Supplementation of cows with organic selenium and the identification of selenium-rich protein fractions in milk
The essential trace element selenium (Se) is fundamental to mammalian health, influencing growth, immunity, muscle and neuromuscular function and fertility. Westernised cultures, including some European nations, have identified suboptimal intakes of Se according to recent accumulated survey data, a result that may be exacerbated by some modern agricultural practices (Rayman, 2000).
A similar situation applies in Australia and New Zealand where Se-deficit status has been identified in farm animals and the human population (Thomson and Robinson, 1980; L. Daniels, personal communication).
Recently the potential of organic Se as an anticancer agent was shown when provided at supranutritional levels (200 μg Se) in the form of Se yeast to humans (Clark et al., 1996).
The intervention trial in middle-aged men in the southwestern USA showed fewer prostate, colon and lung tumours in those who had taken the daily Se supplement for a 7.5 year period, the total period of observation being over 10 years. The anticancer response was evident in those in the lowest tertile of Se serum concentration at the start of the trial (Rayman, 2000; Combs et al., 2001). While the result with regard to these cancers was not the primary aim of the study, it provided important evidence of the potential of this trace element in cancer prevention when supplemented in organic form at levels exceeding (about 2 to 3 times) that recommended for meeting nutritional requirements.
A study in southeastern Finland, where Se was provided in fertiliser on the pasture over a number of years, showed that dairy foods and eggs contributed approximately one third of the Se intake (J. Salonen, personal communication), other major sources coming from meat and grains. Selenium blood levels significantly increased in the population over the period of land fertiliser application. The contribution of dairy foods is not normally reported, as milk levels can often be very low. However, Se in the form of sodium selenite is often supplemented in diets fed milking cows where a selenium deficit is expected, usually with the health and productivity of the cows in mind. Selenium yeast provides Se in an organic form (mainly as L-selenomethionine), which has been proposed as a safer and more effective supplement that would enhance Se incorporation into tissue proteins (Jacques, 2001).
Dairy proteins, caseins and whey proteins, are major sources of sulphur amino acids with caseins being particularly rich in methionine (casein-2.9% methionine, 0.3% cysteine) while the whey proteins are also rich in cysteine (whey protein concentrate- 2.1% methionine, 2.3% cysteine) (McIntosh et al., 1995). They may therefore provide a potentially useful vehicle for dietary selenoprotein enrichment, because selenomethionine is non-specifically incorporated into protein in place of methionine.
Selenomethionine would appear to be an optimal form for Se supplement of animals and humans, since it is the form in which Se is commonly presented in foods, and is effectively stored in body protein (Schrauzer, 2000). A significant opportunity exists to improve human Se status via dairy foods, if it can be established that such an approach benefits both animal and ultimately human health.
Materials and methods
In the first experiment, nine mature mid-lactation Friesian-Holstein cows in a 300 cow herd at the SARDI Flaxley Agricultural Research Centre (Flaxley, South Australia) were given 0, 2 or 6 mg organic Se daily in the form of selenised yeast (Sel- Plex™, Alltech Inc.) for a 12 week period. The cows in this study were of matched age, milk production, milk somatic cell count and stage of lactation and were grazed on summer-irrigated pasture during the day. A preliminary period of six weeks preceded this study, where an inorganic Se supplement (as selenite) being used was removed from the grains fed during milking. Cows were previously receiving a daily supplement of 6 mg Se, with an additional 2 mg Se present in the daily grain concentrate and pasture consumed. Sel-Plex™ was mixed with crushed barley and lupin grain (3:2) and pelleted. The animals were electronically recognised at each milking and a computer automated system delivered the correct amount of Se supplement to each animal.
Milk samples were taken periodically (mostly weekly) and assayed for Se concentration at the Adelaide laboratory of CSIRO Health Sciences and Nutrition. Somatic cell counts were performed by Herd Improvement SA Co. Ltd. Bulk samples were used to isolate the protein fractions, casein and whey, or were processed to skimmed and whole milk powders. These milk samples and fractions were then assayed for Se concentration and protein levels.
A second feeding period was undertaken with the aim to supplement the animals with sufficient Sel- Plex™ to provide a bulk milk sample for extraction of Se-rich casein for use in a colon cancer trial using a rat model. A level of 25 mg Se per day was chosen to provide sufficient Se in milk to achieve a goal of 2-3 ppm Se within the casein component of a rodent diet. Twenty cows were supplemented for 16 days to produce 3000 L of milk for casein isolation and purification, which provided enough casein for a five month rat colon cancer trial. Each batch was collected over 2.5 days and shipped at ≤4 °C to Food Science Australia in Melbourne for processing into casein isolate.
Selenium-rich casein was assayed for Se and protein concentration. Selenium was analysed by Atomic Absorption Spectroscopy (Varian model Spectra AA-400, Mulgrave Vic, Australia) using a modified analytical method No. AA-44 (involves hydride generation). Briefly, samples were digested in concentrated nitric acid overnight at room temperature, and then temperature increased to 130 °C for 1 hr, then to 150 °C for a further hour. A small volume of concentrated perchloric acid was added and the temperature increased to 170 °C for 1 hr or until digest solution was clear. The digest was cooled and washed twice with deionised water, then temperature was brought back up to 170 °C, to remove any traces of nitric acid. Finally, concentrated perchloric and 40% hydrochloric acids were added and the solution heated to 90 °C for 30 min to convert all Se from valency (VI) state to (IV) state for reading.
Protein isolation and purification of the 3000 L bulk milk samples was carried out by Food Science Australia’s Highett dairy technology research laboratory in Melbourne. This involved the acidification of whole milk and the subsequent precipitation of the milk protein fraction, which was re-precipitated and washed to give a high quality protein casein product. Proteins were assayed using a gas chromatographic nitrogren assay method (Carlo Erba NA 1500, Italy). Protein was calculated using the factor 6.25.
All animal work was approved prior to commencement by the Primary Industries & Resources SA, and CSIRO Health Sciences and Nutrition animal experimentation ethics committees.
The results in the dairy cow study were statistically assessed using one way ANOVA and Tukey’s post hoc test or with Students t-tests, significance being indicated at P≤0.05. Results: Selenium content of milk and protein fractions from cows given 0, 2 or 6 mg Se from Sel-Plex
MILK SELENIUM CONCENTRATIONS
During the six week preparative period in which all Se supplementation was stopped (a supplement of 6 mg Se as selenite in mineral mix had been fed), Se levels in milk dropped from a background of about 15 μg/L to a baseline of 7 μg/L. There was a significant and rapid increase in milk Se concentration with the introduction of 2 and 6 mg Se daily supplements as Sel-Plex™ (Figure 1). The mean values of each treatment (± SD) were: 0 mg Se, 6.9 ± 1.2 μg/L; 2 mg Se, 15.0 ± 4.2 μg/L (differed from 0 mg, P<0.05); and 6 mg Se, 25.2 ± 9.9 μg/L.
Each value represented three cows/treatment sampled on nine occasions throughout the 12 week period of supplementation. Milk Se concentration was significantly increased with 6 mg Se relative to 0 mg (P<0.001), and relative to 2 mg (P<0.01). Milk Se concentrations peaked within one week of introducing the supplements.
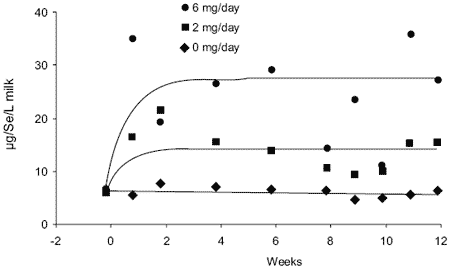
Figure 1. Mean milk Se concentrations over 12 weeks of supplementing at 0, 2 or 6 mg Se per day as Sel-Plex™.
DAILY MILK YIELD
There was no significant influence of Se intake on daily milk output over the course of the study (mean output over study in 9 cows was 26 ± 3 L/day.
There was a trend toward a slight decrease in milk production over the 12 week period in all groups, consistent with the stage in the lactation of the animals.
MILK WHITE CELL COUNTS
The mean milk somatic cell (leucocyte) counts for the individual cows throughout the supplementation period were: 0 mg Se, 155,000 (± 144 x 103) cells/ ml; 2 mg Se, 136,000 (± 61 x 103) and 6 mg Se, 85,000 (± 40 x 103), each mean representing 21 samples from three cows and seven milk samples collected over a 10 week period (Figure 2). There was no significant difference between groups given 0 and 2 mg/day in the somatic cell counts. However, the mean white cell count for the 6 mg Se/day group was significantly lower than for the 0 and 2 mg Se/ day group data combined (t-test, P<0.05).
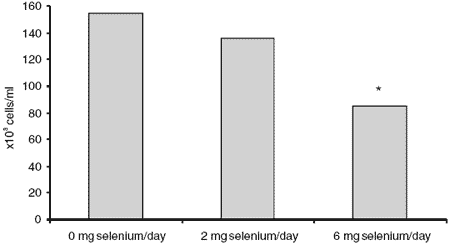
Figure 2. Mean milk somatic cell counts (leucocytes) over the 12 week supplementation period. (Values for the 6 mg Se treatment differed from the 2 mg and 0 mg Se treatments, P<0.05).
SELENIUM CONCENTRATIONS IN ISOLATED PROTEIN FRACTIONS OF MILK
Isolation of the casein and whey protein fractions from milk was undertaken at the Food Science Australia Dairy laboratory at Highett Victoria and at CSIRO Health Sciences and Nutrition in Adelaide.
The isolated milk protein fractions were analysed for Se and protein concentration. The casein fraction contained 50% of the Se in milk while the whey protein extracted contained only 10% of the total Se. A milk sample containing 17.5 μg Se/L was used to isolate casein, which contained 362 μg Se/kg and had a crude protein content of 63.5%. Whey protein concentrate contained 212 μg/kg and had a protein content of 58%. Comparative values for commercial products from New Zealand and Australia are shown (Table 1). It is of interest to note that New Zealand casein contained approximately double the Se concentration of Australian casein, with similar protein concentrations.
Table 1. Selenium content of protein isolates from Holstein- Friesian cows given Sel-Plex and those from commercial sources.
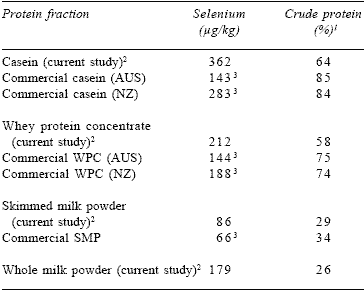
1N x 6.25
2Selenium content of original milk: 17.5 μg/L.
3Values refers to mean of 2 or 3 samples only.
SELENIUM CONTENT IN MILK AND MILK PROTEIN FRACTIONS OF COWS GIVEN 25 mg SE DAILY FROM SEL-PLEX™
When dietary Se content was increased to supply 25 mg Se/day, milk concentrations plateaued at about 150 μg/L, ie. a level about 10-fold that of Adelaide city milk (Figure 3). No clinically detectable signs of excess Se were observed in cows fed 25 mg organic Se daily, albeit this was only maintained for 16 days.
Processing of the 3000 L of milk into casein took place at the dairy technology laboratory at Food Science Australia at Highett, Victoria. The milk concentration averaged 130 μg Se/L, the isolated casein contained 2,317 μg/kg Se with a protein content of 53.8%. Fifty two kg of Se-rich casein powder were obtained for a rat colon cancer study to examine the influence of selenised dairy protein on cancer expression.
Rat colon cancer (azoxymethane induced) study protocol
In an ensuing rat trial, four treatment groups of 25 male rats of the same age and mean group weight were fed a non-supplemented Se (AIN76A) diet for four weeks prior to, and for two weeks during azoxymethane induction. They were then placed on four designated treatment diets for another 24- 28 weeks. The treatments were 1) no Se supplement, 2) 4 ppm Se as Se yeast, 3) 1 ppm as Se yeast, 4) 1 ppm Se as Se-rich casein.
Discussion
Rapid (within 2-3 days) and significant increases in milk Se concentration were observed in this study, with higher concentrations being obtained with the organic Se supplements than were seen with an equivalent inorganic supplement in first milk samples received from cows used in the studies at Flaxley.
The results were consistent with reports by Ortman and Pehrson (1997; 1999).
In Australia some dairy herds receive a recommended supplement of 2-3 mg Se per day, mostly as selenite. Metropolitan Adelaide milk has been shown to contain 13-15 μg/L. In contrast, dairy herds with no Se supplement had approximately 7- 10 μg/L of Se in milk (our data), suggesting a possible doubling of milk Se results from this feeding practice during milking. A higher response was seen in the milk of Jersey cows in a study at Maffra, Victoria where 3 mg Se daily was supplemented (also as Sel-Plex™) over eight weeks, the milk concentration reaching 65 μg/L (J. Batchelor, personal communication), which is more than double that seen with 6 mg Se supplementation in our Holstein-Friesian cow study. The reason for this difference is not known, but may reflect background Se status, or a milk cow breed difference. Daily milk production was less than half that of the Friesian herd.
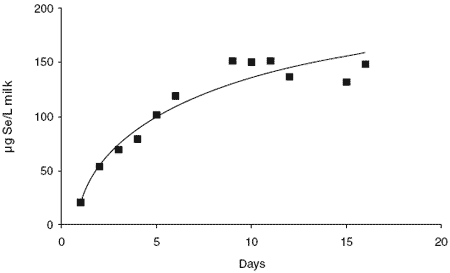
Figure 3. Daily selenium concentration of milk from 20 cows supplemented with 25 mg Se/day as Sel-Plex™.
The level of Se in commercially produced casein and whey protein fractions from New Zealand assayed at approximately double that of Australian samples, which may reflect an increased use of Se supplementation in the dairy industry in New Zealand, the commercial product assayed possibly coming from a different breed, pasture fertiliser application or organic yeast Se being used in preference to selenite. Limited numbers of commercial samples have been assayed, and more data are needed to confirm this observation.
When Se was supplemented at 2 and 6 mg/day over 12 weeks, milk production was not altered.
However, the 6 mg Se level significantly decreased numbers of white blood cells in the milk. This may be a desirable outcome for the reduction of udder infection and mastitis in dairy cattle, as has also been reported by Malbe et al. (1995). However, these values are derived from small numbers of cows and deserve more investigation as to their wider applicability.
The study of different forms of Se used to achieve dietary adequacy and anticancer effects have recently been reviewed (Ip, 1998; Schrauzer, 2001).
Schrauzer summarized the attributes of the L-isomer of selenomethionine in mammalian nutrition. Higher levels of Se supplements (supranutritional) are required to prevent cancer than are needed to address nutritional requirement, as shown by the study of Clarke et al. (1996). Given that the anticancer response was greater in individuals with lower initial Se levels, the relevance of current Se status is of interest, especially since Se status in countries such as Australia and New Zealand is typically lower than in the US. Issues regarding the efficacy of Se sources in relation to critical cancer sites, and avoidance of Se intoxication are topics of ongoing investigation and concern.
While there appear to be some differences of opinion regarding the best form to provide Se in anticancer strategies (Medina et al., 2001), the presence and availability of Se as Lselenomethionine in plant and yeast sources is consistent with a view that it is an appropriate form for supplementation (Schrauzer, 2000). An alternative form, Se-methylselenocysteine, which is more characteristic of Se in plants which are Se accumulators, has been recommended (Ip, 1998).
This form has also been reported to be effective in colon cancer prevention at a concentration of about 1 ppm, as predicted by a significant reduction in aberrant crypt foci (ACF), early preneoplastic markers (Finley and Davis, 2001). Incidentally, they reported that selenomethionine in high Se wheat when fed at 1 ppm was as effective as the broccoli form in prevention. More recently Mukerjee et al. (2001) have reported an effective reduction of colon ACF in a rat MNNG model with selenomethionine supplemented in water at 8 ppm Se. This may, however, approach toxic levels with chronic administration.
Acknowledgements
The authors wish to thank the Australian Dairy Research and Development Corporation for the support provided for this research. Dr. Neville Gregory and Mr. Owen Taylor of SARDI Flaxley Agricultural Centre for their invaluable assistance with the dairy herd management and Mr. Kirthi De Silva of Food Science Australia for protein isolation and purification. Mr. Michael Shields of Alltech Pty Ltd is acknowledged for useful technical information provided.
References
Authors: GRAEME H. MCINTOSH and PETER J. ROYLEClark, L.C., G.F. Combs, Jr., B.W. Turnbull, E.H. Slate, D.K. Chalker, J. Chow, L.S. Davis, R.A. Glover, G.F. Graham, E.G. Gross, A. Krongrad, J.L. Lesher, H.K. Park, B.B. Sanders Jr., C.L. Smith and J.R. Taylor. 1996. Effects of selenium supplementation for cancer prevention in patients with carcinoma of the skin. A randomized controlled trial. J. Am. Med. Assoc. 276(24):1957- 1963.
Combs, G.F., L.C. Clark and B.W. Turnbull. 2001. An analysis of cancer prevention by Se. BioFactors 14:153-159.
Finley, J.W. and C.D. Davis. 2001. Selenium from high-Se broccoli is utilised differently than selenite, selenate and selenomethionine, but is more effective in inhibiting colon carcinogenesis. BioFactors 14:191-196.
Ip, C. 1998. Lessons for basic research in Se and cancer prevention. J. Nutr. 128:1845-54. Jacques, K.A. 2001. Selenium metabolism in animals: the relationship between Se form and physiological response. In: Science and Technology in the Feed Industry. Proceedings of Alltech’s 17th Annual Symposium (T.P. Lyons and K.A. Jacques, eds), Nottingham University Press, Nottingham, UK. Pp. 319-348.
Malbe, M., M. Klassen, W. Fang, V. Myllus M. Vikerpuur, K. Nyholm, S. Sankari, K. Suoranta and M. Sandholm . 1995. Comparisons of selenite and Se yeast feed supplements on Se incorporation, mastitis and leucocyte function in Se deficient dairy cows. Amer. J. Vet. Med. 42: 111-121.
Medina, D., H. Thompson, H. Ganther and CIp, 2001. Se-Methylselenocysteine: a new compound for chemoprevention of breast cancer. Nutrition Cancer 40(1):12-17.
McIntosh, G.H, G.O. Regester, R.K. Le Leu, P.J. Royle and G.W. Smithers. 1995. Dairy proteins protect against dimethylhydrazine-induced intestinal cancers in rats. J. Nutr. 125:809-816.
Mukerjee, B., M. Basu and M. Chatterjee. 2001. Effect of selenomethionine on N-methylnitrosoguanidine- induced colonic aberrant crypt foci in rats. Eur. J. Cancer Prevent. 10:347-355.
Ortman, K., and B. Pehrson. 1997. Selenite and selenium yeast as feed supplements for dairy cows, a comparative study of selenite and selenium yeast (Sel-Plex) as feed supplements for multiparous dairy cows. In: Biotechnology in the Feed Industry. Proceedings of Alltech’s 11th Annual Symposium (T.P. Lyons and K.A. Jacques, eds), Nottingham University Press, Nottingham, UK. pp. 283 – 286.
Ortman, K. and B. Pehrson. 1999. Effect of selenate as a feed supplement to dairy cows in comparison to selenite and selenium yeast. J. Anim. Sci. 77: 3365 – 3370.
Rayman, M.P. 2000. The importance of selenium to human health. The Lancet 356:233-241.
Schrauzer, G.N. 2000. Selenomethionine: a review of its nutritional significance, metabolism and toxicity. J. Nutr. 130:1653-56.
Schrauzer, G.N. 2001. Nutritional selenium supplements: product types, quality, and safety. J. Am. Coll. Nutr. 20:1-4.
Thomson, C.D. and M. Robinson. 1980. Selenium in health and disease with emphasis on those aspects peculiar to New Zealand. Am. J. Clin. Nutr. 33:303-323.
CSIRO Health Sciences and Nutrition, Adelaide, SA, Australia


United States






