I. INTRODUCTION
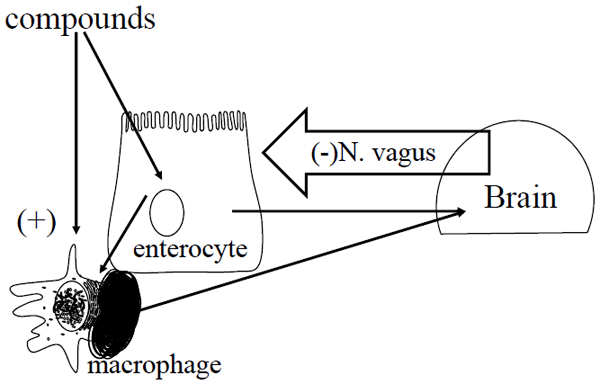
The gut is a very crucial organ for maintaining health. Apart from absorbing nutrients, it is also the barrier against unwanted compounds and germs. The immune system in the intestines plays an important role in this. Immune cells such as inflammatory cells were thought to be central, and until recently, the enterocyte layer was considered a simple physical barrier. Now, it is known that enterocytes are immunocompetent cells as well, in particular in the innate part of the immune system. Enterocytes play an important part in the crosstalk with intestinal microbiota. It is important to realise that as opposed to the systemic counterpart, which is geared towards reaction, the larger mucosal immune system is geared towards tolerance. This is particularly relevant concerning inflammation. Inflammation if not tightly controlled can cause great damage to the intestine itself. On the local level, enterocytes, dendritic cells, macrophages are involved in achieving a balanced response (Wells et al., 2011). Furthermore, the presence of numerous nerve endings suggests involvement of the central nervous system. This was confirmed in the case of post-prandial inflammation. The latter is a low-grade inflammatory response in the intestines after each meal. It is the normal physiological response of the body to a meal, and the degree of inflammation is related to the caloric value, the glyceamic index and specific constituents such as fatty acids and others (Margioris, 2009). If not contained, postprandial inflammation could ultimately lead to unfavourable phenomena such as muscle catabolism, inappetite, and intestinal tissue damage which predisposes for certain pathogens. Because the intestines are constantly exposed to foreign substances such as feed, it is not surprising that the body has evolved an intestinal system to control inflammation and immunity. This is the nervous anti-inflammatory reflex through the nervus vagus which plays a pivotal role in the control and containment of the intestinal inflammatory system, and is therefore essential for health and survival of the animal (Tracey, 2002) (Figure 1). However, this mechanism can be overwhelmed by risk factors such as large amounts of (high) energy feed. This means that gut health in production animals is at risk, and this should be monitored. The determination of the status of intestinal health in general is not that easy, and in particular not in poultry for the absence of suitable biomarkers and methods.
Figure 1 - Schematic representation of the intestinal anti-inflammatory reflex. Feed compounds can give a pro-inflammatory (+) stimulus to enterocytes and macrophages. This leads to the production of pro-inflammatory interleukins (ILs), which also reach the brain. A down (-) regulatory signal is returned to the intestine through the nervus (N.) vagus. Adapted after Niewold, 2013.
II. BIOMARKERS FOR GUT HEALTH IN POULTRY
A major problem in trying to determine intestinal health in vivo is the relative inaccessibility of large parts of the GI-tract. There is a great need for biomarkers for intestinal health, which can be determined in samples obtained in a non-invasive or minimally invasive way, meaning from blood, excreta, saliva, urine, or other bodily fluids. Good candidates should be compounds of the GI-tract itself or related to it. They should be validated as indicators of intestinal health, and reagents and assays should be available. Many biomarkers of (intestinal) inflammation have been validated in humans and experimental animals. Unfortunately, the great evolutionary distance with birds makes that knowledge not directly applicable in poultry. For instance, chickens use a totally unrelated protein (PIT54) for haemoglobin binding instead of the acute phase protein haptoglobin (Wicher and Fries, 2006). Even if a homologous protein is found, immunological reagents often do not cross react. Another complication of chickens is the composition of the excretions, containing a lot of uric acid,
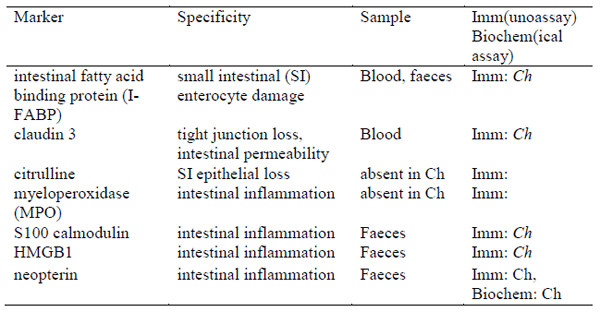
and the fact that biomarkers could derive from the kidneys rather than the intestines. Intestinal markers are related to the physical and immunological intestinal barrier function. Enterocytes and tight junctions between enterocytes are crucial in this respect. Inflammation in the intestinal mucosa is tightly controlled too, because it may cause damage and enhanced permeability. As a consequence, biomarkers for damage to intestinal health could be either constituents of enterocytes such as tight junctions proteins, and intracellular products and proteins, either constitutive or induced. Also, products from inflammatory cells should be useful indicators (Kosek et al., 2013). Concerning the latter, myeloperoxidase (MPO) from neutrophiles has been used extensively in mammals. Faecal MPO is used to establish the degree of intestinal inflammation in humans. Unfortunately, chicken heterophiles do not contain MPO or equivalent activity (Brune et al., 1972). For other faecal inflammatory cell biomarkers such as calmodulin also designated S100, lipocalin 2 and HMGB1, it is not clear if these biomarkers are at all present in chicken. Faecal neopterin is a marker of intestinal inflammation. The advantage of neopterin is that is identical in all species, not requiring species specific reagents. Unfortunately, in chicken excreta neopterin excreted by the kidneys can be derived from inflammatory processes other than the intestines. There are several markers for enterocytes described. Intestinal fatty acid binding protein (I-FABP) is a constitutive cytosolic enterocyte protein of the small intestine, and is a useful marker of enterocyte damage in blood and faeces. The chicken I-FABP gene is present and was expressed only in intestinal tissues (Wang et al., 2005); however, reagents are not available as yet. The same is the case for claudin-3. Finally, the level of circulating citrulline is a parameter for functional enterocyte mass in mammals, but not in chicken (Wu et al., 1995) (Table 1).
Table 1 - Intestinal health biomarkers in mammals, their presence in chicken (Ch), sampling method, and the availability of reagents. In italics: proposed but not proven.
In the case of chronic intestinal diseases in humans, plasma acute phase proteins (APP) such as C-reactive protein (CRP) and haptoglobin are used as markers of intestinal inflammation. It is evident that APP are only good markers for intestinal inflammation in the absence of other inflammatory processes in the body (Margioris, 2009). Several APP, such as haptoglobin can be measured in chicken, using a biochemical method. In any case, it is expected that the level of APP is reciprocal to growth, and that is indeed what is generally observed (Korver et al., 1998).
III. HOW TO REDUCE INTESTINAL INFLAMMATION AND PROMOTE GROWTH
It is clear that increased intestinal inflammation poses a risk to high producing poultry such as broilers. Apart from growth retardation by reduced appetite and muscle catabolism, there is also an increased risk of infections because many pathogens such as Clostridiae benefit from inflammation (Ng et al., 2010). Whereas it is economically hardly feasible to reduce the energy content to reduce the inflammatory stimulus, there are other possibilities. In the past, anti-inflammatory components have been added to feed, and the most prevalent of those were the antimicrobial growth promoters (AGP). Earlier, the beneficial effects of AGP were attributed to their antibiotic character (e.g. Dibner and Richards, 2005), but this is unlikely for a variety of reasons, the main one being the sub therapeutic concentrations used. AGP such as oxytetracycline (OTC) work by inhibition of the intestinal inflammatory response by direct inhibition of inflammatory cells (Niewold, 2007), and indeed there proved to be a perfect correlation between (past) use as AGP and the direct anti-inflammatory properties of antibiotics (Table 2).
Table 2 - The relationship between the direct anti-inflammatory properties of antibiotics and their use as antimicrobial growth promoters (AGP). Adapted after Niewold, 2007

With the increasing pressures to restrict the use of antibiotics either as AGP or as therapeutics, there is a great need for effective alternatives. These alternative additives could be selected on the basis of known anti-inflammatory activity, and tested directly in vivo for their effect on intestinal health and growth. In addition, one could look at the pro- and anti-inflammatory properties of existing normal feed constituents. Intestinal inflammation could be alleviated or prevented by an anti-inflammatory feed composition by for instance removing the pro-inflammatory constituents and maintaining or increasing the levels of anti-inflammatory constituents. New compounds could be selected for anti- and pro-inflammatory properties (and toxicity) using simple in vitro assays available (Wu et al., 2003, Niewold and de Backer, 2010). If found to be effective (and non-toxic) these compounds can be tested in vivo. This approach makes it possible to select effective compounds from large amounts of candidates. Several promising compounds have been successfully selected using these techniques. The preferable sources of compounds are plants (extracts). Plants are seen as natural, and do contain a host of candidate substances. Alternatives should preferably not be (registered) drugs, because regulatory action can be foreseen. As reviewed earlier (Niewold, 2009), there is a long list of feed components with proposed immunomodulatory properties, including macronutrients, polyphenols, essential oils, herbal compounds etc. For most of these compounds, the results reported are quite variable, and are from empirical trials often built on an inadequate theoretical basis. Of only a few compounds the anti-inflammatory activity and associated growth promotion are fairly well established and three examples from different origin are given. Concerning herbal extracts, sanguinarine containing extracts were previously shown to have growth promoting activity in broilers (Vieira et al., 2008). It also had in vitro anti-inflammatory activity (Niewold and De Backer, 2010), which is consistent with the in vivo claim. Furthermore, polyphenols were described as anti-inflammatory (Chen et al., 2006), and indeed anti-inflammatory and growth promoting effects in piglets were demonstrated (Deng et al., 2010), whereas the evidence in chicken is much less clear, which is likely related to the anti-nutritional properties of some polyphenol extracts used. Fat and fatty acids as normal or added feed components are most interesting concerning inflammation. In the first place, the n-3 and n-6 polyunsaturated fatty acids (PUFA), and in particular the ratio between the two are very important (Margioris, 2009). The n-3 PUFA are anti-inflammatory, whereas the n-6 PUFA are pro-inflammatory. A higher 3 to 6 ratio leads to better performance in chicken, and to attenuated growth retardation after inflammatory challenge at the optimum dosage (e.g. Korver et al., 1998). It should be realised, however, that fat is largely responsible for the high caloric value of a meal. This means that when applied in higher concentrations the pro-inflammatory effect may override the anti-inflammatory effect.
IV. CONCLUSION
Inflammation is inversely related to growth (Korver et al., 1998, Niewold, 2007). As discussed here, post-prandial inflammation is a major factor in growth in broilers. Therefore, the research in feed should focus on anti-inflammatory compounds and anti-inflammatory feed composition. Large amounts of new components can easily be screened in the in vitro anti-inflammatory assay before costly feed experiments are performed. In vivo, the anti-inflammatory theory predicts effective compounds to promote growth to coincide with lower levels of markers of inflammation such as the plasma levels of acute phase proteins.
REFERENCES
Brune K, Leffell MS & Spitznagel JK (1972) Infection and Immunity 5: 283-287.
Chen X, Li W & Wang H (2006) Medical Hypotheses 66: 660-663.
Deng Q, Xu J, Yu B, He J, Zhang K, Ding X & Chen D (2010) Archives of Animal Nutrition 64: 12-21.
Dibner JJ & Richards JD (2005) Poultry Science 84: 634-643.
Korver DR, Roura E & Klasing KC (1998) Poultry Science 77: 1217-1227.
Kosek M, Haque R, Lima A, Babji S, Shrestha S, Qureshi S, Amidou S, Mduma E, Lee G, Yori PP, Guerrant RL, Bhutta Z, Mason C, Kang G, Kabir M, Amour C, Bessong P, Turab A, Seidman J, Olortegui MP, Lang D, Gratz J, Miller M & Gottlieb M (2013) American Journal of Tropical Medicine and Hygiene 88: 390-396.
Margioris AN (2009) Current Opinion in Clinical Nutrition and Metabolic Care 12: 129-137.
Ng J, Hirota SA, Gross O, Li Y, Ulke-Lemee A, Potentier MS, Schenck LP, Vilaysane A, Seamone ME, Feng H, Armstrong GD, Tschopp J, Macdonald JA, Muruve DA & Beck PL (2010) Gastroenterology 139: 542-552.
Niewold TA (2007) Poultry Science 86: 605-609.
Niewold TA (2009) In: Recent advances in animal nutrition. P. C. Garnsworthy and J. Wiseman (eds.) Nottingham University Press, Nottingham, UK. pp 191-205.
Niewold TA & de Backer K (2010) In: Duclos M, Nys Y (eds.) Proceedings of the XIIIth European Poultry Conference Tours, France, pp. 718.
Niewold TA (2013) Quality Assurance and Safety of Crops and Foods, in press Tracey KJ (2002) Nature 420: 853-859.
Vieira SL, Oyarzabal OA, Freitas DM, Berres J, Pena JEM, Torres CA & Coneglian JLB (2008) Journal of Applied Poultry Research 17: 28-133.
Wang Q, Li H, Liu S, Wang G & Wang Y (2005) Animal Biotechnology 16: 191-201.
Wells JM, Rossi O, Meijerink M & van Baarlen P (2011) Proceedings of the National Acadamy of Science USA 108(Suppl 1): 4607-4614.
Wicher KB & Fries E (2006) Proceedings of the National Academy of Sciences USA 103: 4168-4173.
Wu CH, Chen TL, Chen TG, Ho WP, Chiu WT& Chen RM (2003) Journal of Trauma 55: 540-545.
Wu G, Flynn NE, Yan W & Barstow DG (1995) Biochemical Journal 306: 717-721.










.jpg&w=3840&q=75)