INTRODUCTION
Alphitobius diaperinus is one of the most predominant poultry litters inhabiting insect species all over the world. It has been shown to be able of transmitting a large variety of poultry pathogenic organisms, such as viral agents (IBDV…) [1], bacterial disease organisms (Salmonella spp, Campylobacter spp…) [2] and several poultry parasites.
Understanding the biology, the epidemiology and the behaviour of this insect is primordial to determine its control measures.
In this paper, we have studied the spatial distribution and the population fluctuation of Alphitobius diaperinus in a broiler breeding house during a raising cycle in the Province of Constantine (North-East of Algeria).
MATERIAL AND METHODS
The Breeding House: The study was carried out between February and March 2008 in a naturally infested greenhouse-like poultry house located in the Community of Hamma Bouziane, Province of Constantine (North-East of Algeria). It is 08 m wide by 48 m long, earth floored with sides made of multiple layers of reed and straw covered with a plastic foil. It held one flock of 3000 chicken for 50 days. No chemical treatment was used and the litter consisted of wood shavings.
Insect Sampling: Adults and larvae A. diaperinus were sampled using the sandwich type traps (wooden boxes of 20 cm / 15 cm / 8 cm, with cover and two openings of 1 cm in the longitudinal direction, next to base of the box. The traps are filled with corrugated paper) [3]. Traps were placed at 04 locations across the house and 8 locations down the house (Fig. 1). At each location, temperature (at 06 cm depth) and humidity (at the surface) of the litter were measured using a digital hygro-thermometer (Oregon Scientific, model ETHG913R). The contents of the traps were collected weekly in plastic bags and then frozen. The traps were refilled with new corrugated paper after each collection. Beetles and larvae counts and environmental data (temperature and humidity) were recorded with correct corresponding spatial coordinates (X, Y) (expressed in meters).
Fig. 1: Broiler house sampling grid
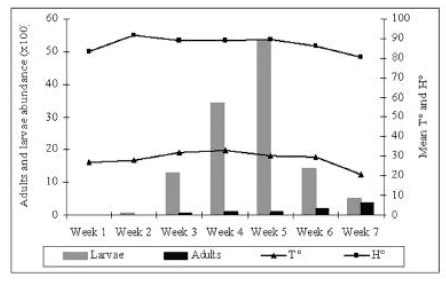
Fig. 2: Population fluctuation of larvae and adults Alphitobius diaperinus related to the temperature (T°) and the humidity (H°) of the litter.
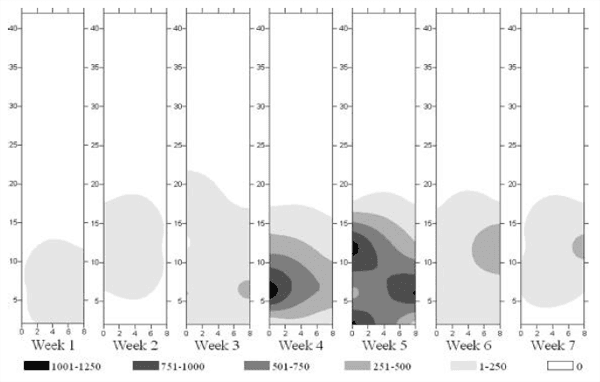
Fig. 3: Spatial distribution of Alphitobius diaperinus population in the litter of the poultry house from week 1 to week 7
Spatial Distribution: Contour plots of insects were obtained by using the geo-statistical software Golden Surfer 8 [4] with the kriging algorithm, a statistical linear method estimating the best value based on weighted linear combinations of neighbouring values while aiming to minimize the error variance between samples.
RESULTS
Population Fluctuation (Fig. 2): The total larvae trapped increased from the first week to the fifth week, then decreased through week 6 and 7, whereas adults increased from week 1 to the end. Larvae were more abundant in all of the samples.
Temperature beneath the litter was always approximately similar during the fist six weeks then decreased by the 7th week. Humidity showed a more or less comparable evolution.
Spatial Distribution (Fig. 3): By the first week the insect population was concentrated in quarter the surface of the facility and then it spread through the half by week 3, 4, 5 and 6. By the seventh week the population moved far from the southern side of the house. 70% of the population was concentrated in the edges of the breeding zone (Which represents 6% of its total surface) and 30% in its centre.
Insects' concentration in the brooder section was not correlated to the poultry distribution, which was not uniform all over the house.
DISCUSSION
The average temperature and humidity during the raising period (28.60°C and 86.91% H°) are ideal to the development of the different stages of this insect. Its life cycle could be achieved in 70 to 89 days at 27°C and 65% [5] and in 26 days at 31°C [6]. This explains the fact that the number of immature forms in our results reached its maximum by the 5th week and decreased by week 6 and 7 during what larvae became pupa then adults. As in Chernaki et al's findings, the results of our study proved that there is no correlation between the numbers of trapped insects and temperature records [7]. Lambkin et al, in compact earth flour, reported that the number of lesser mealworms increased over the 14 first days, picking at 16 and declined over the 28 last days [8].
Strother and Steelman found darkling beetle populations initially concentrated along the perimeter of the house and spread toward the centre as population density increased [9]. Lower densities are found in open locations and under drinkers, while high densities occurred under feeders, support pillars and along the house edges [8, 10].
CONCLUSION
The greenhouse-like poultry house (mostly used in our region) provides an excellent environment to the installation, the development and the spread of the darkling beetle A. diaperinus. The environmental conditions within this kind of poultry houses allow A. diaperinus to complete its life cycle during a breeding period.
The earth floor is an excellent medium where eggs can be deposited and final instars tunnel and pupate. The facility sides are also ideal for pupating. The insects hide in earth floor and insulation materials (reed and straw) during the depopulation period and then emerge by the start of a new flock.
Further studies seem to be necessary to understand the biology and the behaviour of this insect in this type of poultry houses along the year.
REFERENCES
1. Mc Allister, J.C., C.D. Steelman, L.A. Newberry and J.K. Skeels, 1995. Isolation of infectious bursal disease virus from the lesser mealworm, Alphitobius diaperinus (Panzer). Poultry Science, 74(1): 45-49.
2. Skov, M.N., A.G. Spencer, B. Hald, L. Petersen, B. Nauerby, B. Carstensen and M. Madsen, 2004. The role of Litter beetles as potential reservoir for Salmonella enterica and Thermophilic Campylobacter spp between broiler flocks. Avian Disease, 48(1): 9-18.
3. Safrit, R.D. and R.C. Axtell, 1984. Evaluations of sampling methods for darkling beetles (Alphitobius diaperinus) in the litter of turkey and broiler houses. Poultry Science, 63(12): 2368-2375.
4. Golden Co: Surfer 8 user's guide. Golden software, Inc 2002.
5. Dass, R., R.A. Agarual and A.U.N. Paul, 1984. Feeding potential and biology of lesser mealworm Alphitobius diaperinus (Panz.) (Col., Tenebrionidae), preying on Corcyra cephalonica St. (Lep., Pyralidae). Zeitschrift fuer Angewandte Entomologie, 98: 444-447.
6. Chernaki, A.M. and L.M. De Almeida, 2001. Thermal Requirements, development and survival of the immature Stages of the Alphitobius diaperinus (Panz.) (Coleoptera: Tenebrionidae). Neotropical Entomology, 30(3): 365-368.
7. Chernaki, A.M., L.M. De Almeida, D.R. Sosa-Gomez, A. Anjos and K.M. Vogado, 2007. Population fluctuation and spatial distribution of Alphitobius diaperinus (Panz.) (Coleoptera: Tenebrionidae) in a poultry house, Cascavel, Parana state, Brazil. Brazilian Journal of Biology, 67(2): 209-213.
8. Lambkin, T.A., R.A. Kopittke, S.J. Rice, J.S. Bartlett and M.P. Zalucki, 2007. Distributions of lesser mealworm (Coleoptera: Tenebrionidae) in litter of a compacted earth floor broiler house in subtropical Queensland, Australia. Journal of Economical Entomology, 100(4): 1136-1146.
9. Strother, K.O. and C.D. Steelman, 2001. Spatial analysis of Alphitobius diaperinus (Coleoptera: Tenebrionidae) in broiler production facilities. Environmental Entomology, 30(3): 556-561.
10. Cogan, P., D. Webb and M. Wakefield, 1996. A comparison of four residual insecticides for the control of the lesser mealworm beetle (Alphitobius diaperinus Panzer) in turkey broiler houses in the UK. International Pest Control, 38(2): 52-55
This article was originally published in the Academic Journal of Entomology 2 (2):88-91, 2009 ISSN 1995-8994, 2009













.jpg&w=3840&q=75)