Managing Algal Blooms and the Potential for Algal Toxins in Pond Water
Published: September 14, 2017
By: Gary J. Burtle. / Extension fisheries specialist, University of Georgia, Department of Animal and Dairy Science.
Summary
If a pond looks stagnant with an algal scum, take precautions against animal exposure. Choose an algal control plan that includes early monitoring for cyanobacteria and selecting a control method. Copper or sodium percarbonate are good treatments for bluegreen algae control. Consider moving animals away from the pond or restricting access to the water when toxins are discovered. Install animal drinking water structures so that grazing animals do not drink from the pond edge. Consider a nutrient control program to prevent the pond from developing harmful algal blooms in the future. Copper or sodium percarbonate are good treatments for blue-green algae control.
Introduction
Cyanobacteria, blue-green algae, harmful algae, and toxic algae are words that can confuse people who are trying to decide on pond water safety. Certain algae can cause serious problems when they become very abundant in drought, hot weather, unusual rainfall patterns, or after nutrient accumulation in pond water. Fortunately, most ponds do not experience toxin problems or dense algal blooms. Watching for the warning signs that lead to toxic algal bloom development is the best way to prevent toxin problems.
Cyanobacteria is a descriptive name for blue-green algae, used by taxonomists and scientists. Cyanobacteria are common algae that usually float in the water column or attach to the pond bottom or another surface in the pond. Harmful algae are a very small number of species that may produce toxins, odors or off-flavors. Toxic algae is the name given to an even smaller number of species, including cyanobacteria (responsible for most cases of algal toxins), euglenoids, and dinoflagellates that produce chemicals that irritate skin, damage the liver, or affect the brain. These effects can be quite serious but are uncommon in fish ponds.
A toxin producing algae must be present at a high density and must be producing toxin for dangerous conditions to be present. Since many ponds do not have dense algal blooms, few pond owners have experience with toxic algae.
Weather conditions may be responsible for the development of toxic algal blooms. Under normal conditions, the nutrients phosphorus and nitrogen gradually move downstream from ponds after rainfall. During dry weather, pond water volume recedes, and runoff no longer flushes the pond. Reduced water volume concentrates phosphorus and other nutrients. Nutrients, heat, and sunlight allow algae to grow at a rapid rate, and dense algal blooms may develop. Certain cyanobacteria species can dominate the algal population to the point of becoming a single-species bloom. Algal bloom density can increase until a thick scum resembling pea soup or latex paint is formed.
The color of the bloom may be light blue-green, bright green, green-brown, or even chocolate. At that point, the danger from toxins is most apparent. Animals are in danger when they drink the algae along with water. Dense single-species blooms are subject to sudden die-offs. When toxic algae die, the toxin they contain is released. More often, odors and taste from chemicals produced by algae cause drinking water or fish to become unacceptable to humans.
Toxins from Algae
Algal toxins form inside cyanobacteria cells and, in most cases, are retained inside the cell until after the cell dies and is ruptured (U.S. EPA 2012). Pond water with a small amount of toxin may suddenly have a large amount of toxin after cyanobacteria are killed with an algicide or by natural causes. These events are called “sudden deaths,” “die-offs,” “turnovers,” or “death phases.” In microbiology, growth is described in phases up to the point that a limiting parameter is depleted and the colony or population dies. Depletion of a nutrient, lack of light during cloudy weather, or extremely bright sun can kill the algal bloom. Under normal circumstances oxygen depletion occurs as the algal population dies. If a toxin-producing species is present, the toxin contained inside the cells can be released into the pond water.
Many chemicals produced by cyanobacteria may be harmless, but a few are severely toxic. Earthy, musty odors coming from ponds or off-flavors in fish are caused by chemicals produced by cyanobacteria. Those are common and nontoxic events, though unpleasant. The earthy, musty odor of algae does not usually mean a toxin is present. However, even slight amounts of algal off-flavor in fish can make those fish undesirable.
Toxin producing algae do not always produce toxins, and toxin producing algae strains cannot be identified by visual means alone. Specific testing is used to identify algal toxins. The most common algal toxin is microcystin, which may cause liver and kidney damage, dermatitis, abdominal pain, or death, depending on the amount ingested. Toxins produced by algae may be toxic to mammals, fish, or all animals, depending on the chemical. Rare but dramatic animal deaths have been caused by algal toxins around the world and throughout recorded history. Most cases involve cattle, horses, or dogs because they can ingest the algae scum. Few cases of toxic algae deaths were reported in Georgia until 2012 and 2013 (Haynie et al. 2013) when drought and unsettled weather following the drought were blamed. Humans are rarely affected by algal toxins, and no drinking water limits are set for algal toxins in the United States at this time (U.S. EPA 2012).
In addition to microcystin, freshwater algae produce the toxins cylindrospermopsin, anatoxin-a, saxatoxin, or eugleonophycin. Laboratory observation of algal species that are present in pond water can indicate the probability that these toxins may be present. When suspect algae are identified, further testing for specific toxins may be needed.
Monitoring for Potential Toxic Conditions
Monitor for potential toxic conditions by frequent pond visits and careful observation. Watch for the development of algal blooms in ponds as well as animal behavior. By using algae control methods, you can minimize the chance that dense algal blooms that favor toxin production will develop. And, if algal blooms do develop, you can take steps to eliminate the danger of algal toxins.
Figure 1. Measuring visibility in pond water with 4 inches of visibility. (Images by G. Burtle, UGA CAES.)

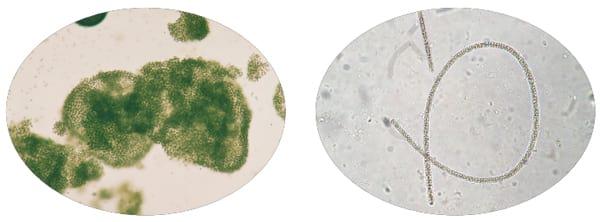
Figure 2. Algal scum on left compared to filamentous algae on right. (Images by T. Wynne and R. Sinyard, UGA Extension.)

Ponds with static water and the receding water level should be watched carefully. Water quality can decline when ponds do not collect enough runoff to cause the pond to overflow. The water level may drop, causing microorganisms, including algae, to grow more rapidly.
Pond management uses static water to make pond liming and fertilization work. It is acceptable for algal density to prevent light from penetrating more than about 12 inches into the water column. Less light penetration caused by dense algal blooms should be a warning that the pond water is enriched with nutrients. A white target called a Secchi disc is used to measure visibility into the water column (Figure 1). A plate attached to a yardstick is a good target to use for that purpose.
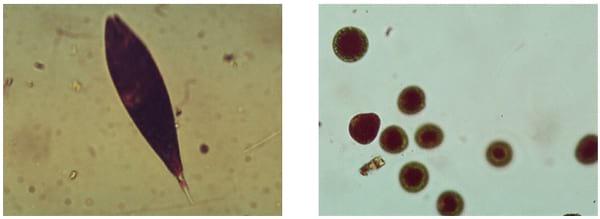
The point of concern exists when algal blooms prevent visibility of less than 12 inches into the water or if scums are observed (Figure 2). Then algal control methods should be considered, and a toxin check might be in order. Examination of a pond water sample, using a microscope, can determine if potential toxin producers are present and how abundant they are (Figures 3 and 4). If high numbers of cyanobacteria are observed, a microcystin test could be considered. Pond owners may order algal bloom testing through their local Cooperative Extension office. The test involves checking the species and number of algae present, and then requesting the toxin test if justified. If the pond water is treated to control toxic algae, a follow-up sample should be submitted for toxin testing.
Figure 3. Microscopic views of Microcystis aeruginosa and Planktothrix sp. (Images by G. Burtle, UGA CAES)
Measures to consider when evaluating cyanobacteria blooms:
1. Is light penetration into the pond water less than 12 inches? If yes, this is an early sign of an algal problem.
- Stop pond fertilization (if that practice is used).
- Consider a method to reduce algal density by using an algicide or algae-eating fish.
2. Has an algal scum begun to form? If yes, this is evidence of an algal problem.
- Consider water testing for harmful algae and toxins.
- Consider the exclusion of humans and animals until algae are reduced.
3. What could have caused the dense algal bloom to develop? Check the water supply from springs, runoff, or high volume water well.
- Allow pond water to be diluted or flushed. (Note: In severe drought, pumping water from a well may not help.)
4. Can an algicide be applied or has one been used in the past?
- Consider using chemical control to thin the algal bloom. (Note: Conditions may not allow the use of chemicals.)
- Can the pond be aerated in the case of oxygen depletion caused by the algicide?
5. Would animal relocation be a good option (if algal toxins are present)?
- Fence the pond or move the animals to a better water source.
- Consider installing a water tank and hauling water to animals.
6. Can nutrients be excluded from the pond (for future algae control)?
- Maintain a vegetation buffer around the pond, fence out animals, and/or control subsurface nutrient contamination.
- Reevaluate pond fertilizer additions that may have been used for fish population management.
Figure 4. Euglena sanguinea and cysts are found in late summer and fall in Georgia. (Images by G. Burtle, UGA CAES.)
Methods for Algal Control and Water Quality Improvement
By controlling nutrients that reach the pond water, algal growth can be limited. Grazing animals should have limited access to the pond edge in order to limit manure contamination and limit pond bank erosion (Blocksome and Powell 2006, Schumacher 2002). Maintain grass buffers around the pond edge to catch or filter nutrients from animal waste or crop fertilization. Allow animals to water in limited areas of the pond, or pipe pond water to tanks located away from the pond edge, in order to limit manure entry from animal access. When ponds are over-fertilized, dense algal blooms will develop. However, after a period of time, when nutrients are kept away, the pond can recover, and stable pond ecology may return.
The Natural Resources Conservation Service has published standards (614 Watering Facility) for the construction of animal watering areas using ponds. Livestock farmers may even consider drilling a horizontal well (Welchert and Freeman 1973) under or beside the pond to allow soil filtration of pond water before animal watering. These methods prevent animals from drinking at the water edge where they would consume algal cells, which might contain toxins.
Natural flushing from rainfall must occur to dilute nutrients that enter ponds from the fish feed, fertilizer, or pastured animals. Phosphorus moves downstream through a watershed over time. Phosphorus remains in ponds in the sediment, in fish, and in plants. Water movement through the pond carries nutrients away from the pond. Ponds with a high rate of water exchange are not good subjects for pond fertilization and liming programs because of the potential for loss of lime and fertilizer. A detention time of three to four weeks is recommended for effective pond fertilization programs (Boyd and Snow 1975).
Drought conditions do not allow for regular flushing (detention greater than four weeks) and contribute nutrient accumulation. If water is available, enough to change at least 25% of the pond volume in 24 hours, flushing the pond by exchanging over one quarter of the pond water volume per month (Ghate et al. 1993) may help lower nutrient concentration even when nutrients are continually added to the pond. However, during a drought, water tables are low and pumping into a pond may not raise pond water levels but only add water to the shallow aquifer as it seeps out the pond sides.
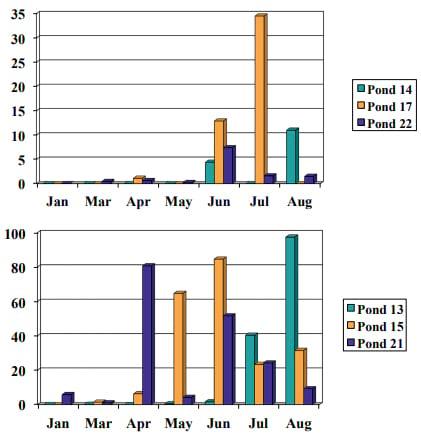
'Threadfin shad are stocked as a forage for largemouth bass and may have an effect on algal blooms. The larger algae colonies and larger algae may be consumed by shad (Drenner et al. 1984), and a shift in the species of algae may occur when threadfin shad are stocked (Green et al. 2010). About 600 threadfin shad per acre prevented dense blue-green algal blooms in fish ponds at Tifton (Figure 5). Threadfin shad should be stocked before dense blooms occur. As with most biological controls, filter-feeding fish may take several months to control the algal bloom. A disadvantage to stocking threadfin shad includes the need to restock if the shad fail to reproduce.
Figure 5. Blue-green algae cells (in millions) with threadfin shad (top) and without (bottom). (Images by G. Burtle, UGA CAES.)
Threadfin shad are stocked as a forage for largemouth bass and may have an effect on algal blooms. ). (Image by G. Burtle, UGA CAES.

Algicidal chemicals are the most common methods of controlling algal blooms. Copper chemicals and sodium percarbonate (2Na2CO3- 3H2O2) are popular choices. Copper chemicals are effective on most algae and can be relatively inexpensive. When used early and before dense algal blooms can develop, copper chemicals are very effective in preventing algal problems. However, if copper chemicals are added when dense algal blooms develop, the release of algal toxins may occur. Copper sulfate, added at 0.25 ppm, can effectively control most cyanobacteria species without killing other aquatic plants in the pond. Copper as a liquid formulation can be more easily applied than crystalline copper sulfate. See specific labels for each copper product in order to obtain proper application methods.
Repeated copper application may be needed each week until visibility into the water column is more than 12 inches. Stop copper treatments as soon as algal control is achieved. Avoid applying copper more than three weeks in a row so that desirable algae may survive. In soft water, when hardness is measured below 40 ppm, copper may be toxic to fish (Watson and Yanong 2012). Rainbow trout, koi carp, and blue catfish are more sensitive to copper than channel catfish, largemouth bass, and bluegill sunfish. So, test pond water hardness before beginning a copper treatment program. Copper will bind to the pond bottom soils within a day or two after application, leaving the water column and becoming unavailable for long term algae control.
Sodium percarbonate has an algicidal effect and also oxidative effect when percarbonate releases peroxide. When sodium percarbonate kills algae and toxins are released, toxin degradation is enhanced due to the presence of peroxide. Algal toxin degradation occurs naturally from ultraviolet light exposure and bacterial degradation (He et al. 2012, Barrington and Ghadouani 2008). In addition to peroxide, potassium permanganate and chlorine have been used to oxidize algal toxins. Activated carbon has been used to absorb algal toxins from contaminated drinking water.
Formulations of sodium percarbonate are labeled by the U.S. Environmental Protection Agency for use in the pond under trade names such as GreenClean, PAK- 27, and Phycomycin. Sodium percarbonate has been used in fish ponds at Tifton at 16 pounds to 25 pounds per acre-foot for control of blue-green algal blooms. Repeated application is necessary, since peroxide has a short residence time in pond water, degrading to oxygen and water molecules after 24 hours.
Alum and lanthanum have been used to reduce soluble phosphorus in ponds and lakes, binding with phosphorus and settling to the bottom. However, algal toxin reduction does not always occur with those treatments, possibly because some soluble phosphorus remains for algal growth (Lurling and Faasen 2012).
Any chemical addition to ponds may cause dissolved oxygen depletion. Hot weather increases the probability of oxygen depletion. A source of aeration should be present before chemical addition in order to reduce the danger of oxygen depletion after chemical-induced algal death.
This article was originally published in UGA Extension Bulletin 1445.
References:
Barrington, D. J., and A. Ghadouani. 2008. Application of hydrogen peroxide for the removal of toxic cyanobacteria and other phytoplankton from wastewater. Environmental Science & Technology, 42(23), 8916-8921.
Blocksome, C. E., and G. M. Powell (eds.). 2006. A handbook for waterers and watering systems: A handbook for livestock producers and landowners, Kansas State University, Agricultural Experiment Station and Cooperative Extension Service, Manhattan, KS. S-147.
Boyd, C. E., and J. R. Snow. 1975. Fertilizing farm fish ponds. Auburn University Agricultural Experiment Station Leaflet 88. Auburn, Alabama. 6 p.
Drenner, R. W., J. R. Mummert, F. DeNoyelles, and D. Kettle. 1984. Selective particle ingestion by a filter feeding fish and its impacts on phytoplankton community structure. Limnol. Oceanogr. 29:941-948.
Ghate, S. R., G. J. Burtle, and G. J. Gascho. 1993. Reuse of water from catfish ponds. In K.L. Hatcher (ed.), Proceedings of the Georgia Water Resources Conference, Institute of Natural Resources, The University of Georgia, Athens, Georgia. 391-393.
Green, B. W., P. Perschbacher, G. M. Ludwig, and S. E. Duke. 2010. Threadfin shad impacts phytoplankton and zooplankton community structures in channel catfish ponds. Aquaculture Research. 41:e524-e536.
Haynie, R. S., J. R. Morgan, B. Bartelme, B. Willis, J. H. Rodgers Jr., A. L. Jones and S. B. Wilde. Harmful algal blooms and toxin production in Georgia ponds. Proceedings of the Georgia Water Resources Conference. Athens, Georgia. April 2013. 5 p.
He, X., M. Pelaez, J. A. Westrick, K. E. O’Shea, A. Hiskia, T. Triantis, T. Kaloudis, M.I . Stefan, A. de la Cruz, and D. D. Dionysiou. 2012. Efficient removal of microcystin-LR by UV/C-H202 in synthetic and natural water samples. Water Research 46:1501-1510.
Lurling, M., and E. J. Faasen. 2012. Controlling toxic cyanobacteria: Effects of dredging and phosphorus-binding clay on cyanobacteria and microcystins. Water Research 46:1447-1459.
Natural Resource Conservation Service. 2010. Watering Facility. Conservation Practice Standard 614. USDA NRCS, NHCP. 3 p.
Schumacher, J. 2002. Surface water pollution from livestock production. ExEx 1010 Extension Extra. South Dakota State University. College of Agriculture and Biological Sciences. 2 p.
Sigler, W. A., and Bauder, J. 2004. Suitability of water for livestock fact sheet. Montana State University Extension Water Quality Program and Department of Land Resources and Environmental Sciences. http://watergua litv.montana.edu/docs/WELL EDUCATED/fact sheets/Livestock Suitability.pdf.
U.S. EPA. 2012. Cyanobacteria and cyanotoxins: Information for drinking water systems. United States Environmental Protection Agency Office of Water, 4304T, EPA-810F11001. 9 p.
Watson, C., and Yanong, R. P. E. 2012. Use of copper in freshwater aquaculture and farm ponds. Fact Sheet FA-13 of the Department of Fisheries and Aquatic Sciences, University of Florida, Florida Cooperative Extension Service. 2 p.
Welchert, W. T., and Freeman, B. N. 1973. Horizontal Wells. Journal of Range Management 26(4): 253-256.
Related topics:
Authors:

Recommend
Comment
Share
Recommend
Reply

Would you like to discuss another topic? Create a new post to engage with experts in the community.







