Introduction
The poultry industry is perhaps the most sensitive sector of animal production. The high degree of genetic development seeking greater productivity could have been responsible for the development of animals more susceptible to diseases. Therefore, as a result of the constant challenges experienced by these birds, their immune system must be running smoothly so they can respond to different stimuli (Santos et al., 2009b).
The immune system has a key role in the protection of poultry against pathogens. As in mammals, birds immune system is complex and involves a variety of cells and soluble factors that must work together to produce a protective immune response (Santos et al., 2009b). Several immunosuppressive pathogens occur frequently in endemic areas of poultry production and exposure to these pathogen agents can affect the immune system functions. Immunosuppressed lots have an increased incidence of secondary infections with a lower yield, where one of the main immunosuppressive agents are mycotoxins (Santos et al., 2009b).
Mycotoxins affect the immune system by interacting with cellular DNA, affecting protein synthesis and inhibiting mainly lymphocytes, plasma cells and macrophages (Corrier, 1991). The first scientific studies on the immunosuppressive action of mycotoxins are mainly related to aflatoxin poisoning (Leeson et al., 1995). But in the world in which hundreds of mycotoxins are known, studies related to the issue of immunosuppression are concentrated on not more than ten. However, in general, it is believed that all mycotoxins can reduce or suppress resistance against infectious diseases (Freire, 1995). Therefore, mycotoxins can render birds more susceptible to the development of various pathologies, as in the case of salpingitis.
Salpingitis is an inflammation process of the oviduct and is mainly caused by the bacterium Escherichia coli. The process is generally chronic and found in egg-producing birds. When these birds are affected, can show weight loss and often death without any clinical sign. The gross appearance of salpingitis is characterized by a mass of cheese-like and dried material inside the oviduct, and also the walls of this organ get extremely thin (Santos et al., 2009a)
Materials & Methods
The study material was obtained from a processing plant with Federal Inspection of the State of Minas Gerais. Consisted of 10 female broiler carcasses of 45 (5 carcasses, lot A) and 47 days of age (5 carcasses, lot B). The carcasses were condemned due to salpingitis and were referred to the Avian Health Study Unit, Department of Veterinary Medicine, Federal University of Viçosa for microbiological (liver and oviduct) and histopathological (liver and bursa) analysis.
For microbiological analysis, liver and oviduct content swabs were performed. Swabs were placed in BHI broth and were left at a temperature of 37°C for 24 hours. Then, with a bacteriological loop, the material was placed in Petri dishes with eosin methylene blue agar and kept in incubation at 37°C for 24 hours.
Materials for histopathology were fixed in formalin in 10% saline solution and kept for 24 hours. After fixation, samples were dehydrated and clarified by following histological techniques. Histological sections after being stained with hematoxylin and eosin (Prophet et al., 1992), were examined by light microscopy.
Results & Discussion
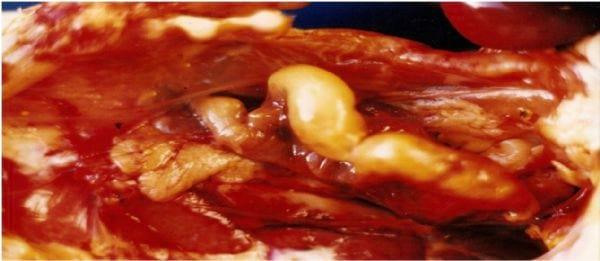
All 10 carcasses showed an oviduct with distended and thin walls, with a mass of yellow material and cheese-like appearance in the lumen (Figure 1). In the bacteriological analysis of liver and oviduct contents swabs, only the bacterium Escherichia coli was isolated in the latter.
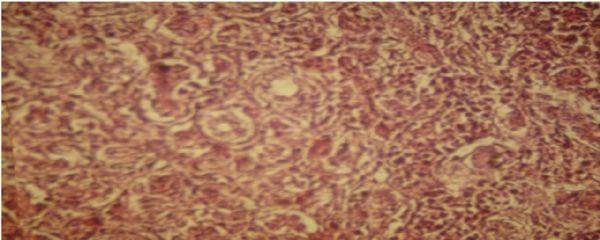
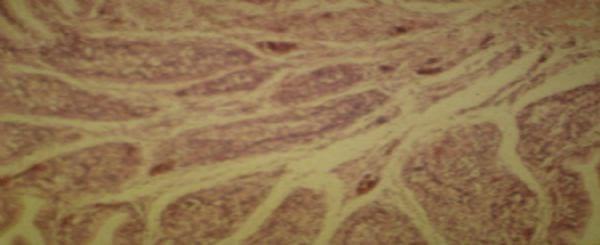
At histological examination, liver showed cell proliferation of bile ducts, degeneration and necrosis of liver cells (Figure 2), added heterophils and further evidence of fibrous conjunctive tissue. Bursa showed the presence of degeneration and necrosis of follicular cells and fibroplasia (Figure 3).
Muneer et al. (1988) believe that the immunosuppressive effect of mycotoxins is because of decreased mobility and the phagocytic activity of blood monocytes. Mycotoxins, mainly aflatoxins, increase susceptibility to bacterial diseases, namely salmonellosis (Tizard, 1998) and colibacillosis (Santos et al., 2009a). In addition, mycotoxins decreased production of immunoglobulins by lymphoid organs, and reduce the size of the bursa, spleen and thymus (Hoerr et al., 2008).
Figure 1 . Oviduct of a female broiler with distended walls and a mass of yellow material.
Figure 2 . Photomicrograph of histological section of liver with proliferation of the bile ducts cells.
Figure 3 . Photomicrograph of histological section of the bursa with reduced size lymphoid follicles, degeneration and necrosis of follicular cells and fibroplasia.
Conclusions
In this study, female broilers developed colibacillosis as salpingitis, likely due to immunosuppression caused by mycotoxins.
References
Corrier DE. 1991. Mycotoxicosis: mechanisms of immunosupression. Vet. Immunopathol. 30:73-87.
Freire RB.1995. Micotoxinas e imunossupressão em aves. pp. 115-126. In: Simpósio Internacional sobre micotoxinas e micotoxicoses em aves. Anais, Curitiba.
Hoerr FJ. 2008. Mycotoxicoses. p.1197-1229. In: Diseases of poultry. Saif YM, Fadly AM, Glisson JR, McDougald LR, Nolan LK, Swayne DE. (eds.) Ames: Blackwell publishing. Iowa USA.
Leeson S, Diaz G, Summers JD. 1995. Poultry metabolic disorders and mycotoxins. Guelph, Canada: University Books.
Muneer MA, Farah IO, Newman JA, Goyal SM. 1988. Immunosuppession in animals. Rev. Br. Vet. J. 144:288-301.
Prophet EB, Mills B, Arrington JB 1992. Laboratory methods in histotechnology. Washington, EUA:: American Registry of Pathology.
Santos BM, Moreira MA, Dias CCA. 2009a. Manual de doenças avícolas. Editora UFV. Viçosa, Brasil.
Santos BM, Pereira CG, Marin SM, Morato TG. 2009b. Prevenção e controle de doenças infecciosas nas aves de produção. Editora UFV. Viçosa, Brasil.
Tizard IR.1998. Imunologia veterinária. 5 ed. São Paulo: Roca. Brasil.