Introduction
Highly pathogenic avian influenza virus (HPAIV) H5N1 persists in natural reservoirs of aquatic birds and waterfowl. In 1996 it was discovered that infected wild geese in Southern China were responsible for transmitting the virus to poultry [1]. In 2003, HPAIV H5N1 began to spread from Southeast Asia to other regions, and in the past decade it has caused numerous outbreaks on poultry farms in over 60 countries in Asia, Africa, and Europe. While the virus remains highly pathogenic to poultry and humans, it has also evolved to become pathogenic to domestic ducks beginning in 2002, and has killed wild waterfowl beginning with the outbreaks in Qinghai Lake in 2005 [2–5]. HPAIV H5N1 has caused severe agricultural and economic burdens with hundreds of millions of poultry culled, and poses a serious public health threat with over 370 human deaths reported since 2003 [6, 7].
HPAIV H5N1 was first transmitted to Africa in 2006 and spread quickly, with outbreaks reported in Nigeria, Egypt, Cameroon, and other African countries [8]. Notably, since then it has become endemic only in Egypt, which is the largest poultry producing country of the Arab nations [9], with regularly reported outbreaks, enormous economic losses in poultry industry, and confirmed human cases with highmortality [10]. While breeders' farms may have strict biosecurity, most commercial farms and household backyards, which comprise the majority of poultry producers in the country, practice poor biosecurity measures [11]. In addition, Egyptian customers rely heavily on live bird markets, where birds of different species and ages and from various locations are offered for trade and slaughter, providing numerous opportunities for viral transmission [12]. Since early control plans including culling infected birds, implementing quarantine measures, and movement restrictions have failed to contain the virus since its emergence in early 2006, control strategies have shifted to massive vaccination programs with inactivated H5N1 or H5N2 viruses, virus surveillance in poultry sectors, and preemptive culling of infected birds [11– 13]. These new strategies have significantly reduced outbreaks in recent years. However, surveillance data reveal continuous and wide circulation of the virus in vaccinated and non-vaccinated commercial farms, household backyards, and live bird markets [14]. In addition, the virus may be undergoing genetic divergence, evolving into multiple genotypes. Remarkably, the H5N1 isolates circulating in Egypt that originated from the Qinghai Lake H5N1 viruses possess several critical genetic hallmarks, including the mutations in HA154-156, where a glycosylation site is missing, and PB2627K [15, 16]. The first renders the virus capable of transmission in mammals, and the second increases viral replication in humans, causing concern that HPAIV H5N1 variants may emerge in Egypt with increased potential for transmissibility in mammals [17].
The HPAIV H5N1 viruses circulating in Egypt since 2006 are highly pathogenic to chickens and can be lethal to ducks [12]. Since becoming endemic in Egypt, the virus has spread from farm to farm, even under immune pressure produced from vaccinated H5 viruses, which may lead to its evolution and mutation [14]. There is limited understanding of the viral pathogenicity and pathogenesis of the current Egyptian H5N1 viruses, the most common cause of outbreaks in poultry in the country.
In this study we report a recent outbreak of the HPAIV H5N1 in commercial poultry farms and backyards in Sharkia Province, Egypt. Our data indicate that the mortality rates were 22.8– 30% in broilers and layers that were vaccinated once with inactivated H5 vaccines, in contrast to the mortality rate of 0.14% in layers which were vaccinated twice. The mortality rates for two backyard flocks of ducks were 28.5 and 40%, respectively, and none of the ducks were vaccinated. Using immunohistochemistry staining we detected viral nucleoprotein in multiple organs and tissues, including trachea, lung, brain, spleen, pancreas, liver, proventriculus, bursa of Fabricius, and testis. The resultant HPAI H5N1 viruses A/chicken/Egypt/IT-1/2013, A/chicken/Egypt/IT 2/2013, and A/duck/Egypt/IT-3/2013 were isolated, and their HA and NS genes analyzed by comparing with the H5N1 viruses previously isolated in Egypt, neighboring countries and worldwide.
Materials and Methods
Viruses
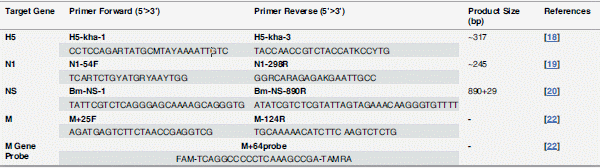
HPAIV H5N1 strains were isolated from chicken and duck tissue samples in commercial poultry farms or backyards in Sharkia Province, Egypt, from February to May 2013. Tissue samples were collected and ground in DMEM cultural medium and centrifuged at 500xg for 10 min. The supernatants were collected and inoculated into 9- to 11-old embryonated chicken eggs, which were then incubated at 37°C. Allantoic fluids were collected and tested with a standard hemagglutination (HA) assay. For HA positive samples, viral RNA was extracted and viral subtypes were identified by reverse transcriptase-polymerase chain reaction (RT-PCR) using a set of subtype-specific primers for viral genes (Table 1), and then further confirmed using nucleotide sequencing. The viruses were aliquoted and stored at -80°C. All animal work was performed with accomplice to the rules and approved ahead by the ethics committee for animal studies at Zagazig University, Egypt.
Table 1. Sequences of the Oligonucleotide Primers Used in the Study.
RNA Extraction, RT-PCR, and Realtime RT-PCR
Viral RNA was extracted from allantoic fluids with Blood/Liquid Sample Total RNA Rapid Extraction Kit (Bioteke Corporation, China) following manufacturer’s instructions. Approximately 1 μg of RNA (5 μl) was reverse transcribed to cDNA using cDNA DiaStarRT Kit (Solgent Co., Korea) according to the manufacturer’s protocol. The cDNA were used as templates for PCR with specific primers to H5 and N1 [18, 19], which was carried out for 35 cycles. PCR amplicons were subjected to electrophoresis in a 1.5% agarose gel, and fragments of H5 subtype HA and N1 subtype neuraminidase (NA) genes of expected sizes were detected (Fig 1F).
For realtime RT-PCR, total RNA were prepared from various tissue and serum samples and reverse transcribed to cDNA. TaqMan-based realtime RT-PCR (Applied Biosystems, USA) was performed with 1 μl of cDNA in a volume of 25 μl to measure viral Mgene transcripts in the tissues using a set ofM gene specific primers and probe (Table 1). The probe was labeled with 5’-FAM and 3’-TAMRA. The reaction was run with ABI 7500 and each assay was repeated three times with positive and negative controls included with the tested samples.
Viral Gene Cloning, Sequencing, and Phylogenetic Analysis
Viral RNA extracted from the allontoic fluids was reverse transcribed with Superscript III Reverse Transcriptase (Invitrogen, USA) and a Uni-12 primer [20]. The cDNA was amplified by PCR with Taq DNA polymerase (Takara, Japan) and primers specific for influenza virus gene segment [20]. PCR products were cloned into a TA cloning vector (Invitrogen, USA) and the nucleotide sequences determined by DNA sequencing.
Phylogenetic analyses of the HA and NS gene were based on nucleotides 791–1100 (309 bases) of HA and 39–704 (666 bases) of NS. All gene sequence data of known H5N1 strains were collected from the NCBI Influenza Database. Multiple alignments were constructed using ClustalW Multiple Alignment with the MegAlign module of DNAStar software (Lasergene version 7.2, DNASTAR, Madison, WI, USA). The neighbor-joining method with Kimura twoparameter distances was used for constructing the phylogenetic tree using the Mega 5.0, and the tree was rooted to the A/goose/ Guangdong/1/1996 virus sequence. The reliability of the internal branches was assessed by the p-distance substitution model and 1000 bootstrap replications. The HA and NS genotypes was determined using the Influenza A Virus FluGenome web server, (http://www.flugenome.org/) [21].
Immunohistochemistry
Freshly collected tissues were fixed by 10% formalin and embedded in paraffin. For histopathology, cross-sections were prepared at 5 μm and stained with hematoxylin and eosin (HE). For immunohistochemistry (IHC) staining, a duplicate 4-μm section was cut, mounted on positively charged SuperFrost Plus microscope slides, dewaxed, and rehydrated. Antigen retrieval was performed by pressure-cooking for 25 mins in pH 6 citrate buffer. Endogenous peroxidases were blocked using 3% H2O2 for 15 mins. Non-specific bindings were blocked using normal goat serum at 1:10 in TBS for 15 mins. Slides were incubated overnight at 4°C with primary rabbit anti-influenza A nucleoprotein (NP) polyclonal antibody (Catalog ID19382; LifeSpan Biosciences, WA) at 1:50 dilution. The EnVision AP (DAKO K1396, Carpinteria, CA) detection system and nuclear fast red (DAKO K1396) were used as chromogens. Sections were counterstained with Mayer’s hematoxylin. Positive and negative control sections were included in each IHC run. All tissues were systematically screened for microscopic lesions. The intensity of viral antigen staining in each section was also scored as following: (-), no antigen staining; (+), infrequent; (++), common; and (+++), widespread staining.
Statistical Analysis
Data were collected and continuous variables were analyzed using one-way analysis of variance (ANOVA), then comparison of means was carried out with Duncan’s multiple range tests (DMRT) and summarized as mean ± standard deviation.
Results
Outbreaks of HPAIV H5N1 in commercial poultry farms and backyards
Beginning in February 2013, chickens and ducks in commercial poultry farms and backyards in Sharkia Province, Egypt, became sick with symptoms including ecchymosis on the shanks and feet, cyanosis of the comb and wattles, subcutaneous edema of head and neck in chickens, and nervous signs (torticollis) in ducks, characteristics of HPAIV infection, as shown in Fig 1A–1D. Birds began to die within 48–72 hrs of the onset of illness. The mortality rates for two unvaccinated backyard flocks of ducks consisting of 14 and 20 birds were 28.5 and 40%, respectively (Table 2). On the other hand, the mortality rates were 22.8 to 30% in broilers and layers that were vaccinated once with inactivated H5 vaccines, and 0.14% in layers that were vaccinated twice (Table 2). The flock sizes of the commercial chicken farms ranged from 3,900 to 5,000 for both broilers and layers.
Nine- to11-day-old chicken embryonated eggs were inoculated with samples from ground pulmonary, tracheal, and testicular tissues of dying or dead chickens or ducks. Embryos with extensive hemorrhage died within 48 hrs post inoculation. Severely congested embryos were observed in eggs inoculated with testicular tissues after the first passage (Fig 1E). Allantoic fluids were tested for hemagglutination (HA) reactivity with a routine HA assay and positive titers were detected. Further viral RNA was prepared from the allantoic fluids, which was reverse transcribed to cDNA for amplification of viral HA and NA fragments with polymerase chain reaction (PCR) using primers specific for theH5N1 HA and NA genes (Table 1). Results indicated that the isolates wereHPAIVH5N1 positive. The PCR results showed specific bands in 1.5% agarose gel at 317(Fig 1F) and 245 base pairs in size (data not shown) for HA and NA, respectively. HPAIV subtype H5N1 was detected in34 of 46 birds including both ducks and chickens (Table 2) with a detection rate of73.9%, including 30 of 42 (71.4%) chickens and 4 of 4 (100%) ducks, respectively (Table 2). Further nucleotide sequencing of the purified PCR products confirmed the isolates to be of the HPAIVH5N1 subtypes, and three viral isolates were designated as A/chicken/Egypt/IT-1/2013, A/chicken/Egypt/IT-2/2013, and A/duck/ Egypt/IT-3/2013. The relevant nucleotide sequences were deposited at the NCBI GenBank database with deposition numbersKJ192204, KJ192205, KJ192206,KP311329, and KP311330 for the three viral strains, respectively.
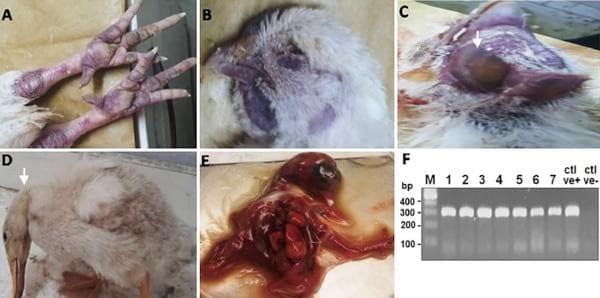
Fig 1.Naturally infected chickens and ducks by HPAIV H5N1. (A) Chickens: subcutaneous hemorrhages in non-feathered skin. (B) Chickens: cyanosis of comb & wattles. (C) Chickens: facial edema. (D) Ducks: nervous signs, torticollis. (E) Infected chicken embryos: chicken embryos were inoculated with supernatants of homogenized testicular tissue of naturally infected layer chickens; the inoculated embryos died within 48hrs with severe congestion and hemorrhages after the first passage. (F) PCR amplification for HA gene showing bands of 317bp in size. First lane: Molecular marker of 100 bp, Lane 1–7: Positive samples, Ctrl +ve: Positive AIV subtype H5N1, Ctrl-ve: Negative control.
Table 2. The Outbreaks of HPAIV H5N1 in the Egyptian Commercial Poultry Farms and the Virus Identification.
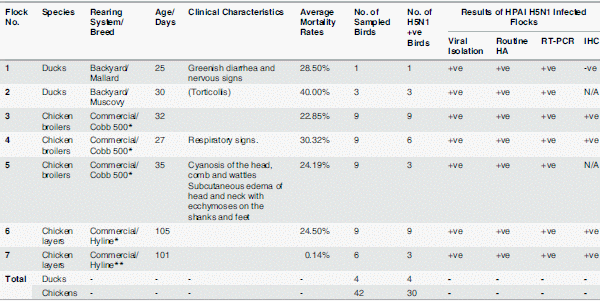
* Chicken broiler and layer flocks were vaccinated once with H5 inactivated vaccine.
** This chicken layer flock was vaccinated two times with H5 inactivated vaccine.
Backyard ducks were not vaccinated.
N/A: Not applied
+ve: Positive
Viral RNA in chicken and duck tissues
Chicken and duck tissues including trachea, lung, liver, spleen, intestine, brain, testis, and serum were taken from sick or dead birds for total RNA preparation. After reverse transcription, cDNA of the samples were subjected to TaqMan-based semi-quantitative realtime RT-PCR (qRT-PCR) [22]with primers specific to theM gene to examine viral RNA in each organ type.
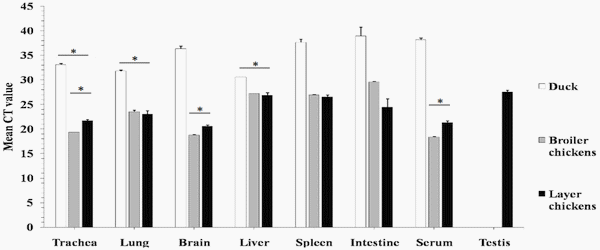
As shown in Fig 2, viral RNA of the HPAIV H5N1 Mgene was detected in all tissues tested both in chickens and ducks with the exception of testis, which was only positive for chicken layers. Higher levels of viral RNA were in general detected in tissues of broiler and layer chickens, including trachea, lung, spleen, intestines, brain, and serum, than in those of ducks (Fig 2).
However, these samples from different birds cannot be compared because they came from natural outbreaks with uncertain timing of the course of infection. In broiler chickens, however, higher viral RNA levels appeared in brain, trachea, and serum samples. No significant differences in viral RNA levels were observed in various tissues of ducks, except for higher levels detected in trachea, lung, and liver tissues (Fig 2).
Fig 2.Detection of M gene transcripts by RT-PCR. Results were expressed as Ct values; Ct values lower than (35) were considered positive; Ct values between 15 and 25 were considered strong positive, while those between 25 and 30 were considered moderate positive; Ct values between 30 and 35 were considered weak positive. The tests were performed in triplicates for each sample and the Ct numbers are average with 3xSD (* P<0.05).
Viral nucleoprotein detected in organs of chickens
Organs and tissues from sick or dead chickens from farms where outbreaks of HPAIV H5N1 occurred were prepared for cross-section slides with routine paraformaldehyde fixation, and subjected to IHC. An antibody to HPAI H5N1 nucleoprotein (NP) was used to stain the samples for detection of viral NP in tissues of infected birds.
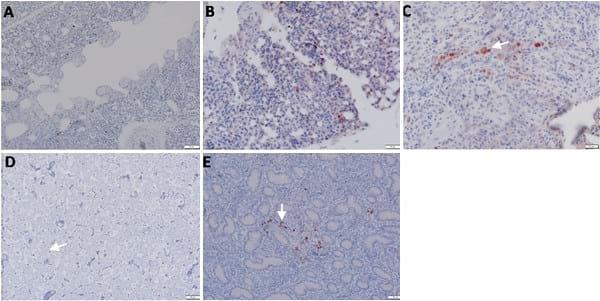
As shown in Table 3 and Figs 3 through 7, viral NP was detected in all tested tissues including brain (Fig 4), pancreas and spleen (Fig 5), proventriculus and bursa of Fabricius (Fig 6), and lung and liver (Fig 7). Remarkably, the viral antigen was also detected in testicular tissue in between seminephrous tubules, even sticking to heads of sperm inside them (Fig 7E). The viral antigen was often associated with histologic lesions, although it was also observed in areas without detectable lesions.
Staining of most organs revealed a common feature characteristic of the viral antigen detected in their endothelial and mononuclear cells, which suggests that viral pathogenesis of the HPAIV H5N1 may be associated with endothelial invasion, and that the virus could be carried by infected monocytes. We identified viral NP in endothelial (Fig 3D) and epithelial (Fig 3F) cells of the trachea, neurons, glial cells of Perkinji cell layer (Fig 4C and 4D) and endothelial cells of brain (Fig 4E), lymphocytes and mononuclear cells of spleen (Fig 5D), acinar epithelium of pancreas (Fig 5F), glandular epithelium of proventriculus (Fig 6B), and lymphocytes of the follicular layer of the bursa (Fig 6D). A summary of viral antigen staining in various tissues examined is shown in Table 3.
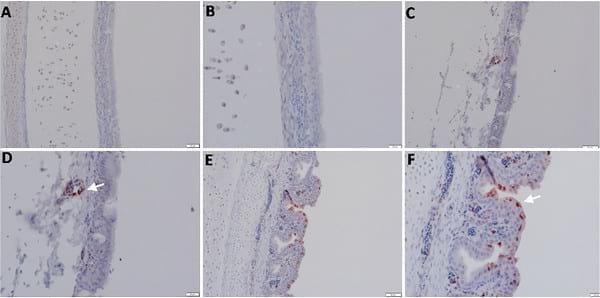
Fig 3.Detection of viral NP antigen in trachea by IHC. (A and B) Trachea of non-infected birds (X = 50 and 20μm, respectively). (C and D) Endothelial cells of trachea of infected birds (X = 50 and 20μm, respectively). (E and F) Epithelial cells of trachea of infected birds (X = 50 and 20μm, respectively).
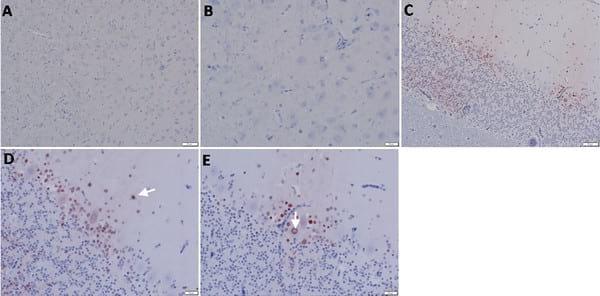
Fig 4.Detection of viral NP antigen in brain by IHC. (A and B) Brain of non-infected birds (X = 50 and 20μm, respectively). (C) Perkinji cell layer of cerebellum of infected birds (X = 50μm). (D) Neurons and glial cells of perkinji cell layer of cerebellum of infected birds (arrow) (X = 20μm). (E) Endothelial cells of perkinji cell layer of cerebellum of infected birds (arrow) (X = 20μm).
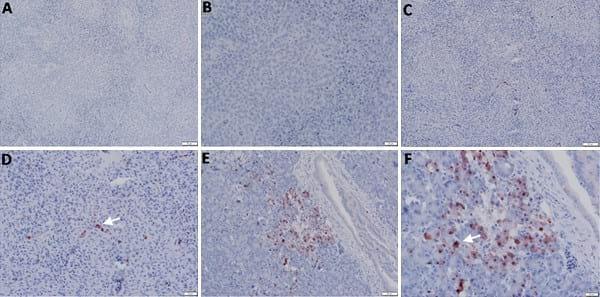
Fig 5. Detection of viral NP antigen in spleen and pancreas by IHC. (A and B) Spleen of non-infected birds (X = 50 and 20μm, respectively). (C) Lymphocytes in spleen of infected birds (X = 50μm). (D) Lymphocytes and mononuclear cells in spleen of infected birds (arrow) (X = 20μm). (E and F) Acinar epithelium of pancreas of infected birds (X = 50 and 20μm, respectively).
Fig 6. Detection of viral NP antigen in proventriculus and bursa of Fabricius by IHC. (A) Proventriculus of infected birds (X = 50μm). (B) Glandular epithelium of proventriculus (arrow) (X = 20μm). (C and D) Lymphocytes in follicular layer of bursa of Fabricius of infected birds (arrow) (X = 50 and 20μm, respectively).
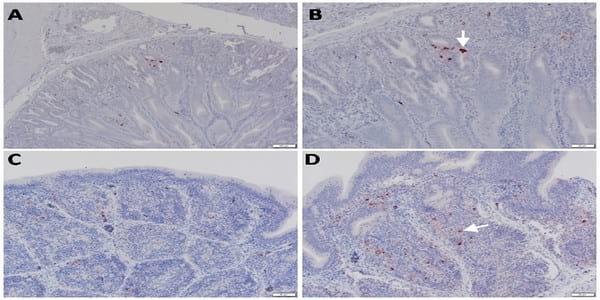
Fig 7. Detection of viral NP antigen in lung, liver and testisby IHC. (A) Lung of non-infected birds (X = 50μm). (B and C) Macrophages and mononuclear cells in lungs of infected birds (arrow) (X = 50 and 20μm, respectively). (D) Liver sinusoids of infected birds (inside van Kuppfer cells) (arrow) (X = 50μm). (E) Testis of infected birds (between seminephrous tubules of testicular tissue) (arrow) (X = 50μm).
Table 3. Summary of Virus Distribution in Tissues of the HPAIV H5N1-Infected Chickens IHC scoring system. (-) no viral antigen detected; (+) sporadic; (++) common; (+++) widespread

IHC scoring system: (-) no viral antigen detected; (+) sporadic; (++) common; (+++) widespread
We prepared samples from dead ducks and performed the same IHC staining on duck tissues as chicken tissues with the anti NP antibody. However, we failed to detect viral NP in all tissues examined (data not shown). Our failure could be attributed to the preparation of the samples, since they were collected from dead ducks in the field. Although viral RNA was still present and live virus isolated successfully (Figs 1 and 2), no sufficient viral antigens existed in the tissues due to prolonged exposure at ambient temperature.
Sequence characterization of the HPAIV H5N1 HA and NS genes
Genomic sequences of the HA genes from the isolated viruses were sequenced and the cleavage sites were compared with those of known H5N1 viruses. We found that the HA of our isolates encodes a multibasic amino acid motif, 321-PQGERRRK/KR_GLF-333, at the HA cleavage site, which is a characteristic feature of all HPAIV H5N1 strains. Interestingly, we found that one of our isolates has a substitution of amino acid (R325K) at this cleavage site, to make it PQGEKRRK/KR_GLF. A phylogenetic tree was constructed, indicating that the HA gene belongs to (5J) genotype (Fig 8A). Our isolates formed a uniform cluster, together with the HPAIVH5N1 viruses from Egypt isolated in 2009, 2010, 2011, 2012, and 2013; however, this cluster was not identical to the HPAIV H5 strains used for commercial vaccine development in Egypt, and was also phylogenetically distant from the viruses isolated from Egypt in 2006, 2007, and 2008.
We also sequenced the NS genomic segments, which are 822 nucleotides in length and encode for NS1 and NS2/NEP proteins. A phylogenetic tree was constructed indicating that the NS1 gene of our isolates belongs to the NS1EQinghai Lake-like genotype (Fig 8B). Our isolates also formed a uniform cluster, together with the HPAIVH5N1 viruses from Egypt isolated in 2010, 2011, 2012, and 2013; however, this cluster was phylogenetically distant from other HPAIV H5N1 isolated from Egypt in 2006, 2007, 2008, and 2009.
We found that the NS1 protein of our H5N1 viruses had a deletion of the 80TMASV84motif and was 225 amino acids in length. The NS1 had a glutamate at position 92 (92E) and its carboxyl terminus harbored the E-S-E-V motif, both of which have been identified as pathogenic determinants of highly pathogenic viruses [23].
Fig 8. Phylogenetic analysis of nucleotide sequences of the HA and NS genes. (A) Phylogenetic analysis of HA gene nucleotide sequences at the cleavage site. The sequences of known HPAIV HA genes were obtained from GenBank. Vaccine H5 strains were included in the tree and marked with solid circles. The tree was constructed with multiple alignment of a 309 base-nucleotide sequence of HA genes using the Neighborhood-joining method in MEGA5. The tree topology was evaluated by 1,000 bootstrap analyses. The viruses isolated in this study are marked with solid triangle. (B) Phylogenetic analysis of NS gene on the basis of nucleotide sequences of the complete coding region (822 bases). The tree was constructed using Neighborhood joining method with bootstrap values calculated for 1,000 replicates and cut off value of 50%. Sequences of the isolates from this study are marked with solid triangles.
Discussion
In this report we demonstrated that the HPAIV H5N1 is still circulating in commercial poultry farms and backyards and causing outbreaks with significant mortality to both chickens and domestic ducks in Egypt. Our data showed that vaccination with H5N1 and H5N2vaccines may provide significant protection, especially to layers which were vaccinated twice. For both broilers and layers in commercial farms, there appears to be protection with even one-time vaccination when compared to a mortality of over 70% in non-vaccinated chickens [24]. Unfortunately, we did not perform serological test to evaluate the immunological status or vaccine efficacy in chickens of the commercial flocks, and therefore are unable to assess how well the mortality is correlated to the vaccination. Therefore, we cannot conclude emphatically that the much lower mortality in the commercial layers, which were vaccinated twice, was absolutely attributed to the boosted immunity.
Vaccine efficacy may best be assessed in the setting of natural outbreaks, which apparently differs from that designed experimentally. In naturally infected free-living birds, the clinical and pathologic manifestations of an HPAIV infection may be influenced by multiple factors including the age of the bird, the dosage of virus and routes of viral exposure, the presence of concomitant infections, and the levels of immunity acquired from vaccination or during previous exposure to influenza viruses [25, 26], which could be significantly different from trials with experimental challenges. Data about vaccine protection and efficacy from natural outbreaks may be more valuable when case studies are carefully planned and serological survey is thoroughly performed and assessed. Even though the mortalities were lowered, the pathogenicity of the infection appeared to be severe, and the virus was highly virulent in sick chickens as shown in Fig 1 and Table 2.
Pathogenicity of infection by HPAIV H5N1 has been studied experimentally in chickens and domestic ducks. Vietnam HPAIV H5N1 caused high mortality in two- and four-week-old SPF White Leghorn chickens (G. gallusdomesticus) (100%, 48/48) with mean death times (MDT) from 36 to 48 hrs, and two- and five-week-old Pekinwhite ducks (Anasplatyrhynchus) (98.4%, 63/64) with MDTs from 2.7 to 4.4 days [27]. Pathogenicity of the Egyptian HPAIV H5N1 have been tested experimentally only in domestic ducks [28]. While A/ck/Egypt/08 killed 8/8 (100%) with an MDT of 4.1 days, A/ck/Egypt/07 killed 4/8 (50%) with an MDT of 7 days, indicating that HPAIV H5N1 isolates differ in their virulence. Although these two isolates are considered to have evolved from the same origin [28], they are far apart within the clade 2.2 in the phylogenetic tree and have evolved different pathogenicity since the HPAIV H5N1 of clade 2.2 was introduced into Egypt. On the other hand, pathogenicity observed in these experimental challenges cannot be directly compared with that in natural outbreaks. Apparent differences exist between SPF birds with a certain age group infected experimentally by the intranasal route (IN) and poultry of various ages in commercial farms by natural exposure.
The mortality rates in our report were lower in duck and even much lower in once- and twicevaccinated chickens during the outbreaks. However, for sick birds the symptoms were severe. Systemic infection was observed and nervous disorders were obvious in both chickens and ducks, and infection was confirmed by viral antigen detection.
The current H5N1 viruses, A/chicken/Egypt/IT-1/2013 and A/chicken/Egypt/IT-2/2013, clearly demonstrate their pantropism in tissues and organs as virulent HPAIV strains. The viral RNA and NP antigen were detected in multiple tissues, including trachea, lung, brain, liver, spleen, pancreas, intestines, serum, proventriculus, bursa of Fabricius, and testis in infected chickens, similar to those observed in chickens infected with the Vietnamese H5N1 virus [28]. Neurotropism of the HPAI H5N1 virus has been previously described [29–32]. Our study shows that high levels of viral RNA were detected in the brains of infected chickens (Fig 2) and that viral NP antigen was observed in the nuclei of neurons and glial cells of the brain, clear signs that the virus replicates in brains.We could also conclude that even though vaccination may lower mortality rates, it does not change pantropism of HPAIV H5N1 in sick birds.
Different pathways have been proposed by which the HPAI H5N1 virus infects the central nervous system (CNS) in chickens. It has been hypothesized that the virus could reach the CNS through the olfactory nerves [33], the peripheral nervous system [34, 35], or even the bloodstream [36]. Interestingly, we observed the viral NP antigen in endothelial cells of brain, which provides direct evidence that the HPAIV H5N1 likely invades the CNS by replicating in blood vessels in the brain, and contributes to the development of severe nervous symptoms. Based on our evidence, we consider this to be one of the routes for HPAIV H5N1 to invade CNS. Severe CNS disorders in birds are probably one of the main causes for mortality when neurons are infected; massive edema due to virus infection-induced altered vascular permeability and multi-organ failure are commonly blamed for high mortality in HPAIV infected birds.
Mechanisms for viral penetration of the blood-brain barrier in the brain have been investigated previously. The virus may invade neurons through the opening of endothelial cell junctional complexes (para-cellular route) [37] or through vesiculo-tubular structures (transcellular route) [38]. It could reach the vessels in the brain through the bloodstream, or via a “Trojan horse mechanism” where viral particles are transported through infection of leukocytes and/or mononuclear cells [39]. In our study, viral RNA or antigen was easily detected in blood or sera and infected macrophages and monocytes, which suggests that the endothelial cells may play a crucial role in viral penetration of the blood-brain barrier, leading to severe necrotizing encephalopathy and death. Our finding that endothelial cells of the cerebellum were also strongly positive in viral NP antigen supports this hypothesis (Fig 4).
In this report, the viral antigen was strongly expressed in the acinar epithelium of pancreas, giving rise to the possibility of a potential role of the pancreas in viral pathogenesis. Evidence showed that the virus also replicated in lymphocytes of follicular layer of the bursa, which may be significant in inducing immunity against the virus, a key process for recovery of sick birds from infection. A remarkable finding in our study is the isolation of virus from testicular tissue samples; infection at this site resulted in embryos with severe hemorrhages and congestion (Fig 1E) leading to death within 48hrs post-infection. Moreover, high viral RNA levels were detected from testicular tissue (Fig 2), with viral NP antigen expressed in between seminephrous tubules (Fig 7E). Strikingly, the viral antigen was also detected inside these seminephrous tubules sticking to sperm. However, sexual transmission for apparently healthy cocks to spread HPAIV H5N1 during the incubation period is probably a scenario of low or unlikely probability. It is likely that sperm collected from infected cocks with healthy appearance could disseminate disease to both uninfected birds and farm handlers and workers either via natural insemination or during application of artificial insemination. Thus, the findings in this report not only provide insights on HPAI H5N1 viral pathogenicity and pathogenesis in natural infection, but also emphasize the importance of biosecurity and proper handling in poultry agriculture.
Acknowledgments
The authors thank Mohamed Azawy for kindly providing clinical samples, Jan Shivers for helping with immunohistochemistry staining, M. Afifi for statistical analysis, and Dr. Rob Porter of Veterinary Diagnostic Laboratory for photographing of slides.
Author Contributions
Conceived and designed the experiments: IH SM CJC ZX. Performed the experiments: IH SM AA EBI AS. Analyzed the data: IH ZRZ CJC JC ZX. Contributed reagents/materials/analysis tools: IH ZRZ CJC JC ZX. Wrote the paper: IH ZX.
This article was originally published in PLoS ONE 10(5): e0120061. doi:10.1371/journal. pone.0120061. This is an Open Access article distributed under the terms of the Creative Commons Attribution License. References
1. Xu X, Subbarao K, Cox NJ, Guo Y. Genetic characterization of the pathogenic influenza A/Goose/ Guangdong/1/96 (H5N1) virus: similarity of its hemagglutinin gene to those of H5N1 viruses from the 1997 outbreaks in Hong Kong. Virology. 1999; 261(1):15–9. PMID: 10484749
2. Sims L, Ellis T, Liu K, Dyrting K, Wong H, Peiris M, et al. Avian influenza in Hong Kong 1997–2002. Avian diseases. 2003; 47(s3):832–8.
3. Sims L, Domenech J, Benigno C, Kahn S, Kamata A, Lubroth J, et al. Origin and evolution of highly pathogenic H5N1 avian influenza in Asia. Veterinary Record. 2005; 157(6):159. PMID: 16085721
4. Chen H, Smith G, Zhang S, Qin K, Wang J, Li K, et al. Avian flu: H5N1 virus outbreak in migratory waterfowl. Nature. 2005; 436(7048):191–2. PMID: 16007072
5. Liu J, Xiao H, Lei F, Zhu Q, Qin K, Zhang X-w, et al. Highly pathogenic H5N1 influenza virus infection in migratory birds. Science. 2005; 309(5738):1206-. PMID: 16000410
6. Claas EC, Osterhaus AD, van Beek R, De Jong JC, Rimmelzwaan GF, Senne DA, et al. Human influenza A H5N1 virus related to a highly pathogenic avian influenza virus. The Lancet. 1998; 351 (9101):472–7. PMID: 9482438
7. WHO. Cumulative number of confirmed human cases for avian influenza A(H5N1); 2003–2014.
8. Enserink M. H5N1 moves into Africa, European Union, deepening global crisis. Science. 2006; 311(5763):932-. PMID: 16484460
9. Freiji M. The poultry industry in the Arab world—present and future. Lohmann Information. 2008: 43: 4–52.
10. WHO. H5N1 avian influenza: Timeline of major events.; 2011.
11. Abdelwhab E, Hafez H. An overview of the epidemic of highly pathogenic H5N1 avian influenza virus in Egypt: epidemiology and control challenges. Epidemiology and infection. 2011; 139(05):647–57.
12. Aly M, Arafa A, Hassan M. Epidemiological findings of outbreaks of disease caused by highly pathogenic H5N1 avian influenza virus in poultry in Egypt during 2006. Avian diseases. 2008; 52(2):269–77. PMID: 18646456
13. Hafez M, Arafa A, Abdelwhab E, Selim A, Khoulosy S, HassanM, et al. Avian influenza H5N1 virus infections in vaccinated commercial and backyard poultry in Egypt. Poultry science. 2010; 89(8):1609–13. doi: 10.3382/ps.2010-00708 PMID: 20634514
14. Abdelwhab E, Selim A, Arafa A, Galal S, Kilany W, Hassan M, et al. Circulation of avian influenza H5N1 in live bird markets in Egypt. Avian diseases. 2010; 54(2):911–4. PMID: 20608538
15. Herfst S, Schrauwen EJ, Linster M, Chutinimitkul S, de Wit E, Munster VJ, et al. Airborne transmission of influenza A/H5N1 virus between ferrets. Science. 2012; 336(6088):1534–41. doi: 10.1126/science. 1213362 PMID: 22723413
16. Imai M, Watanabe T, Hatta M, Das SC, Ozawa M, Shinya K, et al. Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in ferrets. Nature. 2012; 486(7403):420–8. doi: 10.1038/nature10831 PMID: 22722205
17. Neumann G, Macken CA, Karasin AI, Fouchier RA, Kawaoka Y. Egyptian H5N1 Influenza Viruses— Cause for Concern? PLoS pathogens. 2012; 8(11):e1002932. doi: 10.1371/journal.ppat.1002932 PMID: 23166487
18. Njouom R, Aubin JT, Bella AL, Demsa BM, Rouquet P, Gake B, et al. Highly pathogenic avian influenza virus subtype H5N1 in ducks in the Northern part of Cameroon. Vet Microbiol. 2008 Aug 25; 130(3–4):380–4. doi: 10.1016/j.vetmic.2008.02.012 PMID: 18417301
19. Tsukamoto K, Ashizawa T, Nakanishi K, Kaji N, Suzuki K, Shishido M, et al. Use of reverse transcriptase PCR to subtype N1 to N9 neuraminidase genes of avian influenza viruses. J Clin Microbiol. 2009 Jul; 47(7):2301–3. doi: 10.1128/JCM.02366-08 PMID: 19403772
20. Hoffmann E, Stech J, Guan Y, Webster R, Perez D. Universal primer set for the full-length amplification of all influenza A viruses. Archives of virology. 2001; 146(12):2275–89. PMID: 11811679
21. Lu G, Rowley T, Garten R, Donis RO. FluGenome: a web tool for genotyping influenza A virus. Nucleic acids research. 2007; 35(suppl 2):W275–W9.
22. Spackman E, Senne DA, Myers T, Bulaga LL, Garber LP, Perdue ML, et al. Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. Journal of clinical microbiology. 2002; 40(9):3256–60. PMID: 12202562
23. Jackson D, Hossain MJ, Hickman D, Perez DR, Lamb RA. A new influenza virus virulence determinant: the NS1 protein four C-terminal residuesmodulate pathogenicity. Proceedings of the National Academy of Sciences. 2008; 105(11):4381–6. doi: 10.1073/pnas.0800482105 PMID: 18334632
24. Grund C, Abdelwhab E-SM, Arafa A-S, Ziller M, Hassan MK, Aly MM, et al. Highly pathogenic avian influenza virus H5N1 from Egypt escapes vaccine-induced immunity but confers clinical protection against a heterologous clade 2.2. 1 Egyptian isolate. Vaccine. 2011; 29(33):5567–73. doi: 10.1016/j. vaccine.2011.01.006 PMID: 21244859
25. Keawcharoen J, Van Riel D, van Amerongen G, Bestebroer T, Beyer WE, Van Lavieren R, et al. Wild ducks as long-distance vectors of highly pathogenic avian influenza virus (H5N1). Emerging infectious diseases. 2008; 14(4):600. doi: 10.3201/eid1404.071016 PMID: 18394278
26. Bröjer C, Ågren EO, Uhlhorn H, Bernodt K, Mörner T, Jansson S, et al. Pathology of natural highly pathogenic avian influenza H5N1 infection in wild tufted ducks (Aythya fuligula). Journal of veterinary diagnostic investigation. 2009; 21(5):579–87. PMID: 19737752
27. Pfeiffer J, Pantin-Jackwood M, To T, Nguyen T, Suarez D. Phylogenetic and biological characterization of highly pathogenic H5N1 avian influenza viruses (Vietnam 2005) in chickens and ducks. Virus research. 2009; 142(1):108–20.
28. Wasilenko J, Arafa A, Selim A, Hassan M, Aly M, Ali A, et al. Pathogenicity of two Egyptian H5N1 highly pathogenic avian influenza viruses in domestic ducks. Archives of virology. 2011; 156(1):37–51. doi: 10.1007/s00705-010-0813-y PMID: 20882306
29. Brown JD, Stallknecht DE, Berghaus RD, Swayne DE. Infectious and lethal doses of H5N1 highly pathogenic avian influenza virus for house sparrows (Passer domesticus) and rock pigeons (Columbia livia). Journal of veterinary diagnostic investigation. 2009; 21(4):437–45. PMID: 19564491
30. Pantin-Jackwood M, Swayne D. Pathogenesis and pathobiology of avian influenza virus infection in birds. Revue scientifique et technique (International Office of Epizootics). 2009; 28(1):113–36.
31. Tang Y, Wu P, Peng D, Wang X, Wan H, Zhang P, et al. Characterization of duck H5N1 influenza viruses with differing pathogenicity in mallard (Anas platyrhynchos) ducks. Avian Pathology. 2009; 38(6):457–67. doi: 10.1080/03079450903349147 PMID: 19937535
32. Goletic T, Gagic A, Rešidbegovic E, Kustura A, Kavazovic A, Savic V, et al. Highly pathogenic avian influenza virus subtype H5N1 in mute swans (Cygnus olor) in central Bosnia. Avian diseases. 2010; 54(s1):496–501.
33. Majde JA, Bohnet SG, Ellis GA, Churchill L, Leyva-Grado V, Wu M, et al. Detection of mouse-adapted human influenza virus in the olfactory bulbs of mice within hours after intranasal infection. Journal of neurovirology. 2007; 13(5):399–409. PMID: 17994424
34. Tanaka H, Park C-H, Ninomiya A, Ozaki H, Takada A, Umemura T, et al. Neurotropism of the 1997 Hong Kong H5N1 influenza virus in mice. Veterinary microbiology. 2003; 95(1):1–13.
35. Matsuda K, Park C, Sunden Y, Kimura T, Ochiai K, Kida H, et al. The vagus nerve is one route of transneural invasion for intranasally inoculated influenza A virus in mice. Veterinary Pathology Online. 2004; 41(2):101–7. PMID: 15017022
36. Mori I, Komatsu T, Takeuchi K, Nakakuki K, Sudo M, Kimura Y. Viremia induced by influenza virus. Microbial pathogenesis. 1995; 19(4):237–44. PMID: 8825911
37. Lossinsky A, Shivers R. Structural pathways for macromolecular and cellular transport across the blood-brain barrier during inflammatory conditions. Review. Histology and histopathology. 2004; 19(2):535–64. PMID: 15024715
38. Liu NQ, Lossinsky AS, Popik W, Li X, Gujuluva C, Kriederman B, et al. Human immunodeficiency virus type 1 enters brain microvascular endothelia by macropinocytosis dependent on lipid rafts and the mitogen-activated protein kinase signaling pathway. Journal of virology. 2002; 76(13):6689–700. PMID: 12050382
39. Verma S, Lo Y, Chapagain M, Lum S, Kumar M, Gurjav U, et al. West Nile virus infection modulates human brain microvascular endothelial cells tight junction proteins and cell adhesion molecules: Transmigration across the in vitro blood-brain barrier. Virology. 2009; 385(2):425–33. doi: 10.1016/j.virol. 2008.11.047 PMID: 19135695














.jpg&w=3840&q=75)