I. INTRODUCTION
The optimum utilization of nutrients, especially energy and protein, by poultry is essential in commercial production. The efficiency of feed utilization has been improved through genetic development (Rege 1994), alongside feed processing and use of feed additives, especially microbial enzymes. Energy utilization in poultry can be measured by assessing metabolizable energy or net energy, which may be for production (NEp) or for maintenance (NEm). The NEp is regarded as a more sensitive measure of energy utilization by the chickens receiving enzyme because it considers the efficiency of utilization of ME for growth. The utilization of feeds can be enhanced by pelleting the diets as well as enzyme supplementation. Birds fed on pellets use less energy for feeding, thus reserving the available energy for growth (Amerah et al. 2008). The diets that were tested in this study represent typical diets that are utilized by small-to-medium scale (SM) broiler producers in Tanzania, which generally are poorly formulated and have never been evaluated to determine their nutritive value. This study represents an attempt to develop suitable diets for that sector, with the additional benefits of data on energy values and utilization of such energy.
II. MATERIALS AND METHODS
A total of 480 day-old unsexed Ross 308 broiler chicks was randomly allocated according to a 2x4 factorial design. There were 2 feed forms (mash or pellet) and four microbial enzyme treatments (none, AxtraXB, QB and AxtraXB+QB). Each treatment was replicated six times, with 10 birds per replicate. Birds were raised in deep litter pens in climate-controlled rooms at the Centre for Animal Research and Teaching, University of New England, Australia. Eight dietary treatments were formulated from maize, soybean, cottonseed and fish meal, which are commonly used by poultry producers in Tanzania. Diets were fed as-is or supplemented with 100 mg/kg of either Axtra XB (Danisco Animal Nutrition, UK), providing 250 units of βglucanase and 2,500 units of xylanase per kg of the formulated feeds, or 100 mg/kg of Quantum Blue (AB Vista, Marlborough,UK) providing 500 FTU, or a mixture of the two enzymes (50 mg/kg of each). These treatments were used throughout the production cycle (d0-35). Titanium dioxide (TiO2) was added to the grower diets at 0.5 % as an indigestible marker in order to assess the AME and metabolizable energy intake (MEI). Twelve day-old chicks were electrically stunned, killed by cervical dislocation, minced and later analysed to obtain the baseline data of energy, fat and protein contents. Excreta was collected from d19-21 using aluminium foil sheets. The excreta samples were pooled by replicate (per pen), mixed and stored at -20 °C. Two birds per cage were killed at d24 and immediately stored intact at -20 ºC. The intact day-old and adult carcasses (d24) were minced and freeze-dried. Ileal digesta samples were collected from another two birds at d24 of age. They were then ground (0.5 mm) and analysed for DM, CP, GE and total fat. Excreta samples were freeze-dried, ground and analysed for GE and TiO2 contents.
The AME was calculated according to Olukosi et al. (2008):
AME (MJ/kg) = GEdiet – [GEexcreta × (Tdiet /Texcreta)], where GEdiet = diet gross energy (MJ/kg); GEexcreta = excreta GE (MJ/kg); Tdiet = titanium dioxide concentration of diets (g/kg of DM intake); and Texcreta = concentration of titanium dioxide in the excreta (g/kg of DM intake).
The NEp was calculated by subtracting initial carcass GE from the final carcass GE. The MEI was calculated by multiplying AME by bird FI value. The efficiency of ME use for energy retention was calculated as (KRE) = NEp/MEI, while efficiency of ME use for protein retention was calculated as (KREprotein) = REprotein/MEI. Heat production was calculated by subtracting NEp from MEI. Diets were analysed for CP and gross energy. Data were analysed by the general linear model (GLM) procedure of Minitab statistical software version 17 (Minitab 2014).
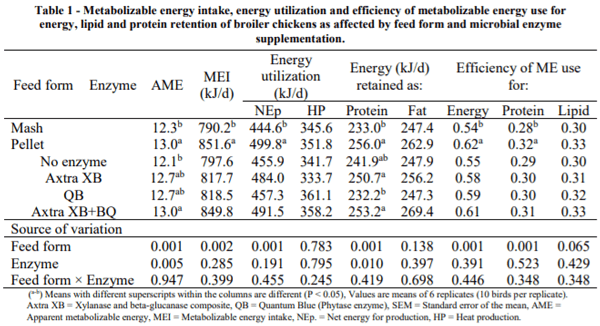
III. RESULTS
The effects of feed form and microbial enzyme supplementation on AME and energy utilization are presented in Table 1. There was no interaction between feed form and enzyme supplementation for either AME or the energy utilization measurements. Birds fed the pelleted diets had higher (P < 0.001) AME than those fed the mash diets. Microbial enzyme supplementation increased (P < 0.005) AME, with the best result observed when Axtra XB and QB were combined in the diet. Metabolizable energy intake, NEp and the energy retained as protein were comparatively improved (P < 0.002) when pelleted diets were fed. Adding microbial enzymes to the diets increased (P < 0.01) energy retained as protein, coupled with a slight increase in MEI and efficiencies of ME usage. Neither feed form nor microbial enzyme addition had significant effect (P > 0.05) on the heat production (HP), energy retained as fat and efficiency of ME used for lipid retention.
IV. DISCUSSION
The increased AME, MEI, NEp and energy retained as protein in the group of birds fed pelleted diets compared to those fed mash could be due to the fact that pelleting diets reduce feed wastage and particle selection during consumption. Furthermore, birds use less energy when eating pelleted diets, thus conserving more energy, some of which could be lost when fed mash diets (Serrano et al. 2012). Pellet processing conditions, including pressing, heating and addition of moisture might deactivate anti-nutritive factors (ANF) and improve the palatability of diets, leading to an improvement in nutrient availability, particularly energy, for the bird (Abdollahi et al. 2013). The current results are supported by the findings of Greenwood et al. (2004), who reported that pelleting diets make more energy available to birds and hence proportionally increase dietary energy in order to achieve maximum utilization and retention. Similar enzymes have been shown to reduce the negative effects of ANF and improvement in the activities of endogenous enzyme activities (Cowieson et al. 2006). The results of this study suggest that the test diets would be better fed as pellet and would benefit further from supplementation with microbial enzyme supplements.
ACKNOWLEDGEMENT: We express our gratitude to the University of New England for financial support of this study.
Abstract presented at the 30th Annual Australian Poultry Science Symposium 2019. For information on the latest edition and future events, check out https://www.apss2021.com.au/. 











