INTRODUCTION
Salmonella has been linked to numerous foodborne illnesses from consumption of poultry products. The FoodNet preliminary data for 2016-2019 identified 25,866 infections, 6,164 hospitalizations and 122 deaths due to foodborne illnesses in United States of which Salmonella contribute about 17.1% of total foodborne infections (Tack et al., 2020; Vaddu et al., 2020). They also reported 5 most common Salmonella serotypes in the US - Enteritidis, Typhimurium, Newport, Javiana, and Infantis. Moreover, European Food and Safety Authority (EFSA) reported that Salmonella Typhimurium and S. Enteritidis are the most common serovars to cause outbreaks of salmonellosis (EFSA, 2017). Approximately 70% of Salmonella foodborne illnesses were associated with consumption of turkey and chicken products in the US (CDC, 2018). While poultry processors have incorporated numerous antimicrobial interventions at processing, it is necessary to reduce both the concentrations and prevalence of Salmonella at preharvest to reduce the incoming load of Salmonella for the processing interventions to achieve USDA FSIS microbiological performance standards. Growing incidence of antibiotic resistant strains posing threats worldwide led to the ban of subtherapeutic use of antibiotics particularly in food animal, making disease control difficult (Levy and Marshall, 2004). With increasing consumer preference for No Antibiotics Ever (NAE) and organic niche markets, poultry producers need alternatives to antibiotics that could reduce foodborne pathogen colonization, and promote overall growth performance of chickens (Lillehoj et al., 2018; Yadav et al., 2020). Various preharvest and postharvest antimicrobial interventions are being used during poultry production and processing to reduce Salmonella prevalence, thus controlling Salmonella associated foodborne illnesses. Some of the most commonly used feed additives during poultry production are organic acids, probiotics, prebiotics, herbal extracts, and nutraceutical products (Abudabos et al., 2018; Teng and Kim, 2018; Yadav and Jha, 2019; Adhikari et al., 2020).
Poultry production has been one of the rapidly developing industries worldwide attributable to a significant increase in the global consumption of poultry meat and poultry products. However, the profitability of poultry sector depends mostly on cost of feed production which contributes to around two-third cost of total poultry production (Berrocoso et al., 2017; Yadav, 2017). The growing discrepancy between demand and supply of traditional corn-soybean meal feed ingredients has compelled poultry producers to search for alternative feedstuffs. However, it is important that these alternative feedstuffs be economical, easily available, and effective in maintaining feed efficiency and overall growth performance of birds to maintain higher poultry production (Yadav and Jha, 2021). Therefore, the exploration of nonconventional substitute for soybean and corn and natural plant based antimicrobial agents that can serve as alternative feedstuff are requisite (Ravindran, 2013). In addition, the products, which could help in reduction of colonization of foodborne pathogens such as Salmonella, as substitutes for antibiotics are necessary to meet the food safety regulatory requirements.
In poultry canola meal, rapeseed meal, and carinata meal are frequently being used as SBM alternative (Rahman et al., 2018; Yadav et al., 2021). The usefulness of these feed ingredients is limited by the presence of antinutritional factors that can limit its inclusion levels in animal feeds and thus reduce the likelihood of antimicrobial properties of the secondary metabolites in animal feeding. Previous studies with inclusion of rapeseed or canola in poultry feed reported a decrease in growth performance at higher inclusion levels (> 25%) which might be due to the presence of glucosinolates, tannins, sinapine, erucic acid, and other antinutritional factors present in these feedstuffs (Mushtaq et al., 2007; Min et al., 2011; Gopinger et al., 2014). Interestingly, allyl isothiocyanate (AITC) inclusion in a controlled release of AITC with modified atmosphere packaging at concentration of 0.6 μg/h was effective against foodborne pathogens and has been used in food preservation (Shin et al., 2010). Most of the plants from Brassicaceae family have natural antimicrobials in the form of secondary metabolites as a part of their self-defense against pathogens (Ishida et al., 2014). This antimicrobial activity of plant could be exploited as an alternative to antibiotics or AGPs in poultry production to reduce Salmonella colonization of the chickens.
Glucosinolates (GLs) are secondary metabolites present exclusively in plants of Brassicaceae family, and isothiocyanates (ITCs) are bioactive compounds produced after the hydrolysis of GLs in the presence of enzyme myrosinase (Romeo et al., 2018). ITCs are known to be effective against foodborne pathogens, plant pathogens, and human pathogens including those of resistant phenotypes (Romeo et al., 2018). Many in vitro antimicrobial studies evaluated the efficacy of ITCs against various foodborne pathogens in chicken meat products (Shin et al., 2010; Olaimat et al., 2014). However, their effectiveness as antimicrobials in vivo for control of bacterial pathogens has not been studied yet. Few limitations for in vivo model could be the instability of ITCs, as naturally they are present as GLs and are activated by physical breakage while chewing by animals, leading to contact of myrosinase, an only known glucohydrolase that can break GLs to produce ITCs (Tripathi and Mishra, 2017). GLs are further classified based on their aglycon chemical structure. For the present study, a common type of glucosinolate-sinigrin was considered, and the antimicrobial effect of its derivative ITC specifically allyl isothiocyanate (AITC) was studied for its mitigation effect on Salmonella Typhimurium. Lu et al. (2016) studied antimicrobial effects of AITC from Wasabia japonica against E. coli and S. aureus, although the mechanism of action was unknown. Other studies using synthetic AITC showed significant antimicrobial activity against Campylobacter jejuni, Escherichia coli O157:H7, Escherichia coli, P. aeruginosa, and S. aureus (Borges et al., 2015; Lu et al., 2016; Kaiser et al., 2017). In the study by Lu et al. (2016), the antimicrobial activity of AITC against Salmonella Typhimurium showed the minimum inhibitory concentration (MIC) and minimum bactericidal concentrations (MBC) of AITC ranging from 100 to 150 ppm respectively. Another study showed concentration of AITC required for MIC varies from 50-1,000 ppm depending on bacteria being tested (Shofran et al., 1998). Although the mechanism of action of the compound was not clearly understood, AITC was reported to show bactericidal effect on E. coli by disrupting the cell membrane integrity and inhibited Shiga toxin production in enterohemorrhagic E. coli. AITC was able to inhibit growth of Methicillin-resistant S. aureus (MRSA) as well as reduced the metabolic activity of P. aeruginosa in mature biofilm (Dias et al., 2014; Kaiser et al., 2017; Romeo et al., 2018). Kim and Lee (2009) showed that naturally produced AITC from horseradish and synthetic AITC had similar antimicrobial activity. Similarly, Romeo et al. (2018) reported that AITC was more effective ITCs against gram-negative bacteria compared to gram positive bacteria. Some of the reported mechanisms of action for bactericidal effects of AITC included disruptions in membrane integrity, redox reaction of bacteria and/or alteration in their metabolism. Thus, the present study explored the use of glucosinolate containing feed ingredients such as rapeseed and canola in vivo in chickens under a Salmonella Typhimurium challenged setting.
Rapeseed, also known as mustard seed or oil seed rape (Brassica napus), is ranked as third highest oil seed crop produced in the world (Nega and Woldes, 2018). Rapeseed constitutes about 40% oil by weight, whereas the residual by-product is of interest to animal nutritionists for its rich nutrient profile, high protein, and balanced amino acid contents compared to other oil crops or cereals (Nega and Woldes, 2018). Regardless of having rich nutrient profile, few limitations still exist, which restricts their inclusion levels in animal diet due to the presence of the antinutritional factors such as GLs, sinapine, its derivatives, tannin, phytic acid, and crude fiber (Zeb, 2002). Various strategies such as physical, chemical, biological, and crop breeding are being used to decrease the GLs in rapeseed and to allow for an increase of the inclusion level in animal ration (Ashnie et al., 2015). Previous studies reported that rapeseed meal (RSM) can be incorporated up to 10-15% RSM or 50% replacement of SBM in broiler diets (Nega and Woldes, 2018). In the present study, an inclusion of 10% rapeseed was incorporated to evaluate the growth performance in comparison to traditional SBM, whereas the inclusion of 30% rapeseed was to investigate if its inherent high concentration of glucosinolates could contribute to any antimicrobial effect against Salmonella Typhimurium.
Canola meal (CLM) often used interchangeably with rapeseed meal vary in the quality of nutrient and antinutritional content where canola was developed through plant breeding of Brassica napus (rapeseed) to reduce glucosinolates (< 30 μmol/g in the fat-free meal) and erucic acid (< 2% of the total fatty acid in oil) and labeled as “double zero” cultivar of rapeseed (Bell, 1993; Anderson et al., 2013); the by-product (meal) from this canola variety could replace conventional SBM protein up to 20%, which is higher than any meals from other Brassica species (Spragg and Mailer, 2007). In contrast, RSM and carinata meal contain higher amounts of glucosinolate, although both feed ingredients are rich sources of protein and sulfur containing amino acids required for animal nutrition (Tripathi and Mishra, 2017). Thus, the present study included 20% CLM based on literature to minimize its negative impact on growth of chickens, whereas 40% CLM was used almost at a 100% replacement for SBM to assess its antimicrobial effect against Salmonella colonization.
Although the impact of including rapeseed and canola meal in broiler diet has been reported, the in vivo effect of the glucosinolate containing feed on Salmonella Typhimurium colonization has not been elucidated. We hypothesize that inclusion of glucosinolate-containing feed ingredients will maintain broiler growth performance in comparison to corn-SBM based diet while reducing Salmonella colonization in broilers. The objective of the study was to evaluate 1) the effects of feeding corn-SBM based control diet, rapeseed (10 and 30%), canola meal (20 and 40%), and AITC (500 and 1500 ppm) on growth performance, intestinal permeability, intestinal histomorphology, and gene expression of tight junction proteins and 2) the antimicrobial effects of dietary treatments in broilers challenged with Salmonella.
MATERIALS AND METHODS
Bird Management
The study was conducted at the Poultry Research Center, University of Georgia, Athens, GA. This study was approved by the Institutional Animal Care and Use Committee. A total of 640 day (d)-old broiler male chicks (Cobb 500) were obtained from a commercial hatchery (Cobb-Vantress Inc, Cleveland, GA). Birds were randomly assigned to 8 treatments with 8 replicates of 10 birds per cage in a total of 64 identical battery cages. In addition, some extra birds were kept in separate cages for serum collection required for gut permeability analysis. All the birds had ad libitum access to feed and water, and the temperature and lighting programs were set as per the recommendation of Cobb 500 broiler management guide (Cobb 2018).
Dietary Treatments
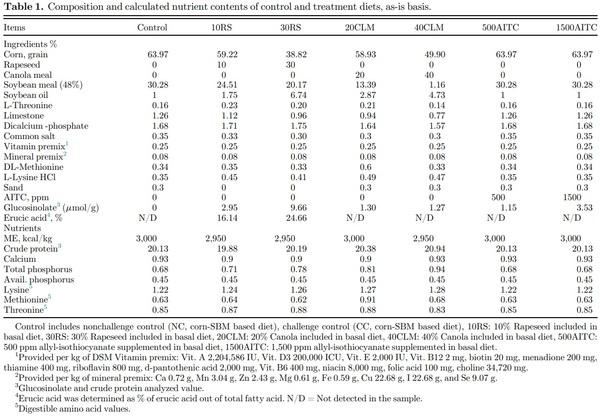
The treatment diets were formulated to meet the nutritional requirements for the starter phase fed from 1 to 21 d of age (Table 1). Whole rapeseed was obtained locally from Athens Seed Co., Watkinsville, GA which provides rapeseed of Dwarf Essex variety originating from Idaho. Rapeseed was further processed in our farm by use of roller to decrease the particle size and increase the surface area. This process improved the solubility of feed ingredient, absorption, and distribution, ultimately increasing the bioavailability (Zajac et al., 2021). Solvent-extracted canola meal with less than 3% oil was obtained in pellet form (Godfrey’s feed, Madison, GA) and ground before incorporating in feed. The glucosinolate content of rapeseed and canola meal containing feed ingredients were tested by HPLC using the AOCS AK 1-92 method performed by BioProfile Testing Laboratories, LLC, Minneapolis, MN. Commercially available allyl isothiocyanate (95% AITC, Sigma-Aldrich, St. Louis, MO) was added to a corn-SBM based basal diet to prepare the AITC treatment diets. The 8 dietary treatments included nonchallenge control (NC, cornSBM based diet), challenge control (CC, corn-SBM based diet), 10% Rapeseed meal in basal diet (10RS), 30% Rapeseed meal in basal diet (30RS), 20% Canola meal in basal diet (20CLM), 40% Canola meal in basal diet (40CLM), 500 ppm (500 mg/kg feed) of allyl- isothiocyanate (AITC) in basal diet (500AITC), and 1,500 ppm of AITC in basal diet (1500AITC). The previous studies have shown that rapeseed can only be included 5−15% in diet, so our study used lower level as 10% that will not affect the growth of birds. Similarly, canola was previously included 15−25% in diet so, our study used 20% at lower level. The higher levels of both ingredients were included to test for its anti-Salmonella effect as they will be high in glucosinolate. On d 1 posthatch, all the birds except NC group were challenged with 0.5 mL of nalidixic resistant Salmonella Typhimurium (5 x 107 CFU/mL) while birds in the NC group were orally gavaged with same amount of 0.1% peptone water.
Growth Performance
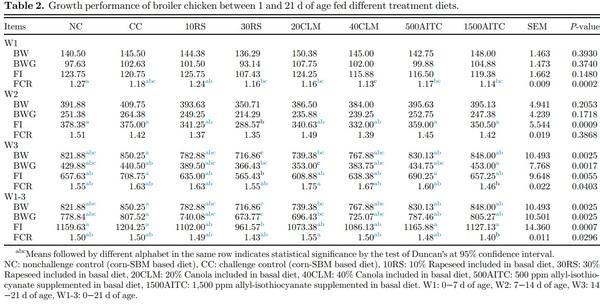
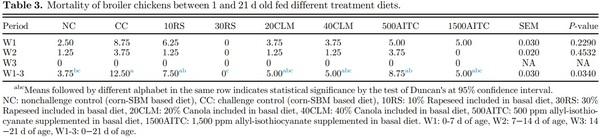
For the growth parameter, the average body weight (BW) and feed intake (FI) of all the birds in each replicate cage were weighed on d 0, 7, 14, and 21. The BW and FI were used to calculate body weight gain (BWG) and feed conversion ratio (FCR; Table 2). Birds were checked twice daily for mortality, and dead birds were removed and recorded to adjust FI and FCR (Table 3).
Salmonella Challenge Protocol
A nalidixic acid resistant Salmonella Typhimurium (STNR) strain was obtained from the U.S. National Poultry Research Center, United States Department of Agriculture, Athens, GA (Vaddu et al., 2020). In brief, a loopful culture from STNR glycerol stocks was streaked onto Brilliant Green Sulfa agar plates (BGS; Difco, Sparks, MD) supplemented with 200 ppm nalidixic acid (Sigma-Aldrich, St. Louis, MO) to get BGSNA and incubated at 37°C for 18−24 h. A single colony from overnight incubated plate was further streaked onto a fresh BGSNA to facilitate adequate resuscitation of bacteria. A day prior to the challenge, a single colony was transferred to trypticase soy broth (TSB; 100 mL) supplemented with nalidixic acid (200 ppm) and incubated for 18−22 h at 37°C. The culture was centrifuged for 10 min at 7,838 g at 4°C. The supernatant was discarded, and the obtained pellet was resuspended in 10 mL of peptone water (PW, 0.1%). The resuspended aliquots were combined and centrifuged again. The supernatant was discarded, and PW (3 mL) was added and vortexed to resuspend the pellet. The OD value of the culture suspension was adjusted with PW to obtain STNR concentration to ca. 7.7 log CFU/mL. The STNR suspension was transported on an ice chest and used for the challenge.
On one-day posthatch, all birds except NC were challenged with STNR by oral gavage with a one-time dose of 0.5 mL (7 log CFU/ bird) STNR. Whereas birds in the nonchallenge group received 0.5 mL of sterile PW. The gavage solution was serially diluted and plated on BGSNA to confirm the dose of challenge.
Sample Collection and Analysis Performed
Salmonella Enumeration Ceca, liver, spleen, kidney, and feces were aseptically collected on 4 (3 d postchallenge, dpc), 7 (6 dpc), 14 (13 dpc), and d 21 (20 dpc) for Salmonella enumeration. On each sampling day, both ceca from 64 birds (1 bird/cage) were aseptically cut and placed in sterile filter bags (Nasco, Weber Scientific, Hamilton Township, NJ). Internal organs, including kidney, liver, and spleen, were collected aseptically in sterile whirl-pak bags (VWR, Radnor, PA). Fecal samples were collected by placing aluminum foil sheets under each pen for 2 h to obtain fresh fecal samples. Collected feces were aseptically transferred to 15 mL sterile conical tubes (Thermo Fisher Scientific, Waltham, MA), and their weights were recorded to calculate fecal shedding as log CFU/g. All the samples were placed in an ice chest to cool the samples and stored at 4°C until further analyses.

Cecal Colonization of STNR Chilled buffered peptone water (supplemented with 200 ppm NA; BPWNA; 10 mL) was added to each cecal sample and homogenized in a stomacher (Neutec Group Inc., Farmingdale, NY) for 1 min. After an aliquot (1 mL) of the homogenized sample was serially diluted, appropriate dilutions were spread plated on BGSNA, and the plates were incubated for 18 to 24 h at 37°C. Typical colonies were counted, and Salmonella populations were expressed as log10 CFU/mL of ceca.
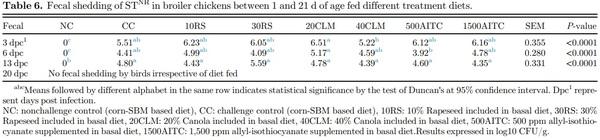
Fecal Shedding of STNR An initial dilution (1:10) of the fecal sample was prepared in BPWNA and thoroughly mixed. An aliquot (10 μL) of the initial dilution was added to microtiter wells containing 90 μL of PW, and serial dilutions were prepared. From each dilution, an aliquot (10 μL) was spotted on duplicate BGSNA plates, and the plates were then incubated for 18 to 24 h at 37°C. Typical Salmonella colonies were enumerated and expressed as log10 CFU/g of feces.
Translocation of STNR to Internal Organs For the recovery of STNR from internal organs, kidney (ca. 2 g), liver (ca. 4 g), and whole spleen were separately added to 5 mL of BPWNA and homogenized in a stomacher and incubated at 37°C for 18 to 24 h. Collected fecal sample in BPWNA were also used to observe prevalence of STNR. The enriched samples were streaked onto BGSNA plates for isolation. BGSNA plates showing typical STNR were counted as positive and the prevalence (%) was calculated.
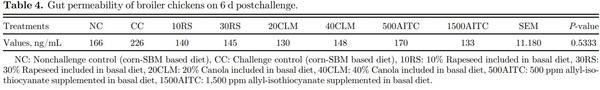
Intestinal Permeability To determine whether translocation of S. Typhimurium into the spleen and liver was associated with altered intestinal permeability, intestinal paracellular permeability was determined by measuring fluorescein isothiocyanate dextran (FITC-d) in the serum according to modified method described by Yadav et al. (2020). Briefly, on 6 dpc birds (6 birds/treatment) were orally administered with 0.5 mL FITC-d (2.2 mg/mL; MW 4,000; SigmaAldrich, St. Louis, MO) per bird. A total of 6 birds from each treatment group were euthanized by cervical dislocation 1 h postoral administration of FITC-d, and 3 mL of blood was collected from each bird (Vuong et al., 2021). Blood from ten extra birds that were reared in the same room but not part of the experiment was collected, and the serum from these birds was used to prepare the standard curve. The serum samples were processed in the dark to avoid direct light exposure. The serum samples and standards were transferred to a black 96-well microplate (Ref. 655077; Greiner Bio-One, Monroe, NC) to measure FITC-d using a spectrophotometer (Spectramax, M5; Molecular Devices, San Jose, CA) at an excitation wavelength of 485 nm and an emission wavelength of 528 nm (Table 4).
Intestinal MorphologyOn 20 dpc, one bird/cage was euthanized by cervical dislocation, and approximately 2 cm mid-sections of the duodenum loop, jejunum, and ileum (with reference to meckel’s diverticulum) were collected, and the digesta were flushed out from the intestine tissues with phosphate buffered saline (PBS; National Diagnostics, Atlanta, GA). The tissues were fixed in 10% phosphate-buffered formalin and stored until further processing. The samples were dehydrated, diaphanized, and embedded in ethanol, xylol, and paraffin respectively. Multiple slices of 4.0 mm sections were made and stained with standard hematoxylin-eosin solution. Each slide was observed for villi height (VH) and crypt depth (CD) by a microscope (BZ-X800, Keyence Inc., Itasca, IL) at 20X magnification. The measurement was done using ImageJ (Version I.X.) application, and VH: CD ratio was calculated.
Gene Expression
On 20 dpc, mid-jejunum tissue samples were collected from 1 bird/cage, flushed with PBS, immediately snap frozen in liquid nitrogen, and stored at -80°C until further processing.
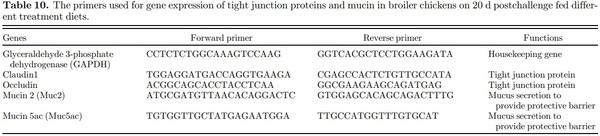
A 40 to 60 mg of jejunum tissue was taken and homogenized in QiAzol lysis reagent (Qiagen, Valencia, CA) to extract total RNA according to manufacturer’s manual. After extraction, the total RNA was quantified using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA). The RNA was normalized and reverse transcribed by high-capacity cDNA conversion kits (Applied Biosystems, Foster city, CA). The cDNA was diluted with 1:5 nuclease free water and stored until running real-time PCR reaction. The reaction was performed using SYBR Green master mix with a Step One thermocycler (Applied Biosystem). Each gene expression sample was run in duplicates, and the target genes were expressed in 2−ΔΔCt method (Livak and Schmittgen, 2001).
Statistical Analysis
All the data except organ translocation of STNR were analyzed using PROC GLM procedure of SAS (Version 9.4; SAS Institute Inc., Cary, NC). One-way ANOVA was performed for growth performance, permeability, histology, and gene expression data, and Duncan’s multiple-range test was used to distinguish significant difference among the treatments set at P < 0.05. For the cecal colonization and fecal shedding data, CFU was first converted to log10 CFU and analyzed similar to other data. For the mortality, the data were transformed to √ (n + 1) and analyzed same as growth performance. Similarly, for the organ translocation and fecal prevalence of STNR, the presence/absence data were transformed to √ (n + 1) and presented as number of positive birds out of total birds (% of positive birds).
RESULTS AND DISCUSSION
Canola or rapeseed are widely cultivated throughout the world, and by-product after oil production (meal) is frequently used as an alternative source of dietary protein in poultry diets as a replacement for SBM (Rahman et al., 2018; Yadav et al., 2021). The crude protein levels of rapeseed and canola meal used in the present study after analysis were 21.7% and 39.3%, respectively. Zeb (2002) reported a relatively higher protein level (36−38%) for rapeseed meal in his study compared to one in the current study, whereas Bell (1993) reported a similar crude protein content in canola meal (38%) to one in the current study. SBM can be easily incorporated up to 30-35%, whereas rapeseed meal inclusion is limited to 8−10% because of the higher concentrations of antinutritional factors such as glucosinolate and erucic acid (European Food Safety Authority (EFSA), 2008; Nega and Woldes, 2018). Canola is developed by intensive plant breeding to contain reduce antinutritional factors such as glucosinolate and erucic acid to increase inclusion levels of up to 20% without any deleterious effects on the growth performance (Khajali and Slominski, 2012; Payvastagan et al., 2012; Nega and Woldes, 2018).
Growth Performance
In the period from d 1 to 7, chickens fed with 10RS and 30RS did not affect the BW or FI, but those fed with 30 RS, 20CLM, 40CLM, 500AITC, and 1500AITC resulted in lower FCR compared to NC (P = 0.0002). For the period of d 7 to 14, there was a reduction in FI for 30RS fed birds compared to NC, CC, 500AITC and 1500AITC (P = 0.0009). For the period from 14 to 21 d, 30RS fed birds had significantly lower BW compared with CC, 500AITC and 1500AITC (P = 0.002). Similarly, 30RS fed birds showed lower FI compared to CC and 500AITC (P = 0.006). On the contrary, even lower inclusion levels of canola meal (20CLM) showed lower BW, and BWG compared to CC, and 1500AITC (P < 0.05). Moreover, 20CLM showed higher FCR compared to 1500AITC (P = 0.04). Overall, in the period of d 0 to 21, significant reduction in all the growth parameters (BW, BWG, and FI) for 30RS was observed compared to the CC group. Similarly, 20CLM had significantly lower BW and BWG compared to CC.
Glucosinolates and other antinutritional factors can lower feed intake, resulting in reduced growth and higher mortality due to hemorrhagic liver syndrome (Tripathi and Mishra, 2007; Khajali and Slominski, 2012). The diminished BW and FI in 30RS fed birds were expected due to the presence of antinutritional factors such as glucosinolate, as they are highly water soluble and can persist in the rapeseed even after its oil extraction and conversion to meal (Egorova and Lenkova, 2015). Elevated levels of glucosinolates, phytic acid, sinapine, and erucic acid present in the higher inclusion level of rapeseed meal (30RS) might have affected the digestibility, reducing overall growth performance (Tripathi and Mishra, 2007). These antinutritional factors also destroy the intestinal mucosa of the gastrointestinal tract, causing decreased absorption of nutrient and ultimately lowering growth (Egorova and Lenkova, 2015). In the present study, the negative effects were not very pronounced, probably due to the use of low glucosinolate rapeseed (12 μmol/g of glucosinolate) which may go up to 200 μmol/g as found by Taraz et al. (2006). The 30RS showed lower FCR which was desirable, and it could be due to well-balanced amino acids, Ca, P, and minerals in rapeseed compared to other feed ingredients used in the study (Ciurescu, 2009). The growth performance in birds fed canola-based diet was intermediate compared to corn-SBM and rapeseed-based diets (overall BW range 717 g to 850 g). The 20CLM showed significant reduction in BWG and increase in FCR. Similar to the findings of our study, Payvastagan et al. (2012) reported that the inclusion of 20% canola meal did not affect FI but significantly reduced BWG and FCR. This reduced performance of birds might be due to the presence of sinapine, tannins, glucosinolate, and fiber or a decrease in the protein digestibility along with FI, leading to decreased BWG (Gopinger et al., 2014). Another reason of reduced body weight gain could be due to the tannins present in canola meal; the tannins can bind to the protein and proteolytic enzymes and therefore reduce protein digestion and growth (Khajali and Slominski, 2012). Canola also has a high fiber content with lignins and polyphenols (Khajali and Slominski, 2012; Gopinger et al., 2014). The fiber content in the diet containing high inclusion levels of canola meal can reduce the nutrient digestibility of protein and energy (Kocher et al., 2000). In contrast, Karimzadeh et al. (2016) reported that when canola bioactive peptides (CBP) were fed to chicken at 200 or 250 mg/kg, there was significant improvement in BWG and reduction in the FCR. The present study was the first report to the effects of AITC in the diet of broiler chickens. The growth performance for the third week was better for 1500AITC, this could be because of antimicrobial property of AITC to mitigate effect of Salmonella and thus improve feed conversion.
Mortality data showed that birds in CC group had higher mortality (12.5%) compared to NC and 30RS groups (P = 0.03). Higher mortality in CC group might be attributed to their efficient growth performance similar to NC group. Because most of the severely affected birds in CC group which might have reduced an average growth performance died, the growth performance from the surviving birds that were healthier and more resistant to STNR challenge appeared to contribute the overall growth performance of CC group. Compensatory growth after recovery from ST infection might also have contributed to the current findings. Surprisingly, 30RS, despite feeding with high levels of antinutritional and harmful toxic compounds, did not result in any high mortality, whereas control group fed with corn-SBM and lower inclusion levels of rapeseed (10RS) showed significantly higher mortality (P = 0.03). Canola and AITC fed challenged birds had similar mortality as corn-SBM based control group. Because feed exposes broiler chickens to different external factors via the gastrointestinal tract, the gut microbiota are also influenced by the types of diets the chicken consumes (Yegani and Korver, 2008). Divergent bacteria have different substrate requirements for growth; certain feed ingredients positively influence beneficial bacteria, whereas others stimulate an increase in the pathogenicity of opportunistic bacteria present in the intestine (Yadav and Jha, 2019). Thus, the potential role of rapeseed meal at higher inclusion rates in regulating intestinal microbiome could have diminished the harmful effects of Salmonella challenge, resulting in no mortality.
Bacteriological Recovery of STNR in Cecum, Feces, and Internal Organs
Experimental gavage of STNR orally was translocated to different organs and persists in the organ for variable periods of time. Cecal colonization of STNR is presented in Table 5. Cecum is a crucial organ for bacterial colonization due to the slow flow of intestinal contents and a site of water absorption and carbohydrate fermentation. It is also a site of highest concentration of microbiota including harmful bacteria such as E. coli and Salmonella (Yadav and Jha, 2019). In the present study, all birds except the NC group were challenged with 7.0 log CFU/mL on d 1. At 3 dpc, all the STNR challenged birds showed colonization (ca. 5.5−6.0 log CFU/mL) with no differences in colonization among the treatments (P > 0.05). However, by 6 dpc, a significant reduction in cecal colonization of STNR was observed in 30RS and 500AITC treatments (P < 0.0001). This reduction in STNR colonization could be due to the sensitivity of Salmonella to the antimicrobial agents such as phenols, phytic acid, and glucosinolate present in 30RS and a bioactive AITC role in 500AITC. The colonization of STNR increased at 13 dpc compared to 6 dpc for all the treatments except for 30RS. At 13 dpc, 500AITC had significantly lower STNR cecal colonization compared to 20CLM (P < 0.0001). The colonization reduced drastically for all the treatments at 20 dpc, with no difference among the treatments.
Fecal shedding of STNR was also quantified at the same dpc as cecal colonization (Table 6). At 3 dpc, the fecal shedding was reduced for 40CLM compared to 20CLM (P < 0.05). At 6 dpc, 500AITC showed significant reduction in fecal Salmonella shedding compared to 20CLM. No difference for fecal shedding was observed at 13 dpc except NC group which did not shed STNR and confirmed the protocols in place prevented cross contamination between the challenge and nonchallenge treatments. By 20 dpc, the birds were recovered and did not shed STNR.
The intestinal microbiota plays a vital role in the maintenance of gut health. The presence of pathogenic bacteria such as Salmonella disrupts the normal niche of microbiota (Yadav and Jha, 2019). Thus, different dietary interventions are needed to modify the microbiota to enhance growth and mitigate enteric infection caused by pathogenic ones (Kim and Lillehoj, 2019; Yadav and Jha, 2019). In a previous study, the use of camelina seed of Brassicaceae family promoted the growth of the Bifidobacterium which plays a role in maintaining intestinal integrity as well as competitive exclusion of pathogenic bacteria (Sergeant et al., 2014; Zajac et al., 2021). Similar effect might be shown in the present study by higher inclusion of glucosinolate containing feed ingredients such as rapeseed and direct supplementation of AITC.
Salmonella recovery from different internal organs was evaluated on the same dpc as cecal and fecal quantification (Table 7). The recovery of STNR in the spleen was similar among treatments at 3, 6 and 13 dpc, whereas at 20 dpc, greater organ translocation in CC group was observed compared to NC, 10RS, 20CLM, 500AITC, and 1500AITC (P = 0.046). In the liver, no translocation was observed at 3 and 20 dpc, whereas at 6 dpc all the challenged treatment groups had higher prevalence compared to NC group (P = 0.042). At 13 dpc, the prevalence of STNR in the liver increased in 20CLM compared to 10RS (P = 0.027). At 6 dpc, 30RS and CC groups showed lower prevalence compared to 10RS and both levels of canola meal (P = 0.0006). Further no difference was observed in kidney translocation at 13 and 20 dpc. Along with fecal quantification, enriched fecal samples were also used to calculate the prevalence. At 6 dpc, 30RS had lower fecal prevalence compared to both levels of canola meal groups (P = 0.004). At 13 and 20 dpc, significant difference was seen between NC and all other challenged groups, whereas no difference was observed at the end of trial as birds were almost recovered at 20 dpc. The presence of polyphenols and sulfated polysaccharides in the rapeseed could also be the reason for inhibited colonization with rapeseed inclusion as they are known to affect the bacterial lipopolysaccharide in the blood (Cartea et al., 2011). Similarly, Sivula et al. (2008) reported lower translocation of STNR to the spleen, with ceca being the most heavily colonized compared to other organs.
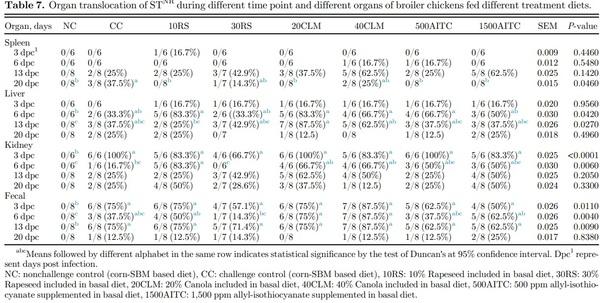
Although liver colonization was higher and cecal colonization increased up to 13 dpc (highest at 6 dpc) in the present study, Sivula et al. (2008) showed higher colonization at 3 dpc. This difference might be due to different challenge days; in the present study, the birds were challenged on d 1, whereas Sivula et al. (2008) challenged the birds at 1 wk of age. Salmonella Pathogenicity Island-1 (SPI-1) which is responsible for internalization of pathogen might vary by age and breed of birds studied. Birds were resistant to STNR at older age, having matured intestinal microbiota as well as immunity (Beal et al., 2006).
Intestinal Health
Intestinal Permeability Appropriate gut barrier function is necessary for overall health of birds (Pastorelli et al., 2013). Optimum tight junction presence and function are necessary for health intestinal integrity, and any damage to tight junctions can disrupt the gut barrier function (Prado-Rebolledo et al., 2017). Birds in both challenge and nonchallenge groups were tested for intestinal permeability on 6 dpc. No significant difference among the treatments was observed, and all the birds had similar gut integrity as nonchallenged birds (NC group; P = 0.533; Table 4).
Under normal conditions, FITC-d will not be found in the serum as it cannot cross the intestinal epithelium barrier. During challenge with STNR, the integrity is compromised, and higher levels of FITC-d quantification in the serum are expected. He et al. (2020) reported no differences in gut permeability at 7 dpc when high nutrient density (CP = 21.8 and ME = 3.16 kcal/g) diet was fed to birds challenged with Salmonella Typhimurium. Similarly, difference in gut permeability at 6 dpc was not observed in the current study contrary to the observations by Leyva-Diaz et al. (2021). The authors reported that Salmonella challenge increased the gut permeability, whereas the treatment with curcumin restored the intestinal barrier parameters with permeability similar to that of nonchallenged control birds. Gut permeability was accompanied by a decrease in intestinal tight junction proteins, whereas in the present study there were no differences in gene expression of tight junction proteins which might be one of the reasons for no significant difference in gut permeability (P > 0.05). The tight junction protein expression and gut permeability could be influenced by the dosage, age of challenge, feeding strategy and STNR virulence (Withanage et al., 2004; Kim et al., 2018). Stecher et al. (2008) reported that inflammation in the intestinal mucosa caused by the presence of pathogenic Salmonella provides a rich source of nutrients that favor virulent and motile Salmonella growth. However, He et al. (2020) stated that providing a nutritious diet helps the birds resist Salmonella colonization by improving gut microbiota and morphology.
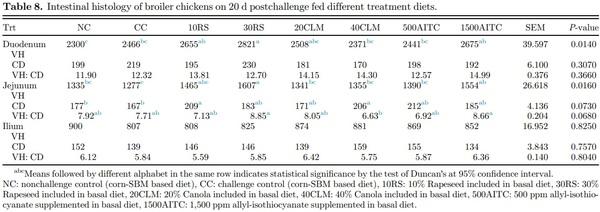
Intestinal Histomorphology At 20 dpc, sections of duodenum, jejunum, and ileum were collected for histomorphology analyses (Table 8). While the VH: CD ratio was similar between the treatments, a significant increase in duodenum and jejunum villus height for 30RS was observed compared to NC, CC, 40CLM, and 500AITC treatments (P = 0.014; P=0.016, respectively). For 10RS and 40CLM groups, the crypt depth at the jejunum was significantly higher compared to NC and CC group (P = 0.07). Jejunum VH: CD ratio was higher in 30RS and 1500AITC compared to 40CLM (P = 0.068). No difference in histomorphology of ileal samples was found among the treatments (P > 0.05).
In comparison to canola meal, the intestinal histomorphology of rapeseed fed birds showed improved VH, CD, and their ratio. This could be due to improved antioxidant activity resulting from higher phenolics contents in rapeseed which could have mitigated the oxidative stress caused by Salmonella (Vuorela, 2005). In contrast to the results of our study, Figueiredo et al. (2003) reported that inclusion of canola meal at 0, 10, 20, 30, and 40% did not show significant differences in the intestinal histomorphology on d 21. A decreased duodenum villus height, jejunum villus height to crypt depth ratio, and increased jejunum crypt depth were observed in the treatments with canola inclusion (20CLM and 40CLM) in the present study. This discrepancy between 2 studies could be due to challenge by STNR in the present study whereas the birds were not Salmonella-challenged by Figueiredo et al. (2003). This also depicts the negative impacts on the histomorphological changes of the intestine caused by Salmonella infection on birds fed canola based diets. Another study by Xu et al. (2012) showed increased villus height under nonchallenged condition with 10% fermented rapeseed meal. Chiang et al. (2010) reported an improved villus height, and VH:CD ratio in the ileum and jejunum of broiler fed fermented rapeseed meal, whereas the present study showed similar results with regards to only jejunum. Consistent with our study, Gopinger et al. (2014) reported a decrease in villus height with 23.6% inclusion of canola meal in the diet. In addition, Gopinger et al. (2014) also found a linear decrease in crypt depth which was not seen in the present study. This could be due to Salmonella challenge which caused an increased cell turnover rate in the intestine by the host in response to the infection.
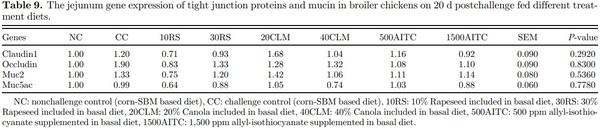
Intestinal Gene Expression Intestinal gene expression was similar among the treatments (P > 0.05) as shown in Table 9 and primer information provided in Table 10. This could be because birds almost recovered from Salmonella infection at 20 dpc. Furthermore, in the present study, birds were challenged on d 1 when the birds have immature intestinal microbiota and improper naive immune system. This could cause the birds in challenged group to face serious consequences, resulting in higher mortality rather than disturbance to intestine function or microbiota dysbiosis (Withanage et al., 2004; Bergmann et al., 2013). He et al. (2020) showed no difference in tight junction proteins and mucin-2 on d 21 same as the present study.
In conclusion, this was the first study to investigate antimicrobial effect of dietary form of rapeseed, canola meal as well as AITC supplementation to the chickens against STNR. Inclusion of rapeseed meal in a chicken diet at a higher level (30%) showed some promising decrease in colonization of Salmonella without increase in bird mortality, decreased FCR, and improved intestinal histomorphology. In the canola meal fed groups, the growth performance, mortality, Salmonella colonization and translocation to different organs, and histomorphology of the jejunum were negatively affected following STNR challenge. AITC at 500 ppm and 1,500 ppm showed better effects for growth performance, and reduction of Salmonella colonization than challenged control group. Further, more research on the mode of action and the improvement in the delivery of AITC through feed such as microencapsulation should be evaluated to increase its efficacy as an antimicrobial against STNR and other pathogens.



















.jpg&w=3840&q=75)