Sel-Plex® organic selenium in male and female broiler breeders
Reproductive responses to Sel-Plex® organic selenium in male and female broiler breeders: impact on production traits and hatchability
Achieving success with broiler breeder management is like hitting a moving target. Modern broiler stocks have been reported to grow at 4.6 times the rate of a 1957 random-bred strain due to increased genetic potential (Havenstein et al., 2003a). The 6-fold improvement in carcass yield of 2001 stocks fed a 2001 diet compared to 1957 stocks fed a 1957 diet is 85-90% due to genetics, and 10-15% due to nutritional changes (Havenstein et al., 2003b). While broiler 42- day body weight is increasing each year, the 42-day target body weight for male and female broiler breeders has remained the same, or even decreased (Rustad and Robinson, 2002).
In 1979, Hubbard male and female breeders were approximately 50% of the 42-day broiler weight. In 2001, this percentage had decreased to 36.1 for males and 30.3 for females. In essence, the degree of feed restriction has continued to increase while there is increased competition for a reduced feed allocation.
Complications in managing this increased growth efficiency is further complicated by the development of ‘yield’ varieties, carrying increased amounts of breast muscle, often on a smaller carcass frame. As selection for broiler breeder egg production is not as heritable or profitable as selection for growth traits, there are continued increases in the growth potential while egg production is not emphasized. As a result, Whitehead (2000) indicates that geneticists continually compound the problem by breeding a bird that, if allowed to exist in its freely expressed adult state, is completely unfit for life.
Understanding the ovarian function of the chicken and its interaction with nutritional status, age, and strain is likely the most important issue affecting poultry breeding companies today. The process involves the conversion of genetic, environmental, and nutritional cues into a cascade of signals from the neuroendocrine system.
These signals must be integrated and responded to by the organs and tissues primarily involved in reproduction, which will in turn produce more signals for both local and distant activities. The resulting eggs produced are the net result of the bird’s attempt to coordinate the demands its body and environment have placed on it. The ability of an embryo to survive the incubation process relies on a balance between hatchery management and breeder management. Specific feed ingredients, bird age, and flock management decisions can directly affect semen quality, the oviduct environment, and the egg environment. These factors combine to influence the potential of the egg to be fertile and ultimately to hatch.
Reproduction in the broiler breeder
The reproductive system of the laying hen is comprised of many organs. The list includes the hypothalamus, the anterior pituitary, the ovary, the oviduct, the liver and the skeletal system. Small follicle steroidogenesis, particularly estrogen production, is responsible for transforming a pullet into a hen. As plasma levels of estrogens increase, externally visible features include reddening and enlargement of the comb and wattles, a prenuptial feather molt (feather drop) and a widening of the pubic bones to permit egg passage. Internally, estrogen stimulates liver production of egg yolk lipids with a significant change in the color and size of the liver. Finally, the oviduct enlarges and becomes a secretory organ for deposition of albumen.
The male focuses on quantity rather than quality when it comes to sperm production. The hen then must screen out unsuitable sperm in order to guarantee production of high quality chicks. Mature sperm spend the majority of their time in the oviduct. Following mating, the hen will store sperm in the highly specialized microenvironments of the sperm storage tubules (SST) are located in the vaginal region of the oviduct. Only about 1 to 2% of the originally inseminated sperm enter the SST (Bakst et al., 1994), where they exist in a near-dormant state. The survival of the sperm to the point of insemination depends on a combination of sperm quality and the ability of the hen to provide a safe environment for the sperm.
The hormone messages being relayed between the ovary and the hypothalamic and pituitary control centers are altered by feed intake. Besides affecting follicle formation, and reproductive control, feeding level can alter the viability of the embryo through changes to the egg and to the early maturation process.
Excess nutrients are diverted into liver lipids, excess ovarian follicle development, and as abdominal fatpad (Etches, 1996). It can be a vicious cycle, with obesity continuing to worsen as the rate of egg production remains low and/or goes into early decline due to excess feed intake. The ovaries of growth-selected strains are particularly sensitive to overfeeding during the sexual maturation process (Renema et al., 2003).
Body weight in broiler breeder hens has been reported to be negatively correlated with duration of fertility and fertile egg production (Bilgili and Renden, 1985). Ultimately, reduced chick production in overfed broiler breeders is the culmination of poor egg production combined with reduced fertility, hatch of fertile, and embryonic viability (Yu et al., 1992).
The female oviduct environment can be hostile to sperm despite their existence in the SST within the oviduct wall. Duration of fertility (measured by monitoring fertility in consecutive eggs) can be reduced under conditions of overfeeding (Katanbaf et al., 1989b; Goerzen et al., 1996). It is known that fewer sperm survive in some bird strains and when excess feed is used (Renema et al., 2001), but it is not clear how the surviving sperm are affected, and if the remaining sperm are of similar quality to the ones originally placed.
Factors affecting hatching egg quality
There are many factors that can affect the potential of the embryo to survive incubation and generate a quality chick. Some of these factors are out of our control, such as hen age, and others can be manipulated through management decisions, such as egg size and hatchery environment. It can be difficult to formulate diets to optimize egg production, fertility, and hatchability as little is known about the nutritional requirements of the embryo (Leeson and Summers, 1991).
Dietary vitamin levels are increased in the diet with the hope they will also be increased in the egg. Yet there can be adverse reactions with this type of approach, as some vitamins have antagonistic relationships with others. Furthermore, there can be stability issues for long-term storage, as well as for feed processing procedures. With current genetic stocks, if the chick hatches in a weakened state due to a vitamin or mineral deficiency, it is more likely to succumb to disease now than with previous stocks.
Growth-selected stocks have low immunoresponsiveness (Siegel et al., 1984) due to either inadvertent or intentional negative selection pressure combined with growth efficiency selection. The developing embryo is especially sensitive to vitamin deficiency, which will result in death, malformation or some other atypical response (Leeson and Summers, 2001).
During embryo development, oxidative metabolism increases substantially over the incubation period and especially in the last few days before hatch (Freeman and Vince, 1974). This normal respiration related to embryo growth results in the production of free radicals, which can cause tissue damage through lipid peroxidation, with polyunsaturated fatty acids being especially vulnerable (Surai, 1999). The chick has developed effective antioxidant pathways to prevent damage.
The primary defense mechanism is a group of three enzymes (superoxide dismutase, glutathione peroxidase, and catalase), which convert free radicals produced by cellular respiration into less harmful alcohols (Ursiny et al., 1997). A second level of defense are the natural antioxidants – vitamin E, carotenoids, ascorbic acid, and glutathione, which protect the developing chick (Surai, 1999). During the last week of incubation, fat-soluble antioxidants are moved into the liver and yolk sac membrane.
The major fat soluble antioxidant, vitamin E, moves from the yolk to the embryo tissue at this time (Gaal et al., 1995). Ascorbic acid (vitamin C) is the major water-soluble antioxidant, and is produced in the yolk sac membrane before transport to tissues like the brain (Surai, 1999). This helps protect membrane lipids during the large metabolic effort of hatching. The third level of antioxidant defense is the generation of enzymes that rebuild damaged membranes (Surai and Sparks, 2001).
ROLE OF DIETARY SELENIUM
Selenium is normally provided in the diet in the form of inorganic sodium selenite. An organic form can be provided (Sel-Plex®), which is selenium yeast.
Yeast, like plants, form selenoamino acids and other organic selenocompounds that exist in very reduced form in comparison to the highly oxidized inorganic selenium forms (selenite and selenate). Organic minerals are transported intact and retained better in target tissues or organs. Higher selenium in eggs reflects increased antioxidative properties of the egg during storage, therefore preserving the egg for incubation and potentially increasing hatchability.
Cantor (1997) and Paton et al. (2000b) found that eggs from Sel-Plex®-fed chickens were significantly higher in selenium than eggs from sodium selenitefed chickens. Organic selenium has a vitamin E-sparing action through its involvement in vitamin E retention in the plasma and through involvement with the primary enzymatic defense system of the embryo against lipid peroxidation. In fact, supplementing organic selenium to breeder diets has been shown to increase levels of other antioxidants (vitamin A, E and carotenoids) in the egg (Surai and Sparks, 2001).
The protective effects of organic selenium are especially apparent during the highly oxidative state of late incubation and the first few days after hatch.
Selenium is an integral part of the antioxidant enzyme glutathione peroxidase (GSH-Px) as well as a component of many other selenoenzymes. Oxygen metabolism produces free radicals, which have potentially toxic effects on all biological molecules (Surai, 2000). Glutathione peroxidase aids in the removal of oxidative compounds in the form of hydrogen peroxide and hydroperoxides from the cell (Burk, 1989). Buildup of these substances can impair cell membrane structure and function, and once the membrane is damaged decreased productivity and reproductive performance can result (Surai, 2000).
Selenoamino acids have been shown to have higher bioavailability than traditional inorganic sources commonly used for dietary supplementation. They are actively absorbed in the intestine compared to the passive absorption of inorganic selenium (Surai, 2002). Furthermore, selenomethionine and selenocysteine can be incorporated non-specifically into structural proteins (particularly muscle tissue) during protein synthesis.
Selenomethionine can be substituted for methionine during protein synthesis due to its similar structure (methionine contains a sulphur atom instead of a selenium atom). This critical difference between selenium sources allows the organic selenium compounds in Sel-Plex® to contribute to a selenium reserve to be available for prevention of lipid peroxidation (through GSH-Px) during stress conditions (Surai, 2002). In the broiler breeder, it also enables enhanced transfer of selenium from the hen to the embryo (Edens, 2002). Increased antioxidant uptake in the hen due to the maternal diet is linked to increased antioxidant concentrations in the developing chick (Surai et al., 1999).
The need for defense against oxidative damage is clear in the male, where antioxidant enzymes play a key role in maintaining the sperm cells (Surai et al., 1998). Sperm cells contain large amounts of polyunsaturated fatty acids, which allow them to maintain flexibility relating to motility (Surai, 2002). However, this means they are also a target for lipid peroxidation. Cellular integrity is maintained by GSH-Px, other selenoenzymes and vitamin E, which protect the cell membranes from oxidative damage (Flohe and Zimmermann, 1970).
Some recent research has demonstrated that the inclusion of selenium in poultry diets enhances sperm numbers, and using an organic source (Sel-Plex®) reduces production of defective sperm, thereby having a positive effect on the fertilizing potential of the male (Edens, 2002). Little information is available regarding the effect of dietary selenium source on the reproductive efficiency of laying hens.
How does dietary selenium source affect the hen’s contribution to fertility? (Study 1)
Egg production and fertility decline with age. The decrease in hatchability and fertility associated with an increase in age might be due to the older hen’s inability to hold sperm in the SST (Fasenko et al., 1992).
Furthermore, the sperm do not retain their viability as long in the SST of older hens, and are released in larger numbers from the SST (Bramwell et al., 1995). Quantification of fertility is determined by the occurrence rate of perivitelline holes caused by the sperm. In past research, killing the hen to obtain the newly ovulated ovum was the only way to determine sperm hole quantities. Bramwell et al. (1995) adapted the technique to use eggs for the determination of perivitelline sperm hole numbers.
This study examined the effects of selenium supplementation form and level on female reproductive performance and egg traits. Its intent was to determine the effects of organic (Sel-Plex®) and inorganic selenium supplementation in the laying hen diet on fertilization potential and egg traits.
METHODS
We housed 75 hens in individual laying cages at 61 weeks of age. Three dietary treatments were imposed, varying in selenium source and level. Twenty-five hens were fed a control diet, 25 were fed a diet enriched with inorganic selenium in the form of sodium selenite and 25 were fed a diet enriched with organic selenium in the form of the Alltech product, Sel-Plex®. All diets contain 19% CP and 2875 kcal ME/kg. The control diet had a selenium inclusion rate of 0.1 mg Se/kg, whereas both enriched diets contained a total of 0.3 mg Se/kg with an added 0.2 ppm Se coming from the organic or inorganic selenium source. The diets were fed for a 3-week period prior to insemination to ensure tissue saturation of the new dietary selenium forms and concentrations.
Following the 3-week acclimation period, all hens were artificially inseminated with 50 μL of neat, pooled semen collected from a group of 22 broiler breeder males (116 million sperm/dose). Eggs were collected from 2 to 7 days after insemination for the quantification of perivitelline layer sperm holes. Eggs traits (weight, specific gravity, yolk and dry shell weight) were measured at 30 and 60 days from the start of dietary treatments.
The sperm penetration assay of Bramwell et al. (1995) was used to quantify the perivitelline layer sperm holes. An approximately 1 cm2 section of the perivitelline layer above the germinal disc was cut free, cleaned, mounted to a microscope slide, fixed, and stained with Schiff’s Acid reagent to generate a contrast with the sperm holes. The holes were counted at 100X magnification. The raw numbers and change in numbers over time were used as a representation of quantifiable fertilization potential.
OBSERVATIONS
Egg traits
The use of Sel-Plex® rather than sodium selenite as the dietary selenium source has previously been shown to increase shell breaking strength after 42 days in laying hens at 80 wk of age (Paton et al., 2000a). Our study found no significant differences in egg traits between the various treatments after 30 days on the diets. However, the Sel-Plex® treatment resulted in numerically the greatest positive change in shell quality during the 30 day period of this trial.
Research with younger birds (26 wk of age) has indicated no difference in shell quality with the use of Sel-Plex® although the comparisons were made after only 28 days on the diet (Paton et al., 2000a).
After 9 wks on the diets, shell weights were higher in the Sel-Plex® group than in the controls, while shell weights in the group given inorganic Se was intermediate (Table 1). Egg specific gravity, a measure of shell quality, was greater in the Sel-Plex® treatment than in either the control or selenite groups.
While higher dietary selenium levels preserved shell quality to some degree, the organic selenium in Sel- Plex® proved to have a more substantial effect on the preservation of shell quality characteristics.
Table 1. Effect of selenium source and level on shell weight and egg specific gravity after 9 weeks.

a,b Means within a column with no common superscript differ (P<0.05).
Perivitelline sperm hole assay
The Sel-Plex® treatment group had the highest mean number of sperm holes in the 2 to 4 days after insemination study period, while controls had the lowest number (Table 2). Although neither the Sel- Plex® nor the control treatment were statistically different from the inorganic Se treatment, they were statistically different from each other.
A similar relationship among the dietary treatments occurred for the 5 to 7 day period, and for a comparison over the entire 2 to 7 day period. The perivitelline sperm hole numbers declined at a similar rate among dietary treatment groups between the 2 to 4 day and the 5 to 7 day study periods. As the control group started with a lower number of sperm holes than either of the other two treatment groups, their final numbers at 7 days were very low.
The Sel-Plex® and inorganic Se treatments both retained higher fertility potential throughout the sampling period allowing for a longer period of time between artificial inseminations. While not statistically different, the average number of sperm holes at the site of fertilization in the Sel- Plex® group was higher than that of the inorganic Se treatment. This indicates that the greater bioavailability of selenium in Sel-Plex® compared to inorganic Se may be advantageous for the fertility of the female based on changes to the oviduct environment.
Selenium seems to play an important role in the maintenance of fertility in older laying hens. This is most likely due to the seleniumdependent GSH-Px improving the environment of the SST (Surai, 2000). The SST need to maintain a stable environment and the elimination or reduction of free radicals within the tubules is essential.
SUMMARY: STUDY 1
Selenium supplementation is beneficial to increasing and maintaining fertility and shell quality in older hens. Supplementation with the organic selenium in Sel-Plex® may have a greater impact on reproductive ability than inorganic sodium selenite. Factors such as age and length of exposure to the diet also play a role in the results of both this and past studies.
The form and quantity of dietary selenium appear to impact the oviduct environment of the hen. Fewer sperm are able to survive under low dietary selenium conditions (control) compared to conditions provided by supplementation with an organic selenium source (Sel- Plex®). Conditions in the SST may be central to the differences noted in the number of sperm being able to reach the site of fertilization. Through a combination of a more stable, antioxidant-free environment with a potentially slowed sperm metabolism, more sperm may be able to survive storage. Selenium source appears to influence the hen’s contribution to the fertility of the breeder flock.
Effects of dietary selenium source on the fertility and hatchability of broiler breeders (Study 2)
Supplying selenium to broiler breeders in the organic selenoamino acid form may have an important impact on poultry reproduction at the level of sperm formation, sperm storage, and in the hatching egg through increased protection from oxidative damage.
This experiment was designed to provide information on the role of dietary selenium form on both female and male fertility. While there is evidence on a flock basis that selenium source affects broiler breeder female fertility, it is not as clear how these benefits are being expressed.
Previous work suggests the use of an organic selenium source (Sel-Plex®) can lead to improved egg production, shell quality, sperm viability, and embryo survival. Egg shell quality may be enhanced through an altered efficiency of calcium metabolism, and sperm quality may be enhanced through protective antioxidant effects in the male and in the female oviduct. This study assessed some of these production and fertility traits in broiler breeders maintained individually.
Table 2.The effect of selenium supplementation and source in the laying hen diet on perivitelline sperm holes.
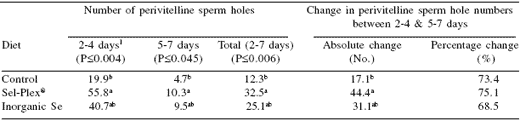
a,b Means within a column with no common superscript differ (P<0.05). 1Number of days after insemination.
The objective of this trial was to characterize specific effects of dietary selenium source on fertility and embryo viability aspects in commercial broiler breeder stocks. A female diet with no added selenium was used to identify the impact of dietary selenium.
Inorganic and organic dietary selenium sources were compared to demonstrate the impact of differences in selenium accessibility and tissue storage on reproductive traits and embryo survival. Higher rates of production, fertility, and ultimately chick quality, would decrease the number of birds required to maintain current rates of production, as well as the overall cost of production.
METHODS
Ross 508 pullets were reared in a light tight facility following the breeder BW profile (Aviagen Inc).
From photostimulation (22 wks of age) pullets were fed a selenium-free laying ration (No added Se), a standard ration containing sodium selenite (0.3 mg Se/kg), or a ration containing selenium yeast (0.3 mg Se/kg from Sel-Plex®).
Thirty hens per treatment were inseminated weekly (from 30 wks) using pooled semen from males fed a standard, sodium selenite diet or a diet containing the same amount of Se from Sel-Plex®. Individual egg production to 58 wk, egg weight, egg specific gravity, and BW were recorded. At 35 and 57 wk of age, eggs from 2 to 5 days after insemination were subjected to the perivitelline sperm penetration assay to measure the number of sperm penetrations near the germinal disk. Eggs were incubated weekly and the hatch residue broken out to determine fertility, hatchability, and embryonic mortality.
OBSERVATIONS
Sperm management
Perivitelline sperm hole numbers of Sel-Plex® and selenite treatment eggs were similar. Both treatments had more sperm holes than eggs from unsupplemented hens by a factor of 2 to 3 (Table 3). Sel-Plex® supplementation improved maintenance of sperm numbers between the day 2 and the day 5 sampling.
By day 5, Sel-Plex® eggs still had an average of 60 perivitelline sperm holes compared to 14 in control eggs, while selenite treatment eggs were intermediate (31 holes). These values represented a decline of 31% in apparent viable sperm population in Sel-Plex® birds between Day 2 and 5 after insemination compared to a 46% and 48% drop within non-supplemented and selenite-fed birds, respectively.
The ability to maintain a viable sperm population for as long as possible reduces necessary frequency of insemination. While selenium appears essential to allow the sperm into the oviduct, organic selenium in Sel-Plex® may have an advantage over inorganic selenium in keeping the sperm population stable and alive. This is especially important as the hens age and have a reduced sperm storage capacity at the uterovaginal junction (Goerzen et al., 1996).
Table 3. The effect of dietary selenium level and source on perivitelline sperm holes of eggs from broiler breeder hens.

a,b Means within a column with no common superscript differ (P<0.05).
1 Number of days after insemination. 20.3 ppm Se
The males on the Sel-Plex® diet produced greater semen volume early in production, with an average of 0.36 ml/bird compared to 0.19 ml/bird in males on the selenite diet (36 weeks of age). At 56 weeks of age, this difference was no longer significant, but remained at nearly the same magnitude. The comparison was complicated at the later ages due to several small males dropping out of semen production part way through the trial (selenite treatment). Testes of all birds are currently being examined for the presence of functionally active sperm producing cells.
Egg production and egg quality traits
Birds on the Sel-Plex® diet entered egg production slightly behind the other feeding treatments (nonsignificant difference), but caught up within a few weeks. Early egg production to 29 wk of age was not different (Table 4). In fact, the rate of lay was similar through most of the production period.
However, during the late lay period (49-58 weeks) the hen-housed rate of lay was 68% in Sel-Plex® birds compared to 61% and 60% in the selenite and nonsupplemented treatments, respectively. The Sel-Plex® birds produced an extra 5 eggs/bird during this period, on average. This is an important time to be producing more eggs, as egg size is higher than in young breeders, which results in a larger chick size and ultimately a greater broiler weight. Edens (2002) also indicated that the egg production of Sel-Plex®- fed hens initially lagged behind, but caught up and even surpassed that of the selenite-fed hens after 5 wk.
Table 4. The effect of dietary selenium level and source on egg production traits of broiler breeders.
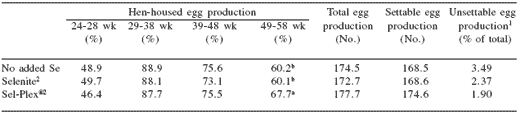
a,b Means within a column with no common superscript differ (P<0.05).
1 Includes double-yolked, soft-shelled, membranous, and abnormally-shelled eggs.
20.3 ppm Se
Ultimately the settable egg production in the dietary groups was 168.5 (non-supplemented), 168.6 (selenite), and 174.6 (Sel-Plex®) eggs/bird (Table 4).
Overall, unsettable egg production ranged from 3.49% in non-supplemented hens to 1.9% in Sel- Plex® hens and was not significantly different. However, during the late lay period (49-58 wk), the Sel-Plex® hens produced significantly fewer unsettable eggs (0.9%) than non-supplemented hens (3.3%), while selenite hens were intermediate (1.7%).
Egg weight and shell quality of settable eggs was assessed throughout the trial and was unaffected. This means that if the hen laid a good egg, it also had a good shell. However, diet affected how many eggs were produced with good shells, as shell defects were the primary egg quality problem in unsettable eggs.
Feeding Sel-Plex® organic selenium to laying hens at 80 wk of age has previously been shown to improve shell breaking strength (Paton et al., 2000a).
Interestingly, dietary selenium affected the change in shell weight as the hens aged. Between 36 and 56 wk of age, shell weight increased by 0.55, 0.80, and 0.76 g in eggs of the non-supplemented, selenite, and Sel-Plex®-fed hens, respectively.
During this time egg size also increased, meaning that the shell was being stretched over more egg, and therefore making up a smaller percentage of total egg weight. The proportion of shell weight dropped by 0.84% of egg weight in non-supplemented hens, 0.80% in selenitefed hens, and 0.57% in Sel-Plex® hens between 36 and 56 wk of age.
The Sel-Plex® hens were significantly less affected by age-related declines in the proportion of egg shell than the non-supplemented hens. While egg specific gravity was not significantly affected, this may be an indicator of increased shell thickness in the Sel-Plex® treatment (not tested). If this were different, there could be implications for incubation success and for defense from contamination in the barn.
Hen body weight followed a similar pattern throughout the production period. However, the nonsupplemented hens grew heavier than the other treatment hens by 42 weeks of age. This difference carried through to 58 weeks of age. This comparison is somewhat artificial, as the body weight profile of the non-supplemented group was inflated by hens that dropped out of lay at a fairly young age. Nutrients they were no longer allocating to egg production went into growth instead. By the end of the trial, 100% of the Sel-Plex® hens were still in active production while only 87% and 90% of the non-supplemented and selenite treatment hens remained, respectively.
Lack of production was due to either birds ceasing production, or to hen mortality (mortality limited to non-supplemented treatment). Reduced mortality has been linked to selenium supplementation (Arnold et al., 1974), particularly under stress conditions such as an immune challenge (Edens, 2001). However, this does not explain the increased proportion of birds still in active lay at 58 weeks of age.
Reproduction in the broiler breeder can be a fragile state and is often the first thing to go when there is stress, or nutrients are insufficient. The fact that all Sel-Plex® birds were still in production may relate to an improved efficiency of nutrient uptake. Presumably the gut benefits from the improved protection from cell membrane damage afforded by the organic selenium. During the late production period, these birds were producing more eggs than hens of the other treatments (Table 4) with no effect on their body weight relative to that of hens on the other treatments.
Fertility, hatchability, and embryonic mortality
Prior to 34 weeks, hatchability averaged 88% in Sel- Plex® treatment eggs compared to 80% in selenite-fed birds and 77% in non-supplemented birds, and was similar in all treatments after peak production.
Overall, fertility, hatchability, and hatch-of-fertile eggs demonstrated the beneficial nature of dietary selenium, but did not differentiate between selenium sources (Table 5). Fertility, for example, was 86.9% in non-supplemented hens compared to 90.1% in hens on selenite and Sel-Plex® diets. Not including selenium in the diet did not seriously harm hatchability, which is in contrast with work by Latshaw and Osman (1974) demonstrating a drop in hatchability to 18% in seleniumdeficient hens. The current study may have provided more naturally occurring selenium in the other feed ingredients and the non-supplemented dietary treatment was not imposed until photostimulation (22 weeks of age).
Embryonic mortality can be a telling identifier of specific dietary or genetic effects. Problems with early embryonic mortality (1-14 days of incubation) can point to nutrient deficiencies. In this study, 5.33% of non-supplemented embryos died during this period compared to 3.72% (selenite) and 3.52% (Sel-Plex®) in the selenium-supplemented hens (Table 5). While selenium source did not make a difference here, clearly selenium supplementation was shown to be important.
A beneficial effect of organic selenium was expected for the late incubation and hatch period, as this is the time of the greatest oxidative load for the embryo, and when the protective antioxidant effects of the Sel-Plex® may be most apparent.
Variability among birds reduced the significance of this comparison, however, and late embryonic mortality, dead-in-shells, and hatchery culls totaled 3.66%, 3.85%, and 3.14% of eggs set for the nonsupplemented, selenite, and Sel-Plex® treatments, respectively (Table 5). Based on these numerical differences, there appears to be a potentially protective effect of Sel-Plex® compared to inorganic selenium in the diet.
Examining this relationship more closely revealed an interesting trend over time. Late embryonic mortality of all treatments was similar at the start of the trial, when all birds were still on a fairly high plane of nutrition. As the hens aged, late embryonic mortality stayed almost constant in non-supplemented and selenite hens, while it decreased in Sel-Plex® hens (Figure 1). As feed allocations were reduced with age, the micronutrients would have been in shorter supply.
The improved efficiency of selenium uptake in the Sel-Plex® diet may not have made a substantial difference on hatchability until a nutrient challenge was faced by the flock. This fits with observations that Sel-Plex® can demonstrate benefits in stressful situations. Heat stress and long-term egg storage are examples of stress situations where Sel-Plex® has been shown to help. Surai and Dvorska (2001) indicate that there are numerous on-farm stress conditions that could be alleviated in part by organic selenium supplementation.
Ultimately what determines the success of a broiler breeder management program is chick production. In this trial, chick production was calculated from the hatchability of settable eggs. The unsupplemented hens produced an average of 131.3 chicks/hen-housed by 58 weeks of age, while selenite hens produced 139.1 chicks/hen, and Sel-Plex® hens produced 145.3 chicks/hen (Table 5). Between the selenite and Sel- Plex® selenium source diets, the numerical differences in settable eggs, embryonic mortality, hatchability, and hatch of fertile culminated in a difference of 5.8 chicks in favor of the Sel-Plex® hens. This difference increased to 14.1 chicks when compared to the nonsupplemented hens.
Table 5.The effect of dietary selenium level and source on fertility, hatchability, embryo mortality and chick production traits of broiler breeder females.
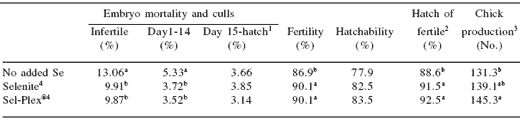
a,b Means within a column with no common superscript differ (P<0.05).
1 Includes embryo mortality to hatch, dead-in-shell, and hatchery culls.
2 Hatchability calculated only from fertile eggs set (infertile eggs excluded).
3 Chick production = hatchability X settable eggs.
40.3 ppm Se.
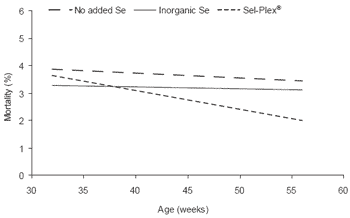
Figure 1.Linear representation of trends in embryonic mortality between 15 days of incubation and hatch (including dead-in-shell and hatchery culls) of broiler breeder hens fed diets containing no supplemental Se, 0.3 ppm inorganic Se, or 0.3 ppm Sel-Plex®.
SUMMARY: STUDY 2
Reproductive traits were improved with the inclusion of dietary selenium, while Sel-Plex® supplementation also improved sperm survival in the oviduct, as well as settable egg production late in lay through increased egg production and reduced shell defects. Ultimately, chick production was improved in the Sel-Plex® treatment through more successful settable egg production and the additive culmination of numerical improvements in hatchability and embryo viability measurements. Selenium is essential in the diet for a successful reproductive effort. Additional benefits of using the Sel-Plex® are also possible. Selenium source appears to influence the hen’s contribution to the fertility of the breeder flock and to beneficially affect semen volume early in production.
Conclusions
Managing the broiler breeder female for optimal chick production requires an understanding of reproductive physiology, nutrition, and their interaction. Besides a thorough knowledge of everyday management, there must also be an awareness of feed ingredients and their interactions both with each other and with environmental effects.
Whereas the basic composition of the egg is fairly constant, diet and specific feed ingredients can affect what and how much of some of the minor ingredients make it into the egg and ultimately the embryo. Specialized feed ingredients are available that behave differently than traditional ingredients and can enhance egg and chick quality under the right conditions. Together these factors can be used to enhance embryo survival.
References
Arnold, R.L., O.E. Olsen and C.W. Carlson. 1974. Tissue selenium content and serum tocopherols as influenced by dietary type, selenium and vitamin E. Poult. Sci. 53:2185-2192.
Bakst, M.R., G. Wishart and J.P. Brillard. 1994. Oviduct sperm selection, transport, and storage in poultry. Poult. Sci. Rev. 5:117-143.
Bilgili, S.F. and J.A. Renden. 1985. Relationship of body fat to fertility in broiler breeder hens. Poult. Sci. 64: 1394-1396.
Bramwell, R.K., H.L. Marks and B. Howarth, 1995. Quantitative determination of spermatozoa penetration of the perivitelline layer of the hen’s ovum as assessed on oviposited eggs. Poult. Sci. 74:1875-1883.
Burk, R.F. 1989. Recent developments in trace element metabolism and function: newer roles of selenium in nutrition. J. Nutr. 119:1051-1054.
Cantor, A.H. 1997. The role of selenium in poultry nutrition. In: Biotechnology in the Feed Industry, Proceedings of Alltech’s 13th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). Nottingham University Press, Nottingham, UK, pp. 155-164.
Edens, F.W. 2001. Involvement of Sel-Plex in physiological stability and performance of broiler chickens. In: Biotechnology in the Feed Industry, Proceedings of Alltech’s 17th Annual Symposium (K.A. Jacques and T.P. Lyons, eds). Nottingham University Press, UK, pp. 349-376.
Edens, F.W. 2002. Practical applications for selenomethionine: broiler breeder reproduction. In: Nutritional Biochemistry in the Feed and Food Industries, Proceedings of Alltech’s 18th Annual Symposium (K.A. Jacques and T.P. Lyons, eds). Nottingham University Press, UK, pp. 29-42.
Etches, R.J. 1996. Reproduction in Poultry. CAB International, Wallingford, Oxon.
Fasenko, G.M., R.T. Hardin, F.E. Robinson and J.L. Wilson. 1992. Relationship between hen age and egg sequence position with fertility, viability and preincubation embryonic development in broiler breeders. Poult. Sci. 71:1374-1383.
Flohe, L. and R. Zimmermann. 1970. The role of GSH peroxidase in protecting the membrane of rate liver mitochondria. Biochim. Bio. Acta 223:210- 213.
Freeman, B.M. and M.A. Vince. 1974. In: Development of the avian embryo. Champman and Hall, London, UK, pp. 249-260.
Gaal, T., M. Mezes, R.C. Noble, J. Dixon and B.K. Speak. 1995. Development of antioxidant capacity in tissues of the chick embryo. Comp. Biochem. Physiol. 112B:711-716.
Goerzen, P.R., W.L. Julsrud and F.E. Robinson. 1996. Duration of fertility in ad libitum and feedrestricted caged broiler breeders. Poult. Sci. 75:962- 965.
Havenstein, G.B., P.R Ferket and M.A. Qureshi. 2003a. Growth, livability, and feed conversion of 1957 versus 2001 broilers when fed representative 1957 and 2001 broiler diets. Poult. Sci. 82:1500- 1508.
Havenstein, G.B., P.R Ferket and M.A. Qureshi. 2003b. Carcass composition and yield of 1957 versus 2001 broilers when fed representative 1957 and 2001 broiler diets. Poult. Sci. 82:1509-1518.
Katanbaf, M.N., E.A. Dunnington and P.B. Siegel. 1989. Restricted feeding in early and late-feathering chickens. 2. Reproductive responses. Poult. Sci. 68:352-358.
Latshaw, J.D. and M. Osman. 1974. A selenium and vitamin E responsive condition in the laying hen. Poult. Sci. 53:1704-1708.
Leeson, S. and J.D. Summers. 2001. Nutrition of the Chicken. University Books, Guelph.
Paton, N.D., A.H. Cantor, A.J. Pescatore and M.J. Ford. 2000a. Effect of dietary selenium source, level of inclusion and length of storage on internal quality and shell strength of eggs. Poult. Sci. 79:75-116.
Paton, N.D., A.H. Cantor, A.J. Pescatore, M.J. Ford and C.A. Smith. 2000b. Effect of dietary selenium source and level on inclusion on selenium content of incubated eggs. Poult. Sci. 70(Suppl. 1):40.
Renema, R.A. and F.E. Robinson, 2003. The impact of varying nutrient allocation from photostimulation on carcass and reproductive traits of conventional and high-yield broiler breeder females. Poult. Sci. 82(Suppl. 1):4.
Renema, R.A., F.E. Robinson and G.M. Fasenko. 2001. Effects of feeding regimen and strain on fertility of broiler breeder hens as indicated by the perivitelline layer sperm penetration assay. Poult. Sci. 80(Suppl. 1):172.
Robinson, F.E., J.L. Wilson., M.W Yu, G.M. Fasenko and R.T Hardin. 1993. The relationship between body weight and reproductive efficiency in meattype chickens. Poult. Sci. 72:912-922.
Rustad, M.E. and F.E. Robinson. 2002. Broiler growth potential and parent stock body weight targets 1972-2001. Poult. Sci. 82(Suppl. 1):52.
Siegel, P.B., E.A. Dunnington, D.E. Jones, C.O. Ubosi, W.B. Gross and J.A. Cherry. 1984.
Phenotypic profiles of broiler stocks fed two levels of methionine and lysine. Poult. Sci. 63:855-862.
Surai, P.F. 1999. Tissue-specific changes in the activities of antioxidant enzymes during the development of the chicken embryo. Brit. Poult. Sci. 40:397-405.
Surai, P.F. 2000. Organic selenium: Benefits to animals and humans, a biochemist’s view. In: Biotechnology in the Feed Industry, Proceedings of Alltech’s 16th Annual Symposium (K.A. Jacques and T.P. Lyons, eds). Nottingham University Press, UK, pp. 205-260.
Surai, P.F. 2002. Selenium in poultry nutrition 2. Reproduction, egg and meat quality and practical applications. World’s Poult. Sci. 58:431-450.
Surai, P.F. and J.E. Dvorska. 2001. Is organic selenium better for animals than inorganic sources? Two different scenarios in stress conditions. Feed Mix 9:8-10.
Surai, P.F. and N.H.C. Sparks. 2001. Comparative evaluation of the effect of two maternal diets on fatty acid, vitamin E and carotenoid in the chick embryo. Brit. Poult. Sci. 42:252-259.
Surai, P.F., I. Ionov, T. Kuklenko, I. Kostjuk, A. MacPherson, B. Speake, R. Noble and N.H.C. Sparks. 1998. Effect of supplementing the hen’s diet with vitamin A on the accumulation of vitamins A and E, ascorbic acid and carotenoids in the egg yolk and in the embryonic liver. Brit. Poult. Sci. 39:257-263.
Surai, P.F., R.C. Noble and B.K. Speake. 1999. Relationship between vitamin E content and susceptibility to lipid peroxidation in tissues of the newly hatched chick. Brit. Poult. Sci. 40:406-410.
Ursiny, F., M. Maiorino and A. Roveri. 1997. Phospholipid hydoperoxide glutathione perioxidase (PHGPx): More than an antioxidative enzyme. Biomed. Environ. Sci. 10:327-332.
Whitehead, C.C. 2000. Nutrition: the integrative science. Brit. Poult. Sci. 41:5-15.
Yu, M.W., F.E. Robinson, R.G. Charles and R. Weingardt. 1992. Effect of feed allowance during rearing and breeding on female broiler breeders. 2. Ovarian morphology and production. Poult. Sci. 71:1750-1761.
Author: ROBERT A. RENEMA
Department of Agricultural, Food, and Nutritional Science, University of Alberta, Edmonton, Alberta, Canada







.jpg&w=3840&q=75)