I. INTRODUCTION
The relationship between calcium (Ca) and phosphorus (P) in broiler nutrition is one that is complex and multifactorial. The digestion and absorption of these minerals is influenced by other dietary vitamins and enzymes as well as endogenous hormones. The modern broiler has a high requirement for Ca and P for energy metabolism and skeletal development. Deficiencies in Ca, P or both nutrients can have negative effects on bird performance as well as skeletal health. Due to the selection for rapid growth in broilers, some indirect welfare consequences have arisen; of particular importance are the adverse effects on skeletal health (Shim et al., 2012). Current research shows that due to the high porosity of the cortical bone it is prone to bone deformities, which can impair broiler mobility, which is considered a welfare concern.
To better understand the complex relationship between Ca and P in poultry nutrition, a novel graphical approach termed the geometric framework was used. The geometric framework allows for a graphical representation of two or more nutrients, and explores the interactive effects of these nutrients. This study aims to investigate the optimum dietary Ca and available P (av.P) concentrations for broiler performance and skeletal health using the geometric framework.
II. MATERIALS AND METHODS
All experimental procedures conducted had approval from The University of Sydney Animal Ethics Committee. A total of 600 Ross 308 day-old male broiler chicks were obtained from a commercial hatchery. Chicks were randomly allocated across 75 cages (750mm x 750mm x 510mm) with eight birds per cage and five replicate cages per treatment. The cages were housed in a temperature controlled room; temperature of 31 °C for the first five days and reduced by 0.5 °C per day until 24 °C (d21). The lighting regime was 23h:1h (light:dark) for the first five days and then 18h:6h (light:dark) for the remainder of the study (to d28). Chicks were fed a commercial starter diet for the first seven days before commencing on dietary treatments. Diets were corn-soy based and fed as a mash. Diets were formulated to one of three densities of total Ca:av.P ; high (15 g/kg), medium (13.5 g/kg) and low (12 g/kg). At each density there were five different ratios of Ca:av.P (4, 2.75, 2.1, 1.5 and 1.14:1). Birds had ad libitum access to feed and water. At day 27 the skeletal health of five birds per cage was evaluated using the latency to lie (LTL) procedure; whereby individual birds were placed into a plastic tub containing a few centimeters of tepid water and were timed until the bird made an attempt to sit down (Berg and Sanotra, 2003). On day 28 birds were euthanized via injection of a lethal dose (1ml/2kg) of sodium pentobarbitone into the jugular vein. Individual body weights were recorded and the right leg removed for tibia ash analysis. Tibia ash analysis, commonly used to assess bone mineralisation was determined as outlined by Garcia and Dale (2006).
All data were analysed using R version 2.15 (R Development Core Team, 2012). All performance data were analysed using a fixed effect model including blocking and a quadratic response surface model was fitted using the mixed model lme() function. Behavioural LTL times were analysed using Cox's proportional hazard survival analysis.
III. RESULTS AND DISCUSSION
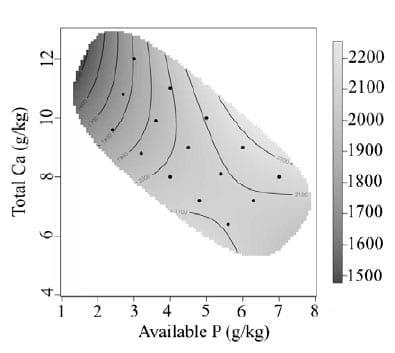
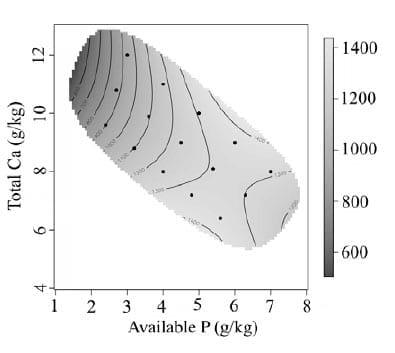
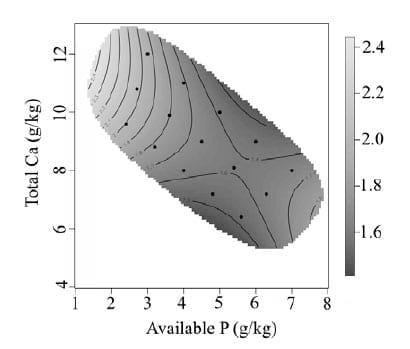
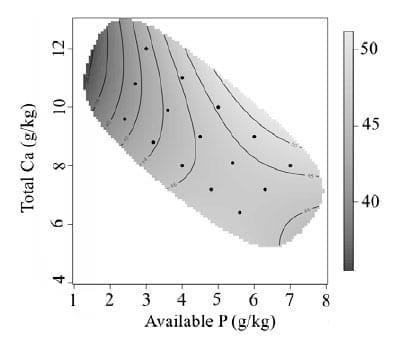
Bird performance is presented as treatment means over the experimental period of day 7 to 28. The effect of dietary Ca and av.P on feed intake is presented in Figure 1. Feed intake increased with increasing dietary av.P (P < 0.0001). Higher feed intakes were associated with high av.P and high total Ca. Feed intake was strongly inhibited by diets with low av.P, and further impaired with the addition of high levels of Ca. Body weight gain (Figure 2) was observed to follow a similar pattern to feed intake. Body weight gain increased rapidly from 1.5 g/kg to 3.0 g/kg of av.P with total Ca having little effect. Concentrations of av.P above 4.5 g/kg had little influence on body weight gain. Total Ca inclusion had a more influential effect on body weight gain at av.P levels higher than 4.5 g/kg resulting in an interaction between Ca and av.P (P < 0.01). Previous studies using low av.P (< 0.2%) and high Ca concentrations (> 0.9 %) have reported a reduction in broiler growth performance (Rousseau et al., 2012), consistent with the results observed. However, the current results contradict other studies, Dhandu and Angel (2003) and Yan et al., (2001). Both studies found no significant effect on growth performance by varying P concentrations with a fixed Ca concentration. However, the study conducted by Dhandu and Angel (2003) focused on broilers from 32-42 days, a developmental stage where broilers are less sensitive to av.P (Nelson et al., 1990). FCR from day 7-28 was shown to be poorest at low dietary av.P, with rapid improvement as av.P concentrations increased (Figure 3). The effect of dietary Ca and av.P on tibia ash is presented in Figure 4. A significant interaction between of Ca*av.P was observed (P < 0.001).
Figure 1 - Graphical representation of the interactive effect of dietary Ca and av.P on feed intake (g/b) from day 7 to 28. Av.P P < 0.0001 Ca P = 0.1167 Ca*av.P P = 0.0311
Figure 2 - Graphical representation of the interactive effect of dietary Ca and av.P on body weight gain (g/b) from day 7 to 28. Av.P P < 0.0001 Ca P = 0.0037 Ca*av.P P = 0.0016
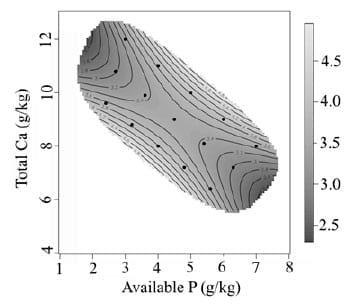
Tibia ash increased with increasing av.P inclusion to around 4.5 g/kg beyond which there was little change in tibia ash. Total dietary Ca was observed to positively influence tibia ash above 4.5 g/kg av.P, resulting in a significant Ca*av.P interaction. Latency to lie data showed that birds fed diets with an approximate 2:1 ratio of Ca:av.P were more likely to stand for longer (Figure 5). When comparing tibia ash and LTL graphs, birds with poorer tibia ash were also observed to have shorter LTL standing times. These data suggest that the birds receiving these diets have poorer bone mineralisation, which is reflected by impaired mobility and locomotion.
Interestingly, birds with higher tibia ash percentage also had shorter LTL standing times. This indicates that these birds also had impaired mobility, however, this was not associated with reduced bone mineralisation. The intense genetic selection that the broiler has undergone for increased muscle yield has also changed the conformation in the broiler stance and stride. Due to the heavier breast muscle the birds' centre of gravity is further forward, which is supported by short legs with large thigh muscles. These physiological changes have led broilers to walk more slowly, taking short wider steps and increasing contact time with the ground (Mench 2004). This abnormal gait of broilers has been associated with chronic pain (Mc Geown et al., 1999; Danbury et al., 2000). These anatomical changes to broiler locomotion, and the associated chronic pain may help to explain why birds may suffer impaired mobility, yet have high tibia ash percentage. Further investigation is needed to determine the cause of impaired broiler mobility with high av.P diets.
ACKNOWLEDGMENTS: This study was funded by Rural Industries Research and Development Corporation (Chicken Meat). The senior author is in receipt of a scholarship from the Poultry CRC. Professor Steve Simpson (University of Sydney) is thanked for his introduction to the nutritional geometric framework.
Figure 3 - Graphical representation of the interactive effects of av.P and Ca on FCR from day 7 to 28. Av.P P < 0.0001, Ca P = 0.0215, Ca*av.P P = 0.0026
Figure 4 - Graphical representation of the interactive effects of av.P and Ca on Tibia ash (DM%) at day 28. Av.P P < 0.0001, Ca P = 0.0031, Ca*av.P P = 0.0009
Figure 5 - Graphical representation of the interactive effects of av.P and Ca on Latency to lie time (in minutes) at day 27. Av.P P < 0.3531, Ca P = 0.5620, Ca*av.P P = 0.004
REFERENCES
Berg C and Santora GS (2003) Animal Welfare 12, 655-659.
Danbury TD, Weeks CA, Chambers JP, Waterman-Pearson AE and Kestin SC (2000) The Veterinary Record 146, 307-311
Dhandu AS and Angel R (2003) Poultry Science 82, 1257-1265
Garcia AR and Dale NM (2006) Journal of Applied Poultry Research 15, 103-109
Mc Geown D, Danbury TD, Waterman-Pearson AE and Kestin SC (1999) The Veterinary Record 144, 668-671
Mench JA (2004) Measuring and Auditing Broiler Welfare, CABI, Oxfordshire, UK
Nelson TS, Harris GC, Kirby LK and Johnson ZB (1990) Poultry Science 69, 1496- 1502
R: A Language and Environment for Statistical Computing, R Core Team, R Foundation for Statistical Computing, Vienna, Austria (2012)
Rousseau X, Létourneau-Montminy MP, Même N, Magnin M, Nys Y and Narcy A (2012) Poultry Science 91, 2829-2837
Shim MY, Karnuah AB, Mitchell AD, Anthony NB, Pesti GM and Aggrey SE (2012) Poultry Science 91, 1790-1795
Yan F, Kersey JH and Waldroup PW (2001) Poultry Science 80, 455-459