The effect of dietary Glutamine supplementation on performance and blood parameter, carcass characteristics, quality and characteristics meat of broiler chickens under continuous heat stress condition
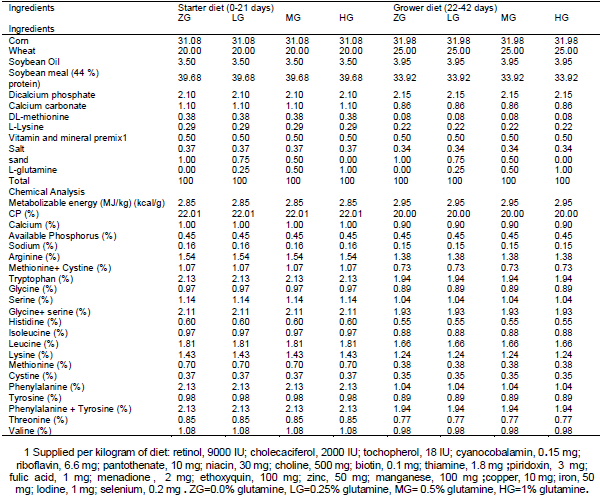
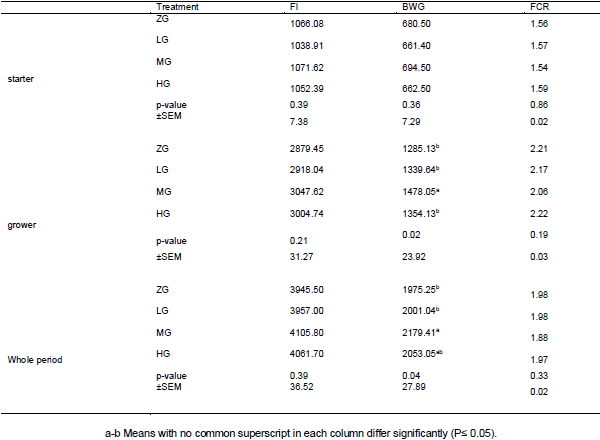
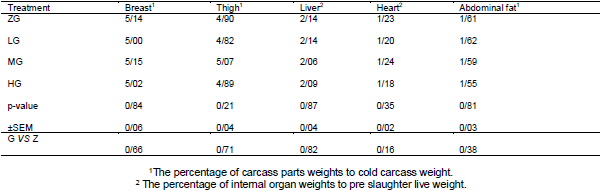
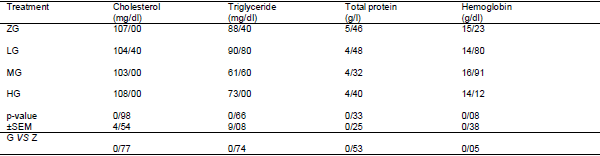
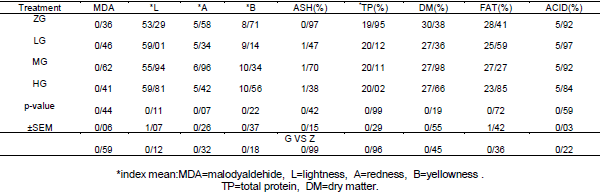
Effects of different levels of 0.0 Glutamine (ZG), 0.25 (LG), 0.50 (MG) and 1 percent (HG) were investigated on performance, quality and characteristics meat, Blood parameters and Carcass characteristics of broiler chickens under heat stress condition. No significant differences were observed between the treatments for feed intake and feed conversion ratio during the starter period, grower period, or entire experiment (P > 0.05). During the grower period, only 0.5% glutamine-fed birds had higher body weight gain than others (P < 0.05). For the entire period, body weight gain of 0.5% glutamine-fed birds was greater than that of 0.00% and 0.25% glutamine-fed ones (P <0.05).None of the blood parameter, carcass characteristics and meat quality affected by dietary treatments at week 6 of age (P>0.05). It was concluded that although dietary Glutamine consumption can improve the performance of broiler chickens during the grower and whole period under heat stress, but it doesn’t affect the performance during starter period or blood parameter at week 6 of age.
Keywords: broiler chickens, heat stress, performance, Glutamine.
Aengwanich W. 2008. Pathological changes and effect of ascorbic acid on lesion scores of bursa of fabricius in broilers under chronic heat stress. Poult Sci Assoc, 1: 62–66.
Bartell SM and Batal AB. 2007. The effect of supplemental glutamine on growth performance, development of the gastrointestinal tract and humoral immune response of broilers. J Poult Sci, 86:1940–1947.
Belay T and Teeter RG. 1996. Effects of environmental temperature on broiler mineral balance partitioned into urinary and fecal loss. Br Poult Sci. 37:423–433.
Bi N. 2007. Poultry meat color. Veterinary Kohkilooye & Boyer Ahmad.p1
Botsoglou N, Papageorgiou G, Nikolakakis I, Florou P, Giannenas I, Dotas V and Sinapi S. 2004. Effect of dietary dried tomato pulp on oxidative stability of Japanese quail meat. J Agri and Food Chem, 52: 2982-2988.
Daghir NJ. 2008. Poultry production in hot climates. second edition. CAB International., 2nd Ed.
Dai F,Gao X, Xu L, Zhang WH and Zhou GH. 2012. Effect of dietary glutamine and gama-amino butyric acid on meat colour, ph, composition and water-holding charecteristic in broilers under cyclic heat stress.J Brt Poult Sic, 471-481.
Dai SF, Gaoa F, Song SX, Zhanga WH, Xub XL and Zhoub GH. 2011. Effects of Dietary Glutamine and Gamma-Amino Butyric Acid on Performance, Carcass Characteristics and Serum Parameters in Broilers Under Circular Heat Stress. J Anim Feed Sci Tech,168:51–60.
De-lian M, Chao S, Jun-hu Y, Gong-she Y and FANG J. 2009. Effect of Aspartate and Glutamine on a Part of the Fatty Traits and the Levels of Adipogenesis Genes mRNA Expression of Broiler. Sci Agricul Sin, 7; 2513-2522.
Ebadiasl G. 2011. Effects of supplemental glutamine and glutamate on growth performance, gastrointestinal development, jejunum morphology and clostridium perfringens count in caecum of broilers, UPPSALA, 07; 1-26.
El Husseiny O and Creger CR. 1981. Effect of ambient temperature on mineral retention and balance of the broiler chicks. J Poult Sci. 60(Suppl. 1):1651.
Feenster R. 1985. High temperatures decrease vitamin utilization. Miss Poult Sci, 38:38–41.
Furlan RL, Faria Filho DE, Rosa PS and Macira M. 2004. Does low protein diet improve broiler performance under heat stress conditions? Bras Sci Avic, Vol.6. No.2.
Geraert PA, Padilha JCF and Guillaumin S. 1996. Metabolic and endocrine changes induced by chronic heat exposure in broiler chickens: Growth performance, body composition and energy retention. Br J Nutr, 75:195–204.
Hai L, Rong D and Zhang ZY. 2000. The effect of thermal environment on the digestion of broilers.J Anim Physiol Anim. Nutr. (Berl.), 83:57–64.
Kannan G, Heath JL, Wabeck CJ, Souza MCP, Howe JC and Mench JA. 1997. Effect of crating and transport on stress and meat quality characteristics in broilers. J Poult Sci, 76: 523-529.
Khempaka S, Okrathok S, Hokking L, Thukhanon B and Molee W. 2011. Influence of Supplemental Glutamine on Nutrient Digestibility and Utilization, Small Intestinal Morphology and Gastrointestinal Tract and Immune Organ Developments of Broiler Chickens. W Academ Science, Engin Techn,80:606-608.
Kitt J, Miller PS, Lewis AJ and Fischer RL. 2002. Effects of glutamine on growth performance and small intestine villus height in weanling pigs. Pages 29-32 in Nebraska Swine Rep. Univ. Nebraska, London.
Kucuk O, Sahin N and Sahin K. 2003. Supplemental zinc and vitamin A can alleviate negative effects of heat stress in broiler chickens. Biol Trace Elem Res. 94: 225–235.
Li J, Langkamp-Henken B, Suzuki K and Stahlgren LH. 1994. Glutamine prevents parenteral nutrition-induced increases in intestine permeability. JPEN. 18:303-307.
Liu T, Peng J, Xiong Y, Zhou S and Cheng X. 2002. Effects of Dietary Glutamine and Glutamate Supplementation on Small Intestinal Structure, Active Absorption and DNA, RNA Concentrations in Skeletal Muscle Tissue of Weaned Piglets during d 28 to 42 of Age. Asian-Aust. J. Anim. Sci. 2; 238-242.
McDougald LR and McQuistionTE. 1980. Mortality from heat stress in broiler chickens influd by anticoccidial drugs. Poult. Sci. 59:2421–2423.
Murakami AE, Sakamoto MI, Atali MRM, Souza LMG and Franco JRG. 2007. Supplementation of Glutamine and Vitamin E on the Morphometry of the Intestinal Mucosa in Broiler Chickens. J Poult Sci. 86:488-495.
Mussini FJ, Goodgame SD, Lu C, Bradley CD, Fiscus SM and Waldroup PW. 2012. A nutritional approach to the use of anticoccidial vaccines in broilers:glutamine utilization in critical stages of immunity acquisition. Int Poult Sci, 4: 243-246.
Newsholme P, Crabtree B and Andrawi MS. 1985. Glutamine metabolism in lymphocytes: Its biochemical physiological and clinical importance. Exp Physiol. 70: 473-489.
Newsholme P, Procopio J, Lima, MMR, Pithon-curi TC and Curi R. 2003a. Glutamine and glutamatetheir central role in cell metabolism and function. Cell Biochem Funct, 21:1-9.
Peng X, Hong Y and You ZY. 2004. Effects of enteral supplementation with glutamine granules on intestinal mucosal barrier function in severe burned patients .Burns, 30, 135–139.
Peter G and Elia M. 1997. The use of glutamine in the treatment of gastrointestinal disorders in man.J Nutr, 13, 743–747.
priya D, jayachandran K, nanjappan S and selvara jK. 2010. Oral supplementation on putrescince and l-glutamine on the growth performance, immunity and intestinal enzymes in the broiler chickens. Tamilnadu VET and Anim Sci, 5:250-254.
Reeds PJ, Burrin DG, Stoll B and Jahoor F. 2000. Intestinal Glutamate Metabolism. J Nutr,130:978s-982s.
Sahin N, Tuzcu M, Ozercan I, Sahin K, Prasad AS and Kucuk O. 2009a. Zinc picolinate in the prevention of leiomyoma in Japanese quail. J Med Food. In press.
Sandercock DA, Hunter RR, Nute GR, Mitchell MA and Hocking PM. 2001. Acute heat stress induced alterations in blood acid-base status and skeletal muscle membrane integrity in broiler chickens at two ages: Implications for meat quality. J Poult Sci. 80: 418–425.
SAS. 2003. SAS Users Guide: Statistics. SAS Institute Inc., Cary, NC, USA.
Shu G, Fu WL, Jiang1 QY, Gao P, Sun FW, Xu PW, Zhou GX and Zhu XT. 2007. Effects of glycyl-glutamine on the growth performance and lipid deposition in yue-huang broilers. J Anim Feed Sci, 16:232-240.
Soltan MA. 2009. Influence dietary supplementation on growth performance, small intestinal morphology, immune response and some blood parameters broiler chickens, Int Poult Sci, 1:60-68.
Souba WW, Klimberg VS, Plumley DA, Salloum RM, Flynn TC and Bland KI. 1990. The role of glutamine in maintaining a healthy gut and supporting the metabolic response to injury and infection. J Surg, 48:383-91.
Tanha T, Amanlou H, Chamani M, Ebrahimnezhad Y, Salamatdost R, Maheri N and Fathi M. 2011.Impact of glutamine on glutathione peroxidase activity (GPX) and total antioxidantstatus (TAS) during transition period in Holstein dairy cows. J Cell Anim Biol, 10: 206-214.
Tapiero H, Mathe G, Couvreur P and Tew KD. 2002. Free amino acids in human health and pathologies - II. Glutamine and glutamate. Biomed Pharmacother, 56: 446-457.
Watford M. 2008. Glutamine metabolism and function in relation to proline synthesis and the safety of glutamine and proline supplementation. J Nutr, 138:2003–7.
Windmueller HG and Spaeth AE. 1980. Respiratory fuels and nitrogen metabolism in vivo in small intestine of fed rats. J Biol Chem, 255: 107–112.
Wolfenson D, Bachrach D, Maman M, Graber Y and Rozenboim I. 2001. Evaporative cooling of ventral regions of the skin in heat stressed laying hens. J Poult Sci, 80: 958–964.
Yardibi H and Turkay G. 2008.The effects of vitamin E on the antioxidant system, egg production and egg quality in heat stressed laying hen. Brit Poult Sci. 32, 319–325.
Yi GF, Allee GL, Frank JW, Spencer JD and Touchette KJ. 2001. Impact of glutamine, menhaden fish meal and spray-dried plasma on the growth and intestinal morphology of broilers . J Poult Sci, 80: (Supplement. 1), 201.
Zhu B y Q, Xu QY, Xu H, Wang CA and Sun DJ. 2011. Dietary Glutamine Supplementation Improves Tissue Antioxidant Status and Serumnon-Specific Immunity of Juvenile Hybrid Sturgeon (Acipenser Schrenck And Husodauricus ). J Appl Ichthyol, 27: 715-720.
Zou XT, Zheng GH, Fang XJ andJiang JF. 2006. Effects of glutamine on growth performance of weanling piglets. Czech J Anim Sci, 51: 444-448.
Dear Mustafa, thanks a lot for your article. The antistress action of glutamine is caused by central position of these amino acids in transferring of amino groups. So, in this way glutamine is highly increasing their spectra of biochemical reactions, responsible for antistress action. You can support glutamine by betaine and carnitine. Probably these substances can give a synergic effect. Best wishes to you.
I understand that the report of Prof. Youssef is consistent with my considerations.
Based on the author's conclusion, it was concluded that although dietary Glutamine consumption can improve the performance of broiler chickens during the grower and whole period under heat stress, but it doesn’t affect the performance during starter period or blood parameter at week 6 of age. Thus, the lack of significant effect during the starter period may be due to high temperature requirements for broilers (34-32) compared to the grower period (~28ºC) after 3 weeks of age. Thus, the critical or broken point for heat stress in broilers in greater than 29ºC after 3 weeks of age.
Good work, with seemingly coherent results. There is a single issue where I would like to make a consideration, regarding the fact that the performance of the birds in the phase from 0 to 21 days, contrary to what happened in the 22 to 42 days, was not influenced by the levels of glutamine. I believe that this variation of results would be related to the fact that the temperature of 32 º represents a lower intensity of stress when applied in the phase of 0 to 21 days, compared to the phase of 22 to 42 days.





.jpg&w=3840&q=75)







.jpg&w=3840&q=75)