Genetic Selection of Cattle for Improved Immunity and Health
Published: September 25, 2014
By: Bonnie A. Mallard, Shannon Cartwright, Mehdi Emam, Kelly Fleming, Natasha Gallo, Doug Hodgins, Marlene Paibomesai, Kathleen Thompson-Crispi and Lauraine Wagter-Lesperance (University of Guelph)
Take-Home Message
- The immune system is the body’s defense system that provides disease resistance
- The immune system is genetically regulated and can be modified by genetic selection
- The University of Guelph award-winning patented High Immune Response (HIR) technology is designed to identify cattle with optimized immune responsiveness and is associated with enhanced resistance to a broad range of diseases
- The Semex Alliance has an exclusive license to utilize this technology to identify high immune responder sires, marketed under the Immunity+ trademark, which can be used to breed for improved bovine immune responses
- The HIR technology offers both a novel management and selection tool to improve herd health
Introduction
The immune system is designed to provide protection from a wide range of pathogenic micro-organisms, as well as cancers. Therefore identifying dairy cows with a natural ability to make superior immune responses reduces disease occurrence, increases farm profit and improves milk quality as well as safety. Using the University of Guelph’s patented High Immune Response (HIR) technology it is possible to classify animals as high, average or low immune responders. The high responders have the inherent ability to produce more balanced and robust immune responses compared to average or low responders. In fact, high responders have about half the disease occurrence of low responders, and can pass these superior immune response genes on to future offspring thereby accumulating health benefits within the dairy herd. In 2012 the University of Guelph granted the Semex Alliance an exclusive licence to utilize the HIR procedure to identify sires with the high immune response classification. These sires are given the designation of Immunity+, marking their enhanced capacity to make protective immune responses. The immune response traits used in the HIR test are highly heritable having heritability estimates of approximately 0.25-0.35, which is in the same range as those for milk production traits. This means that selective breeding using the HIR technology will give producers one more tool to improve herd health at the genetic level. This article will review some of the previous and current research on genetic regulation of the bovine immune system, as well as the commercial development of the HIR technology and its use to genetically improve immunity of cattle.
Background
In Canada, it is estimated to cost the dairy producer about $200 per case of mastitis, and one out of every five dairy quarters in Canada is infected with a mastitis-causing pathogen at any given point in time (Canadian Bovine Mastitis Research Network, “What’s New in the World of Mastitis Research?” http://www.medvet.umontreal.ca/rcrmb/dynamiques/PDF_AN/Results/NewspaperWhatsNew.pdf. 2009). Antibiotics may be the treatment of choice for diseases such as mastitis, but this is less than an ideal treatment in a world making every effort to restrain the use of antibiotics with a goal to limit the emergence of antibiotic resistant pathogens. Therefore there is an urgent need for alternative health strategies, both preventative and therapeutic, in which microbial drug resistance cannot develop. These may include a combination of vaccination, dietary supplementation (e.g. probiotics) and genetic strategies that can act safely and synergistically in the prevention of disease. Our team has focused their efforts on developing a better understanding of the genetic (DNA sequence information) and epigenetic (DNA structural information) regulation of the bovine immune system in order to identify prophylactic methods to improve animal health. The appeal of this particular genetic approach is that it makes use of the animal’s own inherent ability to make appropriate immune responses to a given pathogen. In addition, benefits can accrue to future generations by selective breeding, it is a non-genetically modified organism (non-GMO) tactic that can be implemented in either conventional or organic farms and it works well in combination with other preventative approaches, including vaccination. With these principles in mind, after many years of research, the HIR technology was developed and continues to be refined for additional novel applications.
The Immune System and Breeding for Disease Resistance
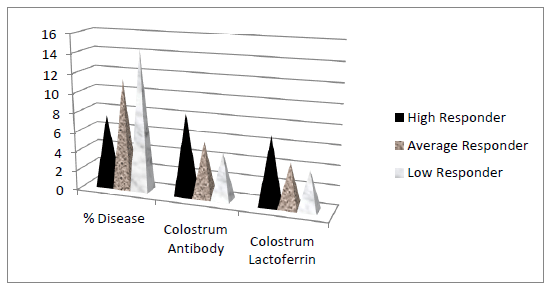
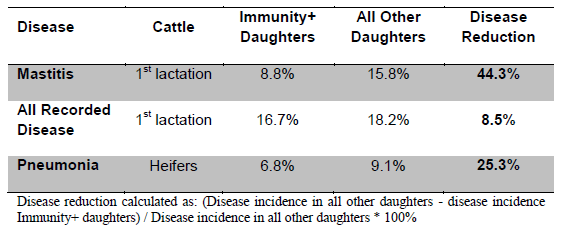
The immune system is the body’s natural defense against infectious disease and cancer. This system has the ability to customize a protective response against a set of diverse pathogens. It can detect danger signals emitted from foreign agents or defective host cells and deliver defensive mechanisms to destroy bacteria, viruses, fungi, worms and cancer cells. These defensive mechanisms are delivered via a collection of genetically regulated cells and molecules that control invading micro-organisms, including those that cause bovine mastitis, metritis, pneumonia, Johne’s disease, BLV and so on. Beneficial health advances, such as use of vaccination, antibiotics, and blood transfusion, to name a few, are based on sound knowledge of the immune system. Additional knowledge of specific immune response genes, such as those within the Major Histocompatibility Complex (MHC), has revolutionized the field of organ transplantation and provided insight into genetic regulation of disease resistance in all mammals. Studies by our group and others have focused on evaluating host defense mechanisms of livestock as indicators of specific and broad-based inherent disease resistance. Genes such as those within the MHC are known to play a key role but there are thousands of genes required to regulate this intricate system. However, using well established quantitative genetic principles, it is possible to identify individuals with enhanced immune response phenotypes following exposure to a specified set of test antigens. Phenotypic characteristics (measureable traits) are composed of two parts – genetic plus environmental components. By comparing the phenotypic traits or responses of interest in related individuals the genetic component of the phenotypic values can be determined. Knowledge of the genetic component allows computation of the heritable fraction (heritability) and the estimated breeding value (EBV) for each individual for the trait of interest. This approach has been used for decades to improve livestock traits, including milk production. The HIR technology uses a similar approach to identify cattle with the highest EBVs for immune response. The heritability of immune response traits (i.e. the genetically inherited component of the trait) are sufficiently high to allow for improvement via genetic selection (Abdel-Azim et al 2005, Thompson-Crispi et al 2012a). Early research by the Mallard group showed health and production benefits following genetic identification of both cattle and pigs with increased immune responses (Mallard and Wilkie 2007). In dairy cattle, this included lower occurrence of mastitis in high immune responders in 2 out of 3 Canadian herds tested, as well as improved response to vaccination and colostrum quality (Wagter et al 2000). A more recent study of 58 dairy herds that were part of the Canadian Bovine Mastitis Research Network (CBMRN) also indicated that cows identified as having high immune responses had lower mastitis incidence, and mastitis that did occur was of lower severity than in cows classified as average or low responders (Thompson-Crispi et al 2013). This included the incidence of both E. coli and S. aureus mastitis, as well as other causative bacteria. Another study on a large US dairy substantiated these findings by demonstrating reductions in the incidence of mastitis, ketosis, metritis and retained placenta of cows with both high antibody and cell-mediated immune responses compared to average or low responders based on their EBVs for these traits (Thompson-Crispi et al 2012b). In general high responders have about half the disease occurrence of low responders (Figure 1). Similarly, daughters of Immunity+ sires are now seen to have lower disease and higher LPI and TPI scores (Tables 1 and 2, Data Courtesy of Jay Shannon, Semex Alliance). Specifically, in a large US dairy, first lactation daughters of Immunity+ sires had an overall disease reduction of 8.5% compared to daughters from sires with unknown immune response status. In fact, daughters of Immunity+ sires had 44% less mastitis and 25% less calf pneumonia. The HIR method can be used to test immune response in dams, sires or calves as early as 2 months of age or older. Since the immune response traits used in the HIR test have heritability values similar to that of milk production traits (~25%), it is possible to make genetic gains in immune response relatively quickly, as has been the case for various production parameters.
Figure 1 - Disease Occurrence (%), Colostral Antibody (ELISA Optical Density x 3) and Colostrum Lactoferrin (ug/ml) in High, Average and Low Immune Responder Cows (Wagter et al 2000, Thompson-Crispi et al 2012b, 2013, and Fleming et al 2013)
High Immune Response, Milk Production and Colostrum Quality
To date, multiple studies have shown that breeding for high immune response using the HIR technology does not negatively affect production traits, and indeed may improve overall herd life. In the CBMRN study there were no differences in 305 day milk yield, protein yield, fat yield or overall lifetime profitability in HIR cows compared to low or average responder cows. It was also worth noting that when relative immune response breeding values of sires were estimated from this study, beneficial associations were noted between immune response and herd life and some reproductive traits, as well as between high cell-mediated immune response and milk yield (Thompson-Crispi et al 2012a), suggesting these traits may improve by breeding for enhanced immune response.
Importantly, high immune responders also produce colostrum of enhanced quality. Early on it was shown that colostrum from high responders contains more specific antibodies induced to experimental test antigens compared to average and low responders (Wagter et al 2000)(Figure 1). This finding was further validated by a recent study showing that antibody to a test antigen in blood was positively and significantly correlated with antibody in colostrum or milk during both early lactation (calving to 5 days post partum) and, late lactation (around 280 days in milk on average). It is worth noting the positive and significant correlation found between antibody in colostrum of the mother and in the blood of their calves two days after administration of colostrum. Thus there was a higher concentration of specific antibody compared to calves of average or low responders (Wagter et al 2012). In addition, the previous study by Wagter et al (2000) reported that cows with high antibody responses to a test antigen responded better to a J5 mastitis vaccine (O111:B4) in comparison to average and low immune responders. These studies demonstrate that high immune responders have an ability to provide enhanced protection of the udder against mastitis during lactation and the dry period, and a greater ability to provide protection to their calf against pathogens that cause disease. The results of these two studies also indicate the potential to improve immune responses among low or average responders through additional or tailored vaccination programs. Depending on immune response classification of cows as high, average or low responders, vaccination strategies can be customized to prevalent pathogens in the environment of cows and their calves. Therefore, the HIR technology provides both a new genetic and management tool to improve herd health. Both of these studies underscore the utility of a milk sample in lieu of blood to evaluate antibody response in lactating dairy cattle in either early or late lactation.
Additionally, it has recently been shown that cows classified as high antibody-mediated immune responders have greater concentrations of total immunoglobulin (IgG) (Fleming et al 2013, unpublished results) and lactoferrin in colostrum compared to low responders (Fleiming et al 2013)(Figure 1). Lactoferrin is an 80 kDa innate host defense glycoprotein found in various biological secretions including bovine colostrum (1.0-5.0 mg/ml) and milk (0.010-0.35 mg/ml). It is known that both lactoferrin and total IgG molecules perform antimicrobial activities against a vast array of pathogens including bacteria, which may partly explain the lower incidence of mastitis in high immune responders. Lactoferrin has also been shown to exert anti-inflammatory (Britigan et al 1994) and anti-cancer activities (Tung et al 2013). For these reasons, lactoferrin and total IgG have been extracted and purified from bovine milk for supplemental use in infant formula and milk replacer. It is also important for producers to bank colostrum with increased concentrations of total IgG from high immune responders for use in unthrifty calves, since high quality colostrum has been associated with enhanced calf health and less disease (Furman-Fratczak et al 2011).
Epigenetic Aspects of Immune Regulation
Epigenetics is a field of study that explores the interaction between genes and environment through characterization of modifications to DNA structure. The epigenome is comprised of alterations to DNA, such as DNA methylation or histone modifications, and through micro RNA that control the activation or repression of target genes (Frésard et al 2013). Generally, decreased DNA methylation at gene regulatory regions, such as promoters, is associated with activation of that gene. For that reason, the epigenome has the potential to explain those differences between individuals that cannot be accounted for by genetic variation. Environmental factors, such as stress, maternal nutrition, hormones, and toxins can influence epigenetic modifications and subsequent phenotypes for not only that generation but also subsequent generations of offspring (Skinner 2011). A key example of this lies in human history; the Dutch famine at the end of World War II left many pregnant women without adequate nutrition throughout pregnancy. This has since been associated with chronic disease later in life for infants born to those mothers (Heijmans et al 2008). It appears that early life experiences, both in utero and postnatally, can have a large effect on adult phenotypes. This is likely also true in dairy cattle. Although there have been numerous studies showing environmental impact on the epigenome the question of epigenetic influence on relevant dairy phenotypes and its place in animal breeding strategies remains. The immune system is a dynamic, responsive system which requires the rapid activation of immune factors, such as cytokines, to direct an appropriateimmune response to a specific pathogen. Our group has previously shown that DNA methylation patterns (epigenetic modifications) correlate with cytokine (hormones of the immune system) production associated with antibody (the cytokine Interleukin- 4) and cell-mediated (the cytokine interferon-gamma) immune responses of dairy cattle (Paibomesai et al 2012). Most recently, we have been examining the association of DNA methylation in high antibody versus high cell-mediated immune responder dairy cows and showed differences in DNA methylation at cytokine gene control regions between these two phenotypes. Those individuals that produced more interferon-gamma cytokine when stimulated with a general stimulus had significantly less DNA methylation at regulatory regions of the interferon-gamma locus before and after stimulation. This suggests that those cattle with heightened cell-mediated immune responses were epigenetically predisposed to rapidly produce more cytokine as compared to the high antibody responder cows (Paibomesai et al 2013, unpublished results). Further studies need to be completed to determine the overall influence of epigenetic marks on individual cow phenotypes, and whether this can be transmitted through generations. One of the most intriguing areas for future research will involve determining how epigenetics can be used as a tool in animal breeding.
Current Research on Genomic Testing for Immune Response
Using the HIR technology and the quantitative genetic method to identify and select individuals with higher breeding values for immune response traits is a novel genetic tool that does not require molecular genetic information or manipulation of the animal and therefore avoids current controversies surrounding production of GMOs. Nonetheless, the advantages of modern molecular genetics techniques are being employed at the laboratory level to identify and study favourable animal genotypes. In fact, the Illumina SNP 50K bovine BeadChip has been utilized to evaluate differences in cows with high or low antibody-mediated and cell-mediated immune responses. In this study, 186 genetic markers that are part of 11 genetic pathways were shown to differ between these cows based on antibody responses, and 17 genetic markers were associated with cellular responses (Thompson-Crispi et al 2012c). Genetic pathways included those within the bovine MHC, an important immune response gene cluster. Results of this work were also validated in the Immunity+ sires and make it possible to estimate genomic breeding values for immune response to improve health in subsequent generations. Although there is still more research to be done in the genomics arena, such as creating a large reference population with immune response phenotypes and genotypes, results from the current studies suggest that in the future it may be possible to identify high or low immune responders from a simple DNA sample.
Practical Implication of the High Immune Response (HIR) Technology
Breeding companies are increasing their focus on health traits (Koech et al 2012) . Currently, this includes distribution of sire proofs to improve mastitis based on physical udder characteristics and somatic cell score (SCS). In the future, information on clinical mastitis may be added to the sire proof. However, these indicators focus mainly on one disease, mastitis, and while this is one of the most costly diseases to the dairy industry there is a need to tackle other disease issues, such as bovine leukosis virus , Johne’s disease and infectious plantar dermatitis. The HIR technology is currently the only commercially available test that can be used to identify and breed cattle for improved general disease resistance. The HIR test measures both antibody and cell-mediated immune responses. Antibody is particularly relevant to the control of extracellular pathogens, such as mastitis- causing bacteria; whereas, cell-mediated immune responses are particulary relevant to control intracellular pathogens, such as viruses and facultative intracellular bacteria, such as mycobacteria. These so called adaptive responses of the immune system possess the attributes of pathogen specificty and immunological memory that allows the individual to produce a greater and more rapid response on second and subsequent exposures to the pathogen. In some cases, these two types of adaptive immune responses can produce lifetime immunity. High immune responders have improved disease resistance and also produce colostrum with greater specific antibody, total immunoglobulin and innate host defense molecules, specifically lactoferrin. Additionally, high responders have the ability to respond better to commercial vaccines such as J5 E. coli. These synergistic effects are beneficial when vaccinating cows prior to calving in order to ensure the production of high quality colostrum with increased amounts of specific antibodies to relevant pathogens within a particular herd. The HIR technology is uniquely designed with demonstrated ability to improve resistance to a diverse range of economically important pathogens, improve colostrum quality and enhance response to certain commercial vaccines. This ultimately translates to increased dairy food quality and safety for the consumer, and improved profitability to the producer. These key research results and practical implications are what paved the way for the HIR/Immunity+ technology to win one of the Dairy Management Innovation Awards at the 2013 World Dairy Expo in Madison, Wisconsin. The future is expected to hold new HIR applications for dairy cattle and other species.
Conclusions
HIR/Immunity+ is an award winning patented technology that allows dairy producers to safely and effectively improve the health of their herd. High immune responders have about half the disease occurrence of low responders, along with improved colostrum quality and response to commercial vaccination. HIR is both a management and breeding tool. Semex has an exclusive license to use the HIR technology to identify sires with the elite high immune response classification. These are marketed as Immunity+ bulls. Immune response traits are heritable and using Immunity+ bulls allows improved disease resistance genes to be passed on to subsequent generations of offspring.
HIGH IMMUNE RESPONSE (HIR) ANIMALS ARE NATURALLY IMMUNE
HIR is a patented evaluation technology developed to identify dairy cattle with high immune response capability.
Identification is safe, fast and effective.
Benefits include:
- Lower disease occurrence and severity
- Reduced treatment and veterinary costs
- Increased response to vaccines
- Increased colostrum quality
- Cattle as young as 2 months or older can be tested
- Animals only need to be tested once in a lifetime
- Testing is safe and does not interfere with other diagnostic testing
- Cost-benefit analyses show significant savings to producers who identify HIR cows and use HIR sires in their herd.
- HIR is both a management and breeding tool that can be used to improve health in future generations of dairy cattle
Acknowledgements
Contributions from all current and previous staff and students in the Mallard laboratory are gratefully acknowledged. Funding from NSERC, OMAFRA, DairyGen and Semex are highly appreciated.
Table 1. Sire Proof Data (August 2013) from Immunity+ Sires Compared to all other Sires Tested for Immune Response using the HIR Method (Data Courtesy of Jay Shannon, Semex Alliance).
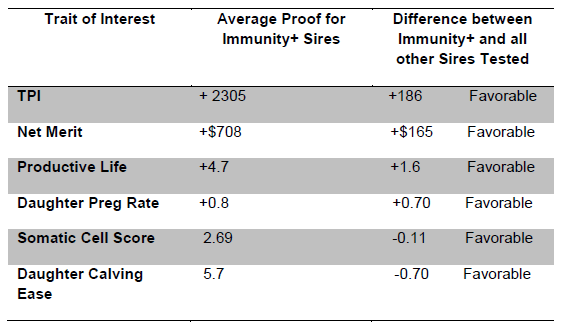
Table 2. Disease Occurrence of Immunity+ Daughters in a Large US Dairy Herd in 2013 (Data Courtesy of Jay Shannon, Semex Alliance).
References
Abdel-Azim, G.A., A.E. Freeman, M.E. Kehrli, Jr., S.C. Kelm, J.L. Burton, A.L. Kuck, and S. Schnell. 2005. Genetic basis and risk factors for infectious and noninfectious diseases in US Holsteins. I. Estimation of genetic parameters for single diseases and general health. J. Dairy Sci. 88(3):1199-1207.
Britigan, B.E., J.S. Serody and M.S. Cohen. 1994. The role of lactoferrin as an anti-inflammatory molecule. In: T.W. Hutchens, S.V. Rumball and B. Lönnerdal (Eds), Lactoferrin Structure and Function. Adv. Exp. Med. Biol. 357: 143-156.
Fleming, K., K. Thompson-Crispi, F. Miglior, M. Corredig, D. Hodgins, and B. Mallard. Bioactive Components in Colostrum and Milk of Dairy Cattle Classified as High, Average and Low Immune Responders. Dairy Cattle Breeding and Genetics Committee Report, September 10 2013, Guelph, Ontario.
Furman-Fratczak, K., A. Rzasa and T. Stefaniak. 2011. The influence of colostral immunoglobulin concentration in Heifer calves’ serum on their health and growth. J. Dairy Sci. 94:5536-5543.
Frésard, L., M. Morisson, J.M. Brun, A. Collin, B. Pain, F. Minvielle, and F. Pitel. 2013.
Epigenetics and phenotypic variability: some interesting insights from birds. Genet. Sel. Evol. 45(16).
Heijmans, B.T., E.W. Tobi, A.D. Stein, H. Putter, G.J. Blauw, E.S. Susser, P.E. Slagboom, and L.H. Lumey. 2008. Persistent epigenetic differences associated with prenatal exposure to famine in humans. Proc. Natl. Acad. Sci. USA. 105: 17046-17049.
Koeck A, F. Miglior, D.F. Kelton, F.S. Schenkel. 2012. Health recording in Canadian Holsteins: data and genetic parameters. J Dairy Sci.; 95(7):4099-4108.
Mallard, B.A., and B.N. Wilkie. 2007. Phenotypic, genetic and epigenetic variation of immune response and disease resistance traits of pigs. Advances in Pork Production 18:139-146.
Paibomesai, M.A., B. Hussey, M. Nino-Soto, and B.A. Mallard. 2013. Effects of parturition and dexamethasone on DNA methylation patterns of IFN-γ and IL-4 promoters in CD4+ T-lymphocytes of Holstein dairy cows. Can. J. Vet. Res. 77:54-62.
Skinner, M.K. 2011. Environmental epigenetics transgenerational inheritance and somatic epigenetic mitotic stability. Epigenetics. 6:838-842.
Thompson-Crispi K.A., A. Sewalem, F. Miglior F, B.A. Mallard. 2012a. Genetic parameters of adaptive immune response traits in Canadian Holsteins. J. Dairy Sci. 95(1):401-409.
Thompson-Crispi K.A., B. Hine, M. Quinton, F. Miglior, B.A. Mallard. 2012b. Short communication: Association of disease incidence and adaptive immune response in Holstein dairy cows. J Dairy Sci. 95(7):3888-3893.
Thompson-Crispi K.A. 2012c. Genetic regulation of immune responses in Holstein dairy cows across Canada. PhD thesis. University of Guelph. Guelph, Ontario, Canada.
Thompson-Crispi K.A, F. Miglior, and B.A. Mallard. 2013. Incidence rates of clinical mastitis among Canadian Holsteins classified as high, average and low immune responders. Clin. Vaccine Immunol. 20(1):106-112.
Tung, Y., H. Chen, C. Yen, P. Lee, H. Tsai, M. Lin and C. Chen. 2013. Bovine lactoferrin inhibits lung cancer growth through suppression of both inflammation and expression of vascular endothelial growth factor. J. Dairy Sci. 96:2095-2106.
Wagter, L.C., B.A. Mallard, B.N. Wilkie, K.E. Leslie, P.J. Boettcher, and J.C. Dekkers. 2000. A quantitative approach to classifying Holstein cows based on antibody responsiveness and its relationship to peripartum mastitis occurrence. J. Dairy Sci. 83(3):488-498.
Wagter, L.C., L. Cain, S. Cartwright, and B.A. Mallard. 2012. Feasibility of transferring High Immune Response (HIR) technology to the marketplace to improve the health and food quality of Canadian Dairy cattle. Proceedings of the Graduate Student Research Symposium, Ontario Veterinary College, University of Guelph. November 2012. Abstract. p. 18 http://www.ovc.uoguelph.ca/academics/graduate/documents/WebGSRSbooklet2012-forweb.pdf
WCDS Advances in Dairy Technology: Strategies to Optimize Performance. Published by the Univ of Alberta, Vol 26;247-257, 2014
Related topics:
Authors:
Poultry Health Research Network
Recommend
Comment
Share

Would you like to discuss another topic? Create a new post to engage with experts in the community.





.jpg&w=3840&q=75)