonella. If biomapping of the plant indicates that the scalder is significantly contributing to cross-contamination of Salmonella between carcasses, then steps should be taken to correct the problem.
Addition of chemicals to the scalder and lowering temperatures
In a survey conducted by the U.S. Poultry and Egg Association at http://www.fsis.usda.gov/PDF/Slides_022406_EKrushinskie.pdf indicated that, of the poultry companies that add anything to the scalders (which very few do), 50% use chlorine and 50% use sodium hypochlorite. Chlorine should not be used in scalders because it is immediately deactivated by the organic load in the scalder and can gas off.
Sodium hypochlorite does not significantly impact the bacterial levels on carcasses during scalding.
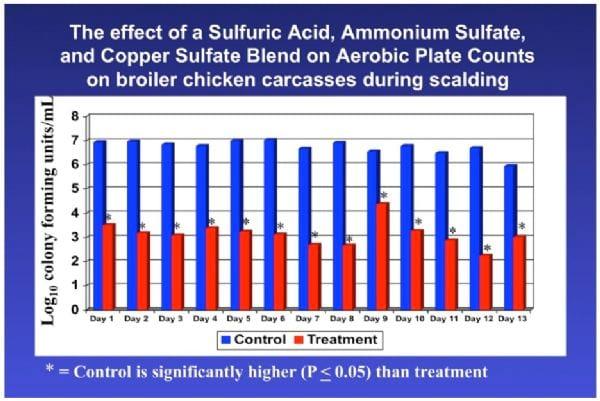
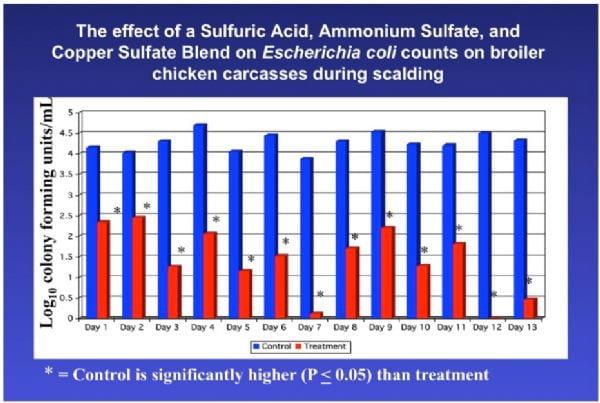
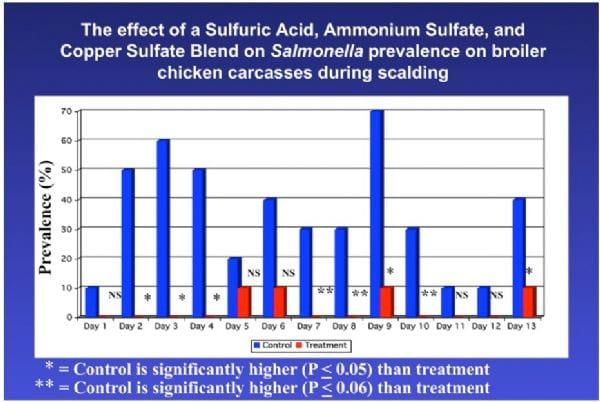
A benefit to adding acidic disinfectant chemicals to the scalder is that the scalder temperature may then be lowered. This has a tremendous impact on the efficacy and expense of scalding. Figures 4, 5, and 6 indicate APC and E. coli counts, and Salmonella prevalence on carcasses before and after scalding for controls (scalded in water only) and for carcasses scalded using sulfuric acid, ammonium sulfate, and copper sulfate as a carcass disinfectant.
Figure 4. APC counts on carcasses as a result of treatment during scalding with an acidic sanitizer.
Figure 5. E. coli counts on carcasses as a result of treatment during scalding with an acidic sanitizer.
Figure 6. Salmonella prevalence on carcasses as a result of treatment during scalding with an acidic sanitizer.
Using a chemical sanitizer in the scalder in these studies had a dramatic impact on Aerobic Plate and E. coli counts and Salmonella prevalence on chicken carcasses. Using a sanitizer, the scalder may be used to prevent growth of Salmonella in the scalder and this allows for the use of lower scalder temperatures that may have the following benefits:
1. Easier pick (acid sanitizers)
2. Less bacterial growth and lower bacterial numbers including Salmonella
3. Less overscald striping of breasts
4. Less energy cost to heat the scalder water
5. Less sub-skin fat cook-off leading to higher yields
Controlling cross-contamination in the pickers
The pickers present a serious problem for processors when it comes to cross-contamination. Fecal material is removed from a carcass by the mechanical action of the picker fingers. Subsequent carcasses are then exposed to this fecal material as they are rubbed by the contaminated picker fingers. Efforts have been made to control this problem by adding antimicrobial compounds to the rubber picker fingers. However, there has been little evidence indicating the efficacy of this approach. When conducting in-plant trials, we discovered that the chlorinated water coming from the bottom of the pickers contained around 106 (1 million) bacteria per milliliter. This indicates a very high level of contamination within the pickers, even when chlorine was added to the water at 40 ppm.
The USDA-FSIS has conducted biomapping studies on plants throughout the U.S. and found that in most plants, Salmonella increases in prevalence as the carcasses traverse the pickers. Thus, there are tremendous opportunities for the use of disinfectants during picking to assist processors in lowering total Salmonella prevalence on finished carcasses. Figure 7 shows how Salmonella prevalence may be increased during picking.
Figure 7. Typical increase in Salmonella prevalence during picking.
Great caution should be used when attempting to use chemicals in the scalder. Oxidants should not be used in the scalder because they bind readily to organic material and are deactivated before they are able to interact with bacteria. Additionally, oxidants generally gas-off under higher temperatures, causing employee health problems. Acidic chemicals or combinations may prove to be effective, however, their corrosivity to equipment should be determined prior to use and may preclude their use. Use of sodium hydroxide has not significantly improved bacterial levels as bacteria tend to be more resistant to increases in pH as opposed to decreases in pH (acids). Wastewater systems should be monitored closely when using chemicals in these systems as they may impact the ability of the plant to treat the wastestream and, if the chemicals have a long residual effect, they may kill the bacterial populations in the treatment ponds.
One major problem associated with the use of chemicals in the scalder and pick systems is that the poultry industry is not accustomed to paying for chemicals in the front of the plant. Thus, many companies, when presented with the additional cost of treatment, which they currently are not paying, balk at the idea of spending the extra money. With the new Salmonella regulations in place, the industry may begin implementing chemical hurdles at the front of the plant, even though they are resistant because of the cost. The cost of treating the scalder and pickers is generally much lower than interventions currently used in the field and it is proving to be far more effective overall.
Chemical disinfection on the processing line
There are three main methods by which poultry processors can reduce pathogenic bacteria on the processing line, excluding the scalder, pickers, chilling, and post-chill dip systems. These methods include: 1) equipment sprays, 2) carcass rinses or washes, and 3) the online reprocessing system (OLR). Each of these processes will be discussed.
Equipment sprayers
The purpose of equipment sprayers is to disinfect the part of the processing equipment that comes into contact with each carcass to prevent pathogens from coming off of one carcass and being transferred to another carcass, resulting in cross-contamination. Generally, high pressure spray nozzles are used in these applications and by far the most commonly used chemical is chlorine because of its cost. Other chemicals that are being used for these applications include chlorine dioxide, peracetic acid, and Zentox or TOMCO water (acidified hypochlorous acid). While chlorine is an excellent sanitizer, there are some general principles that must be taken into account when using it. The incoming water pH should be less than 6.5 after addition of the sodium hypochlorite bleach in order for the chlorine to do its job. This may be accomplished by the addition of citric acid or carbon dioxide to the water.
The main inhibitor to chlorine being effective as an equipment disinfectant is the buildup of fat or other organic material on the equipment, such that even though chlorinated water is being sprayed onto the built up material, it is not able to penetrate the material and kill the bacteria underneath. These bacteria may then be liberated when the next carcass comes by and be transferred to that carcass. This is a common occurrence on carcass brushes intended to remove feces after evisceration. Often, even though chlorinated sprays are used, these systems end up causing an increase in Salmonella prevalence when biomapping is done on carcasses before and after the brush or fecal finger system. The cropper can be especially troublesome in that the ingesta in the crops of birds often contain pathogens such as Salmonella and Campylobacter.
The pH of the equipment rinse waters should be maintained below 6.5 and checked regularly to ensure that the chlorine is in the appropriate form for optimal activity against bacteria. The spray pressure should also be checked and any clogged nozzles should be replaced. Photo 17 shows a spray bar in which the nozzles are set differently and some of the nozzles are not working.
Photo 17. An improperly operating spray bar.
Moreover, the nozzles and bars should be positioned so that they spray the part of the equipment that touches the chicken carcass and not other parts. Used properly, these systems can help to reduce cross-contamination.
Carcass sprays and inside/outside bird washers
The water from these sprayers or rinses should be checked frequently to determine chlorine levels or other chemical levels, pH, pressure and distribution. In one processing facility, the IOBW’s had very little very little water pressure. When confronted, the maintenance manager said, “Oh, do you want this turned up? I had it turned down so that it wouldn’t spray people as they walked by.” This is a case of someone changing the process without understanding the microbiological effect on the carcass. In another instance, a company’s E. coli results became unacceptable within one day and continued that way. The maintenance manager had swapped the nozzles in the IOBW for a different type of low flow nozzle. This had a dramatic influence on bacterial levels.
General suggestions for all washers and rinsers include:
1. Maintain proper nozzle pressure.
2. Maintain proper water pH.
3. Maintain proper chlorine or other chemical level.
4. Maintain proper water distribution on the carcass.
As with equipment rinses, chlorine dioxide, peracetic acid, and Zentox or TOMCO water (acidified hypochlorous acid) are being used in IOBW’s on an experimental basis.
Companies generally use chlorine in IOBW systems; however, a comprehensive research study conducted by Northcutt et al. (2005) of the USDA-ARS clearly demonstrated that adding chlorine to the IOBW has absolutely no impact on aerobic plate counts, Escherichia coli counts, Salmonella prevalence or Campylobacter counts on carcasses. These data correlate well with numerous biomapping studies that show no difference in the microbiological quality of carcasses entering versus those exiting the IOBW systems. This may be explained by the high level of organic material on the carcass at that point in the process. Chlorine is not able to penetrate the organic material and interact with the bacteria. Therefore, little or no effect is observed.
Online reprocessing systems
The purpose of OLR systems is not to reduce Salmonella. The USDA-Food Safety and Inspection Service (FSIS) views the OLR as a process intended to make carcasses that would otherwise have to be reprocessed by hand because they have fecal material or ingesta on them microbiologically equivalent to those carcasses that do not have any fecal material or ingesta on them. This is, in fact, the type of research protocol that companies must run to achieve approval for their chemical as an OLR agent (i.e. they must compare the microbiological quality of fecally contaminated to uncontamined carcasses). That been said, most processors expect to achieve at least a 1 log10 reduction in bacterial levels on carcasses as they traverse the OLR system.
The U.S. Poultry and Egg Association conducted a survey of the poultry industry in February of 2006 and the presentation of those data given by Dr. John Rice may be found at the following web address: http://www.fsis.usda.gov/PDF/Slides_022406_EKrushinskie.pdf .
The following are chemicals that the poultry industry (94 plants responded to the survey out of approximately 247 in the U.S.) is using for OLR purposes and the percentage of companies that use that particular chemistry: 1) acidified sodium chlorite (Sanova® - 33%), trisodium phosphate (Rhodia – 24%), chlorine dioxide (numerous companies – 15%), hypochlorous acid (Zentox and TOMCO – 9%), organic acids (6%), peracetic acid (FMC 323 or Parasafe and Inspexx 100 - 5%), cetylpyridinium chloride (Safefoods Cecure® - 3%), SynerX® (citric acid and HCL – 1%), bromine (BromitizeTM – 1%), sodium metasilicate (AvguardXP® - 1%), and electrolyzed oxidative water (EAU – 1%).
Chemicals not mentioned in the survey include Zentox monochloramine and SteriFx (FreshFx) which was included with organic acids, but contains mostly inorganic acid.
Sanova® (Ecolab) acidulated sodium chlorite
This product is approved as a poultry spray or dip at 500 to 1200 ppm singly or in combination with other GRAS acids to achieve a pH of 2.3 to 2.9 as an automated reprocessing method. In chiller water, sodium chlorite is limited to 50 to 150 ppm singly or in combination with other GRAS acids to achieve a pH of 2.8 to 3.2. Studies have shown that it can reduce Salmonella contamination from 31.6 % prevalence to 10 % prevalence (Kemp et al., 2001). In the survey by the U.S.P.E.A. listed above, the industry mentioned that they had no confidence in Sanova® or Inspexx® as OLR or chiller agents. However, they said that Sanova was an effective post-chill dip solution.
Trisodium phosphate (TSP)
Use of trisodium phosphate (TSP) over the years has been encouraged by the USDA as an approved method for automated reprocessing. TSP is costly to use because of the high concentration (10 %) used on carcasses (Photo 18). There are negative aspects to using TSP in poultry processing plants that should be considered. Residual TSP on carcasses causes the chiller water pH to increase dramatically. In plants where TSP is used, the chiller water will generally be in the pH range of 9.7 to 10.5. This is extremely high and prevents chlorine from being converted to its effective form, hypochlorous acid. Hypochlorous acid forms most effectively when water is in the pH range of 6.5 to 7.5. Thus, plants using TSP are wasting their bleach. This is not a desired situation because chlorine is very effective against Salmonella.
Photo 18: Trisodium phosphate system (TSP).
If a poultry company is trouble with high Salmonella prevalence and have an operating TSP system in place, it must make major adjustments to reduce Salmonella prevalence. CO2 gas systems have been added to the aeration systems of chillers as a means of reducing the pH of the water so that when chlorine is added, it will form hypochlorous acid. Discharging TSP in areas of the country that have strict phosphorous discharge limitations may be a problem as well. One beneficial effect of using TSP is that companies report that a 1% yield increase may be achieved due to increased water holding capacity.
Chlorine dioxide
Chlorine dioxide has had a rocky past within the poultry industry. Early attempts to introduce this chemical were unsuccessful because of the inability to control the levels of ClO2 during use. Gassing off occurred frequently and employees complained. ClO2 is an extremely effective sanitizer. The companies that are most successful with this chemical produce the chemical on site and control it very carefully. Adequate ventilation is necessary to ensure worker safety. In an in-plant trial conducted recently, I found no observable reduction in APC or E. coli on carcasses as they traversed an OLR using ClO2. This may be related to the relatively short contact time used in this plant and should not be used to evaluate all ClO2 used in all OLR applications.
Hypochlorous acid
The success of this technology varies greatly as well. Companies have seen excellent to no positive results depending greatly on the organic loading of the carcasses entering the system. In systems that adequately clean the carcasses prior to introduction to the OLR system, the bacteria may be greatly reduced using this approach. However, carcasses that have high organic loading in plants that use very few carcass washes and little water, may not achieve success using HOCl. Another key to this approach is using precise control of the pH of the water. HOCl will not be formed in significant amounts above a pH of 6.5 to 7.0, depending on the temperature.
Organic or inorganic acids
Acids definitely kill bacteria, however, they must be closely monitored to ensure that they contact the skin of the carcasses for an appropriate period of time and that they do not create product defects. Acid needs more time to kill bacteria than oxidant based chemicals and if the chiller immediately (within 2 min) follows the OLR system, it can be difficult to achieve good results. Often, bacteria become acid stressed when a carcass is treated with an acid. These organisms become hard to recover when doing efficacy studies. This does not mean that the bacteria were killed and will not be discovered by the USDA. Thus, when using acids, make sure that adequate neutralization and recovery steps are used during microbiological analysis or inaccurate results will be obtained.
Peracetic acid
Peracetic acid is a mixture of an organic acid (acetic acid) and an oxidant (hydrogen peroxide). Therefore, this chemical kills bacteria in two separate ways. Most research indicates that a 1 log10 reduction may be achieved using approximately 200 ppm peracetic acid in the OLR system. A precaution when using peracetic acid is that it may react with blood vessels, producing a slightly gray color on the skin of the carcasses in areas that are highly vascularized, such as the neck.
Cetylpyridinium chloride
Cetylpyridinium chloride is a relatively newly approved OLR methodology. It is effective and I have conducted studies that have demonstrated an 83% reduction in Salmonella on carcasses traversing this system. It is a very effective chemical.
Electrolyzed oxidative water (EO)
The machine used to generate EO water is expensive; however, the cost of the raw materials is very low (salt and water). Thus, the total cost is reasonable. Studies have shown that it is very effective and can achieve a 1 log10 reduction in bacteria on carcasses while being completely safe to use. EO water is acidified (pH 1.9 to 2.4) oxidative water that contains some hypochlorous acid (50 ppm) and other antimicrobial ions. It is generated on site, stored, and used as generated. It is not diluted. This material is excellent for post-chill dip applications as well.
Monochloramine
Monochloramine (Zentox) has many of the advantages of chlorine without the negative aspects. Monochloramine is used in a similar fashion as chlorine at about 50 ppm. It is generated by mixing bleach and ammonia under controlled conditions. It kills bacteria but is resistant to deactivation by organic material. Thus, it is more stable under high organic loads. Likewise, Axtell et al. (2005) demonstrated that no carcinogenic compounds were formed when monochloramine was used to chill poultry carcasses. This is a major concern in Europe with regard to the use of chlorine.
Unfortunately, there are still no magic bullets for use with OLR systems. The short contact time and methods of application (generally a spray) make it very difficult for chemicals to eliminate Salmonella during this step. We have observed reductions in Salmonella prevalence of 0% to 90% using various chemistries. It is also possible to see this type of variation from plant to plant using an individual chemical. This is because plants and incoming loads vary tremendously from plant to plant. Thus, it is important to the appropriate type of sanitizer based on an individual plant setup.
Disinfection during chilling
More bacterial reduction (both numbers and prevalence) can be accomplished in a properly balanced chiller than anywhere else in the processing plant. Most studies demonstrate that the chiller can significantly reduce Salmonella prevalence (Izat et al., 1989) if operating properly. As with the scalder, the pH, temperature, flow rate, flow direction, chlorine concentration, and concentration of organic material (digesta, fat, blood) is crucial in order for the chlorine in the chiller to do its job. The pH should be 6.5 to 7.5, the temperature should be below 40ºF, the flow rate should be high (at least one gallon per bird), and the flow direction should be counter-current. The most effective methods for controlling the pH of chill water include addition of carbon dioxide gas (90% of the industry uses this method according to the U.S. Poultry and Egg Association survey at http://www.fsis.usda.gov/PDF/Slides_022406_EKrushinskie.pdf) to the tubes normally used for air agitation, the addition of citric acid (10% of the industry uses this according to the survey) or sodium acid sulfate to the water.
The organic material in the chiller is generally determined by the following factors: the flow rate (amount of fresh make-up water), flow direction (should be counter-current), the use of pre-scald bird brushes and the cleanliness of the scalder, the temperature of the scalder, the number of high pressure carcass sprays used on the line prior to the chiller, and the number of chill tanks (more tanks equals less organics). Excessive organic material (blood, digesta, fat, protein) in the chiller will result in less chlorine being available to kill bacteria, as it will be bound up and rendered useless by the organic material.
Experiments conducted at the USDA’s Western Regional Research Center, Agricultural Research Service concluded that a free chlorine residual could not be established in a commercial poultry chiller even by adding up to 400 ppm of free chlorine (Tsai et al., 1992). When chlorine reacts with organic material, it generally loses its microbiocidal properties, and can no longer act as a disinfectant (White, 1992). Therefore, in order to maximize chlorine use in poultry chillers, efforts should be made to reduce the amount of organic material in these systems. Pre-scald bird brushes, effective carcass rinse systems, proper bleedout procedures, counter-current scalders and chillers, proper fresh water make-up in scalders and chillers, all contribute to lowering organic loading of the chillers. Use of monochloramine can avoid these issues. Monochloramine is not bound by organic material, is effective, and does not result in the formation of carcinogenic compounds (Axtell et al., 2006).
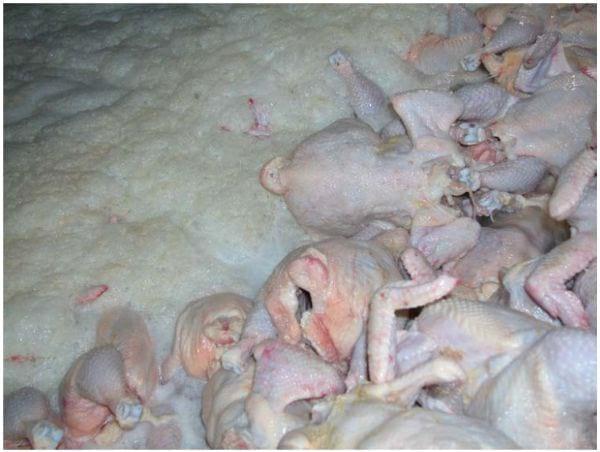
Many of the chillers in the industry are more like a bath than a river. The water is stagnant and organic material builds up during the shift (Figure 11). Also, fat builds up on the chiller paddles and sides of the chiller (Figure 12). This allows for Salmonella to be encased in the fat, offering it protection from the sanitizers used in the chiller. Suggestions for maintaining a balanced chiller include:
1. Maintain proper water flow direction (counter-current).
2. Maintain proper water pH.
3. Maintain proper chlorine level.
4. Maintain water temperature below 40ºF.
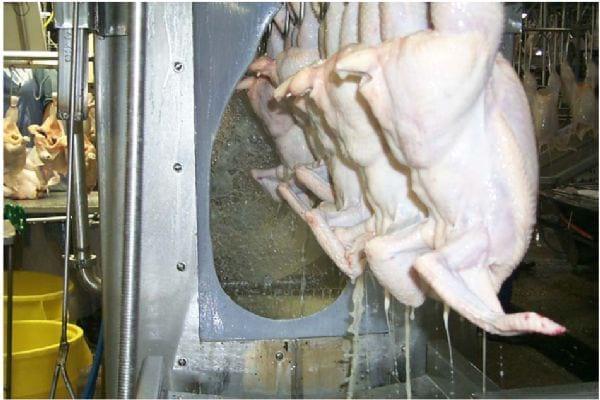
Photo 19. Excessive organic loading in the chiller
Photo 20. Fat buildup in the chill system.
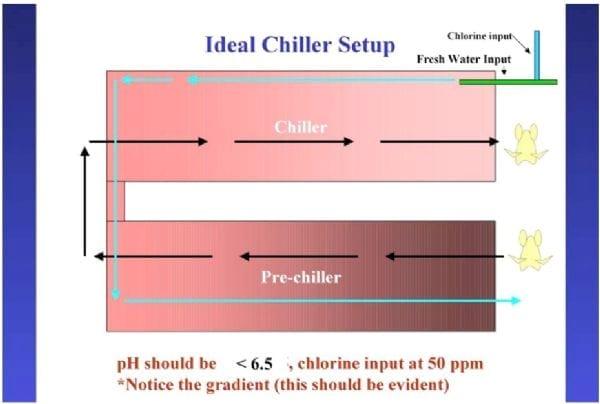
A properly operating chiller should have a visible gradient such that the water at the chiller exit is significantly cleaner than the water at the entrance. This is accomplished by adding all of the fresh water input and newly added chlorine directly to
the exit end of the chiller as close to the exit paddles as possible. This will result in a “clean space” near the exit end of the chiller. In this space, chlorine will be able to act against bacteria, similar to the way a post-chill dip tank works. The ideal chiller setup is depicted in Figure 12.
Figure 12. Ideal chiller setup.
Based on a survey in February of 2006 by the U.S. Poultry and Egg Association found at http://www.fsis.usda.gov/PDF/Slides_022406_EKrushinskie.pdf, the chemicals used in the U.S. for chiller applications and the percentage of plants that use them include hypochlorous acid (72%), peracetic acid (18%), chlorine dioxide (8%), bromine (1%), and monochloramine (1%). Other chemicals not listed in the survey that have been used in the industry include sodium acid sulfate and electrolyzed oxidative water. These chemicals all have advantages and disadvantages associated with their use and some companies may find that one or the other is most appropriate for their specific plant environment. Overall, the chiller, if operated properly can be the most significant intervention step for controlling Salmonella prevalence on broiler carcasses.
Post-chill dips and sprays
Poultry processors are employing the “hurdle hypothesis” to reduce Salmonella at different locations throughout the plant. The “hurdle hypothesis” is the premise that the more hurdles (i.e. interventions) that are employed against Salmonella, the less likely it is that Salmonella cells will be able to survive until the end of the process. As a final intervention and hurdle, companies are now using post-chill dips or sprays.
These systems are advantageous in that the chickens are as clean as they will be throughout the process and the ability of any given chemical to contact bacteria on the surface of the skin without interference from organic material is highest at this point. In general, fecal material, fat, protein, blood, bile, and bacterial biofilms that may be on the surface of the carcass have been removed by the time the carcass exits the chiller. Thus, the bacteria are most susceptible to disinfection at this point.
The chemicals that are being used for this purpose include acidified sodium chlorite (Sanova), hypochlorous acid, peracetic acid (FMC 323 or Parasafe and Inspexx 100), a mixture of hydrochloric, citric, and phosphoric acid (FreshFx), chlorine dioxide (multiple companies), and electrolyzed oxidative acidic water (EAU) to name a few. The U.S. Poultry and Egg Association industry survey found at http://www.fsis.usda.gov/PDF/Slides_022406_EKrushinskie.pdf indicated that of the companies that participated in the survey, 67% of the industry uses sodium chlorite, 25% use chlorine dioxide, and 8% use hypochlorous acid. The dip tanks used for these applications generally vary from small 50 to 100 gallon tanks to much larger (5,000 to 10,000) gallon pre-chiller type tanks. Likewise, the contact time used by these poultry companies varies from 8 seconds to 30 minutes. The spray systems are generally similar to those used for online reprocessing.
Because the organic loading is so low on carcasses after chilling, the efficacy of oxidant type chemicals is very high. These chemicals are most effective in situations where they can directly contact bacteria without interference from organic material. Likewise, acid based sanitizers are very effective as well because they are able to have an extended contact time. Whether a spray or dip is used, if the chemical is applied after the chiller, no other water washes are used. Therefore, the contact time may be hours or days. Because of this effect, sanitizers such as cetylpyridinium chloride, and other chemicals that have a long-term residual effect on bacteria should be used with caution in post-chill dip situations. The reason is that, although chemicals that leave a residual may be very effective in post-chill dips or sprays and they may significantly extend the shelf-life of the product because of this residual, the U.S. Food and Drug Administration (FDA) requires that chemicals that have a “material effect” on the product after packaging, such as extending shelf-life, must be added to the label as an additive (preservative). Thus, the processor would have to add the chemical to the label and this is generally viewed in a negative light in terms of consumer acceptance.
The decision to use a dip or spray system to apply the chemicals post-chill is based on whether the company normally rehangs the carcasses post-chill. For example, for companies that normally process whole ready-to-cook carcasses, these carcasses are packaged after chilling and would not be rehung. Thus, a dip system is more useful in this situation. Whereas, if the company debones most or all of the carcasses, then the carcasses will be rehung on a line and the spray system may be much easier to install and used in this scenario.
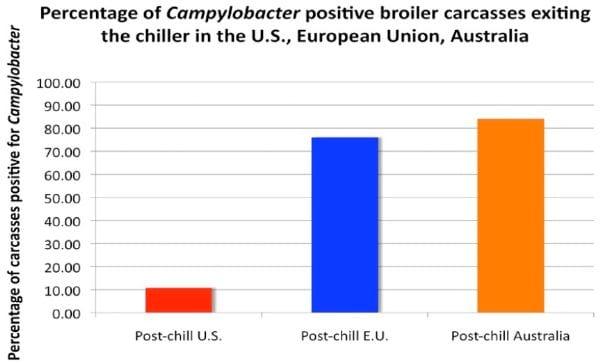
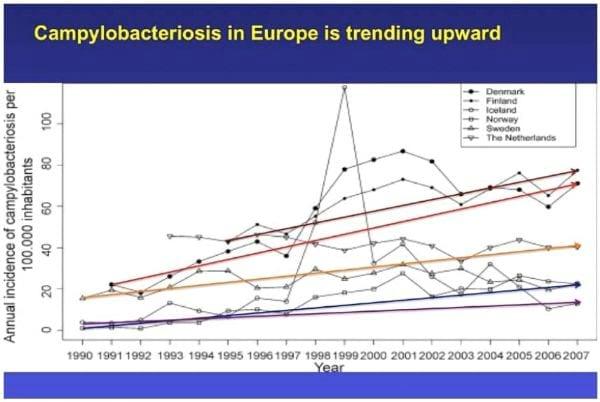
Overall, the companies are achieving success using post-chill dips or sprays and are finding that the interventions throughout the plant, combined with a post-chill dip system can be effective for lowering Salmonella and other bacteria to acceptable levels. It is extremely important to note that European reticence to disallow chemical disinfection of chickens has resulted in a disastrously high food-borne infection rate of humans due to Campylobacter from poultry sources. The following figures tell the story.
Figure 13.
Figure 14.
Figure 15.
There is no question that chemical intervention works and is effective. The European Union cannot control Campylobacter by simply using pre-harvest strategies, as they attempt to do with Salmonella. The interventions simply do not exist for this organism.
References
Abu-Ruwaida, A. S., W. N. Sawaya, B. H. Dashti, M. Murad, and H. A. Al-Othman, 1994. Microbiological quality of broilers during processing in a modern commercial slaughterhouse in Kuwait. J. Food Prot. 57:887-892.
Axtell, S. P., S. M. Russell, and E. Berman, 2006. Effect of immersion chilling of broiler chicken carcasses in monochloramine on lipid oxidation and halogenated residual compound formation. Journal of Food Protection 69(4):907-911.
Barnhart, E. T., D. J. Caldwell, M. C. Crouch, J. A. Byrd, D. E. Corrier, and B. M. Hargis, 1999. Effect of lactose administration in drinking water prior to and during feed withdrawal on Salmonella recovery from broiler crops and ceca. Poultry Science 78:211-214.
Byrd, J. A., B. M. Hargis, D. J. Caldwell, R. H. Bailey, K. L., Herron, J. L. McReynolds, R. L. Brewer, R. C. Anderson, K. M. Bischoff, T. R. Callaway, and L. F. Kubena, 2001. Effect of lactic acid administration in the drinking water during preslaughter feed withdrawal on Salmonella and Campylobacter contamination of broilers. Poultry Science 80:278-283.
Cason, J. A., J. S. Bailey, and N. A. Cox, 1993. Location of Salmonella typhimurium during incubation and hatching of inoculated eggs. Poult. Sci. 72:2064-2068.
Cason, J. A., J. S. Bailey, and N. A. Cox, 1994. Transmission of Salmonella typhimurium during hatching of broiler chicks. Avian Dis. 38:583-588.
Cox, N. A., M. E. Berrang, and J. A. Cason, 2000. Salmonella penetration of egg shells and proliferation in broiler hatching eggs-A review. Poult. Sci. 79:1571-1574.
Cox, N. A., J. S. Bailey, J. M. Mauldin, and L. C. Blankenship, 1990. Research note: Presence and impact of Salmonella contamination in commercial broiler hatcheries. Poultry Sci. 69:1606-1609.
Cox, N. A., J. S. Bailey, J. M. Mauldin, L. C. Blankenship, and J. L. Wilson, 1991. Research note: Extent of salmonellae contamination in breeder hatcheries. Poultry Sci. 70:416-418.
Green, T. A., S. M. Russell, and D. L. Fletcher, 1998. Effect of chemical sanitizing agents on ATP bioluminescence measurements. J. Food Prot. 61(8):1013-1017.
Green, T. A., S. M. Russell, and D. L. Fletcher, 1999. Effect of chemical cleaning agents and commercial sanitizers on ATP bioluminescence measurements. J. Food Prot. 62(1):86-90.
Goren, E., W. A. de Jong, P. Doornenbal, N. M. Bolder, R. W. Mulder, and A. Jansen, 1988. Reduction of Salmonella infection of broilers by spray application of intestinal microflora: A longitudinal study. Vet. Q. 10:249-255.
Hargis, B. M., D. J. Caldwell, R. L. Brewer, D. E. Corrier, and J. R. DeLoach, 1995. Evaluation of the chicken crop as a source of Salmonella contamination of broiler carcasses. Poultry Science 74:1548-1552.
Izat, A. L., M. Colberg, M. H. Adams, M. A. Reiber, and P. W. Waldroup, 1989. Production and processing studies to reduce the incidence of salmonellae on commercial broilers. J. Food Protect. 52:670-673.
Kemp, G. K., M. L. Aldrich, M. L. Guerra, and K. R. Schneider, 2001. Continuous online reprocessing of fecal- and ingesta-contaminated poultry carcasses using an acidified sodium chlorite antimicrobial intervention. J. Food Protect. 64:807-812.
Muira, S., G. Sato, and T. Miyamae, 1964. Occurrence and survival of Salmonella organisms in hatcher chick fluff in commercial hatcheries. Avian Dis. 8:546-554.
Northcutt, J. K., S. I. Savage, and L. R. Vest, 1997. Relationship between feed withdrawal and viscera condition of broilers. Poultry Science 76:410-414.
Northcutt, J. K., D. P. Smith, M. T. Musgrove, K. D. Ingram, and A. Hinton, Jr., 2005. Microbiological impact of spray washing broiler carcasses using different chlorine concentrations and water temperatures. Poultry Science 84:1648-1652.
Okrend, A. J., R. W. Johnston, and A. B. Moran, 1986. Effect of acetic acid on the death rates at 52 C of Salmonella newport, Salmonella typhimurium, and Campylobacter jejuni in poultry scald water. J. Food Protect. 49:500-503.
Russell, S. M., 2003. The effect of electrolyzed oxidative (EO) water applied using electrostatic spraying on pathogenic and indicator bacteria on the surface of eggs. Poultry Science 82:158-162.
Tsai, Lee-Shin, B. T. Molyneux, and J. E. Schade, 1992. Chlorination of Poultry Chiller Water: Chlorine Demand and Disinfection Efficiency. Poultry Sci. 71:188, 194-195.
United States Department of Agriculture, Federal Register, 1996. 9 CFR Part 304 et al., Pathogen Reduction; Hazard Analysis and Critical Control Point (HACCP) Systems; Final Rule. Vol. 61, Number 144, pp. 38846-38848.
White, G. C., 1992. The Handbook of Chlorination and Alternate Disinfectants, 3rd Ed., Van Nostrand Reinhold, New York.