Effect of Dietary Inclusion of Mycodetox B2 on Liveability, Immunity and Organ Pathology During Aflatoxicosis in Turkey Poults
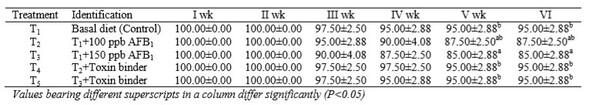
To evaluate the efficacy of Mycodetox B2 in ameliorating aflatoxicosis, day-old Turkey poults (n=200) were divided into five treatment groups, viz. T1: control; T2: T1+100ppb aflatoxin B1 (AFB1); T3: T1+150ppb AFB1; T4: T2+Mycodetox B2; T5: T3+Mycodetox B2. Each diet was fed to four replicates of 10 birds each for 42 days. The study revealed that overall liveability percent in T3 was lower (P<0.05) than T1. The CMI and HA titre values of T2 and T3 was lower (P<0.05) than T1. The CMI and HA titre values in T4 and T5 was higher (P<0.05) than T2 and T3 and similar to T1. The haemoglobin (Hb) value in T2 and T3 was lower (P<0.05) than T1. The Hb value in T4 and T5 was higher (P<0.05) than T2 and T3; and similar to T1. The H/L ratio of T1 was lower (P<0.05) than T2 and T3. The H/L ratio of T4 and T5 was lower (P<0.05) than T2 and T3 and similar to T1. Grossly, in T2 and T3 petechiae were seen in the kidneys and lungs; ecchymotic haemorrhages were also observed in liver and leg muscles; petechial haemorrhages were also recorded in thymus and bursa. The organs of T4 and T5 were normal as in T1.
Histopathologically, liver of T2 and T3, revealed congestion of central vein, mild degenerative changes in the hepatocytes and areas of necrosis associated with mild mononuclear (MNCs) cells infiltration especially in the portal areas. Hepatocytes also showed moderate fatty change, mild hyperplastic changes in the small bile ducts and lymphoid aggregation in the form of nodules. Haemorrhages in the liver parenchyma were quite marked. In T4 and T5, the architectural and cellular organization in the liver was normal as in T1. It was concluded that dietary AFB1 at 100 or 150ppb levels resulted in reduced liveability, immunity; decreased haemoglobin concentration, increased H/L ratio; and morphological and histopathological alterations in the internal organs. Moreover, the dietary incorporation of Mycodetox B2 ameliorated the adverse effects of aflatoxicosis in Turkey poults.
2.1 Production and Analysis of Aflatoxin

Azzam AH and Gabal MA (1998). Aflatoxin and immunity in layer hens. Avian Pathology, 27: 570-577. Bakshi CS (1991). Studies on the effect of graded dietary levels of aflatoxin on immunity in commercial broilers. M.V.Sc. thesis submitted to Deemed University, IVRI, Izatnagar.
Basmacioglu H, Oguz H, Ergul M, Col R and Birdane YO (2005). Effect of dietary esterified glucomannan on performance, serum biochemistry and haematology in broilers exposed to aflatoxin. Czech Journal of Animal Science, 50: 31-39. Chang CF, Hamilton PB and Weeks BA (1976). Impairment of leukocyte chemotaxis and phagocytosis. American Society for Microbiology, 181: 10.
Churchil RR, Praveena PE and Maldhure NA (2014). Effect of esterified glucomannan in amelioration of aflatoxin induced microscopic changes in broiler chicks. Journal of Poultry Science and Technology, 2: 36-37. Churchil RR, Praveena PE and Maldhure NA (2014). Effect of esterified glucomannan in amelioration of aflatoxin induced microscopic changes in broiler chicks. Journal of Poultry Science and Technology, 2: 36-37.
Corrier DE and Deloach JR (1990). Evaluation of cell mediated cutaneous basophil, hypersensitivity in young chickens by an interdigital skin test. Poultry Science, 69: 403-408. Culling CFA (1968). Histological and histochemical staining techniques, (3rd Edn.) Woodsworth Publication Pvt. Ltd., London. DeDuve C and Wattiaux R (1966). Functions of lysosomes. Annual Review of Physiology, 28: 435-492.
Denli M, Blandon JC, Guynot, ME, Salado S and Perez JF (2009). Effects of dietary Afladetox on performance, serum biochemistry, histopathological changes and aflatoxin residues in broilers exposed to aflatoxin B1. Poultry Science, 88: 1444-1451. Deo P, Blaney B and Dingle J (1998). Aflatoxin decreases immunity and stress reaction in poultry. Proceedings on Australian Poultry Science Symposium, 10: 172-175. Ghosh RC and Chauhan HVS (1991). Suppression of cell mediated immunity by purified aflatoxin B1 in broiler chicks. Indian Journal of Animal Health, 30(1): 23-26.
Ghosh RC, Chauhan HVS and Roy S (1991). Immunosuppression in broilers under experimental aflatoxicosis. British Veterinary Journal, 146: 457-462. Giambrone JJ, Diener UL, Davis ND, Panangala VS and Hoerr FJ (1985). Effects of purified aflatoxin on broiler chickens. Poultry Science, 64: 852-858. Giambrone JJ, Ewert DL, Wyatt RD and Eidson CS (1978). Effect of aflatoxin on the humoral and cell-mediated immune systems of chicken. American Journal of Veterinary Research, 39: 305. Gopi K (2006). Influence of melatonin on aflatoxicosis in broiler chickens.
M.V.Sc. thesis, IVRI, Izatnagar. Huff WE, Kubena LF, Harvey RB, Hagler VM, Swanson SP, Phillips TD and Creger CR (1986). Individual and combined effects of aflatoxin and deoxynivalenol (DON, vomitoxin) in broiler chickens. Poultry Science, 65: 1291-1298. Kadian SK, Monga DP and Goel MC (1988). Effect of aflatoxin B1 on DTH and phagocytic activity of reticuloendothelial system in chickens. Mycopathologia, 104: 33-36. Katole SB, Kumar P and Patil RD (2013). Environmental pollutants and livestock health: a review
. Veterinary Research International, 1(1): 1-13. Kececi T, Oguz H, Kurtoglu V and Demet O (1998). Effects of polyvynylpoly-pyrrolidone, synthetic zeolite and bentonite on serum biochemical and haematological characters of broiler chickens during aflatoxicosis. British Poultry Science, 39: 452-458. Khatke PA, Singh R and Mandal AB (2012a). Efficacy of biological adsorbents to ameliorate aflatoxicosis in broiler chicken: effect on immune response and histopathology of liver. Indian Journal of Poultry Science, 48(1): 27-32.
Khatke PA, Singh R, Mandal AB and Tyagi PK (2012b). Evaluation of the ability of Saccharomyces cerevisiae and mannan oligosaccharides to ameliorate the adverse effects of aflatoxin B1 in broiler chickens. Indian Journal of Poultry Science, 47(2): 176-82.
Khatke PA, Singh R, Mandal AB and Tyagi PK (2012c). Ability of biological adsorbents to ameliorate aflatoxicosis in broiler chicken: effect on blood biochemicals and organ weights. Indian Journal of Poultry Science, 48(1): 117-121. NRC (1994). National research council. Nutrient requirements of poultry. (9th revised Edn.), National Academy Press, Washington, D.C. Oguz H, Hadimli HH, Kurtoglu V and Erganis O (2003). Evaluation of humoral immunity of broilers during chronic aflatoxin (50 and 100 ppb) and clinoptilolite exposure. Journal of Veterinary Medicine, 154: 483486.
Patel VR, Choubey M, Trangadiya BJ and Raval AP (2015). Mycotoxins in feed and their amelioration: a review. International Journal of Animal and Veterinary Sciences, 2: 28-33. Pathak GP, Sharma R, Patil RD, Sharma DK and Varshneya C (2017). Effect of dietary supplementation of esterified glucomannan against aflatoxin B1-induced toxicity in broiler chicks. Journal of Poultry Science and Technology, 5(1): 1-6.
Patial V, Asrani RK and Patil RD (2013). Nephrotoxicity of ochratoxin-A in Japanese quail: a clinico-pathological study. Journal of Poultry Science and Technology, 1(1): 07-12. Patil RD and Degloorkar NM (2016a). Protective effect of Bantox® on ochratoxin A-induced liver Damage in broilers: a histopathological study. Journal of Poultry Science and Technology, 4(4): 46-51. Patil RD and Degloorkar NM (2016b). Nephrotoxicity of ochratoxin A in broiler chicken and its amelioration with Bantox®: Histopathological assessment. Journal of Poultry Science and Technology, 4(4): 52-58.
Patil RD and Degloorkar NM (2018). Ameliorative efficacy of commercial mycotoxin binder (Bantox®) against ochratoxin A-induced microscopic pathology in broiler birds. Journal of Poultry Science and Technology, 6(2): 26-30. Patil RD, Degloorkar NM and Pawar PK (2017a). Ameliorating effects of Bantox® on clinical manifestation and growth performance of broiler chicken fed with ochratoxin A. Journal of Poultry Science and Technology, 5(3): 22-27. Patil RD, Degloorkar NM and Pawar PL (2017b). Effects of ochratoxin a feeding on organ weights and gross pathological changes in broiler chicken and its amelioration with Bantox®. Journal of Poultry Science and Technology, 5(4): 44-51. Patil RD,
Degloorkar NM, Moregaonkar SD and Kulkarni GB (2005). Ameliorative efficacy of Bantox in induced ochratoxicosis in broilers: a haemato-biochemical study. Indian Journal of Veterinary Pathology, 29(2): 90-94. Patil RD, Dwivedi P and Sharma AK (2006). Critical period and minimum single oral dose of ochratoxin A for inducing developmental toxicity in pregnant Wistar rats. Reproductive Toxicology, 22(4): 679-687. Patil RD, Sharma R and Asrani RK (2014). Mycotoxicosis and its control in poultry: a review. Journal of Poultry Science and Technology, 2(1): 1-10.
Patil RJ, Tyagi JS, Sirajudeen M, Singh R, Moudgal
RP, and Mohan J (2013). Effect of dietary melatonin and ltryptophan on growth performance and immune responses of broiler chicken under experimental aflatoxicosis. Iranian Journal of Applied Animal Science, 3(1): 139-144. Pons D, Cucullu AP, Lee LS, Robertson JA and Goldblatt LA (1966). Determination of aflatoxins in agricultural products: use of aqueous acetone for extraction. Journal of Analytical Chemistry, 49: 544-552. Shamsudeen P, Shrivastava HP and Singh R (2013).
Biointeraction of chelated and inorganic copper with aflatoxin on growth performance of broiler chicken. International Journal of Veterinary Science, 2(3): 106110. Sharma M (2013). Interaction of aflatoxicosis with methionine and zinc levels in diet of broiler chickens. M.V.Sc. thesis, I.V.R.I., Izatnagar (UP) India. Sharma M, Singh R and Mandal AB (2015). Effect of methionine supplementation in ameliorating aflatoxicosis in broiler chickens. Animal Nutrition and Feed Technology, 15: 161-169.
Sharma M, Singh R and Mandal AB (2016). Effect of aflatoxin, ochratoxin and their interaction on growth performance, immunity and jejunal morphometry of broiler chickens. Indian Journal of Poultry Science, 51(3): 253-258. Sharma M, Singh R, Mandal AB and Gupta VP (2014). Efficacy of zinc in amelioration of aflatoxicosis in broiler chickens.
Indian Journal of Animal Sciences, 84(3): 311-315. Shotwell OL, Hesseltine CV, Stubblefield RD and Sorenson WG (1966). Production of aflatoxin on rice. Applied Microbiology, 14: 425-429. Siegel PB and Gross WB (1980). Production and persistence of antibodies to sheep erythrocytes. 1. Directional selection. Poultry Science, 59: 1-5. Silambarasan S, Singh R and Mandal AB (2013). Evaluation of the ability of adsorbents to ameliorate the adverse effects of aflatoxin B1 in broiler chickens. Indian Journal of Animal Sciences, 83: 73-77. Silambarasan S, Singh R and Mandal AB (2015). Efficacy of certain adsorbents on carcass traits and livability of broiler chickens fed aflatoxin B1 contaminated diet. Indian Journal of Poultry Science, 50(1): 113-117. Silambarasan S, Singh R and Mandal AB (2016). Evaluation of adsorbents to ameliorate the adverse effects of aflatoxin B1 on blood biochemicals, immune response and histopathology of liver in broiler chickens. Indian Journal of Poultry Science, 50(3): 267-271.
Singh M, Singh R and Mandal AB (2019a). Influence of Saccharomyces cerevisiae to ameliorate adverse effects of ochratoxin A on histopathology of kidney and liver in broiler chickens. Livestock Research International, 7(1): 26-31. Singh M, Singh R and Mandal AB (2019b). Ameliorative effects of vitamin E on renal and hepatic microscopic changes during experimental ochratoxicosis in broiler chickens. Journal of Poultry Science and Technology, 7(1): 15-20.
Singh R (2019a). Efficacy of choline in ameliorating aflatoxicosis in broiler chickens. International Journal of Current Microbiology and Applied Sciences, 8(2): 2356-2365. Singh R (2019b). Effect of dietary supplementation of mycodetox b2 on production performance, organ weights and serum biochemicals during aflatoxicosis in Turkey poults. Livestock Research International, 7: 3239.
Singh R (2019c). Effect of supplementation of toxin binder (Mycodetox B1) on liveability, immune response and organs pathology in induced aflatoxicosis in Japanese quails. Livestock Research International, 7(1): 5-11. Singh R (2019d). Effect of supplementation of toxin binder (Mycodetox B2) on liveability, immune response and pathology of organs during aflatoxicosis in Japanese quails. Journal of Poultry Science and Technology, 7(1): 1-7.
Singh R (2019e). Ameliorative effects of Mycodetox B1 on production performance, organ weights and serum biochemicals during aflatoxicosis in Turkey poults. Livestock Research International, 7(1): 40-48. Singh R (2019f). Ameliorative efficacy of mycodetox b1 on liveability, immunity and organ pathology during experimental aflatoxicosis in Turkey poults. Livestock Research International, 7(1): 49-54.
Singh R and Mandal AB (2013). Efficacy of ascorbic acid and butylated hydroxyanisole in amelioration of aflatoxicosis in broiler chickens. Iranian Journal of Applied Animal Science, 3(3): 595-603. Singh R, Mandal AB and Biswas A (2013b). Efficacy of DLMethionine in amelioration of aflatoxicosis in coloured broiler chicken. Indian Journal of Animal Sciences, 83(12): 1329-1334.
Singh R, Mandal AB and Divya (2015). Efficacy of methionine hydroxy analogue in ameliorating aflatoxicosis in Japanese quails. Animal Nutrition and Feed Technology, 15: 227-234. Singh R, Mandal AB and Shrivastav AK (2013a). Amelioration of aflatoxicosis in coloured broiler chickens by dietary butylated hydroxytoluene. Animal Nutrition and Feed Technology, 13: 235-242.
Singh R, Sharma M, Mandal AB and Tyagi PK (2016). Comparative efficacy of DL Methionine vis a vis methionine hydroxy analogue in ameliorating aflatoxicosis in Japanese quails. Indian Journal of Poultry Science, 51(2): 168-173. Singh R, Shrivastava HP and Shrivastav AK (2011). Effect of dietary aflatoxin on the performance of Turkey poults. Indian Journal of Poultry Science, 46(2): 185-188.
Thaxton JP, Tung HT and Hamilton PB (1974). Immunosuppression in chickens by aflatoxin. Poultry Science, 53: 721-725. Tung HT, Wyatt RD, Thaxton P and Hamilton PB (1975). Concentrations of serum protein during aflatoxicosis. Toxicological Application Pharmacology, 34: 320-326.
Virdi JS, Tiwari RP, Saxena M, Khanna V, Singh G, Saini SS and Vadehra DV (1989). Effects of aflatoxin on immune system of the chicken. Journal of Applied Toxicology, 9: 271-275. Yunus AW, Razzazi-Fazeli E and Bohm J (2011). Aflatoxin B1 in affecting broiler’s performance, immunity, and gastrointestinal tract: a review of history and contemporary issues. Toxins, 3: 566-590.







.jpg&w=3840&q=75)