Eimeria tenella Elongation Factor-1a (EF-1a) Coadministered with Chicken IL-7 (chIL-7) DNA Vaccine Emulsified in Montanide Gel 01 Adjuvant Enhanced the Immune Response to E. acervulina Infection in Broiler Chickens
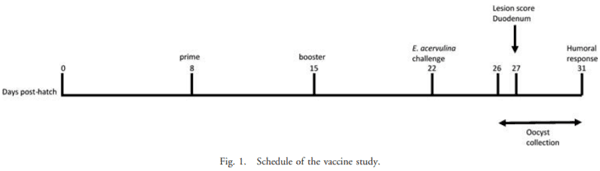
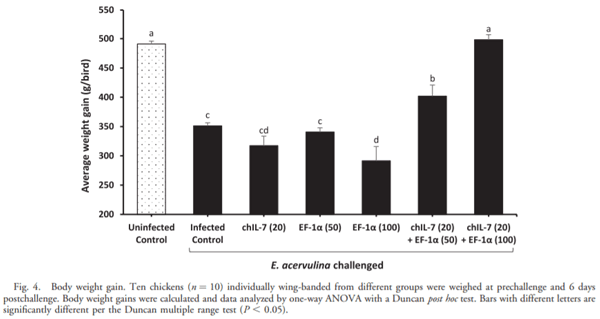
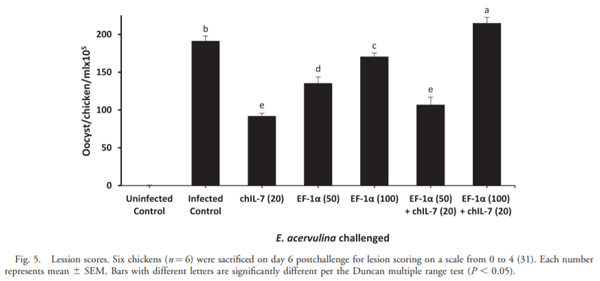
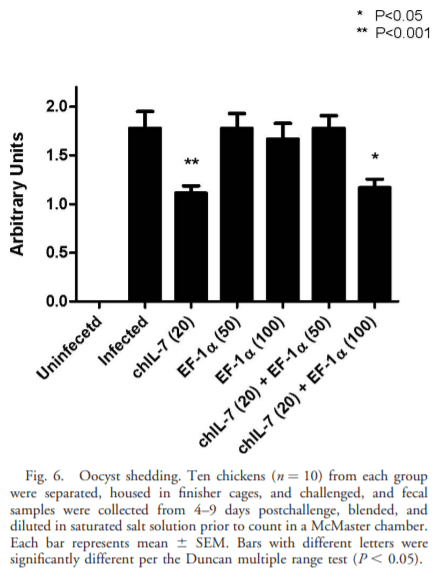
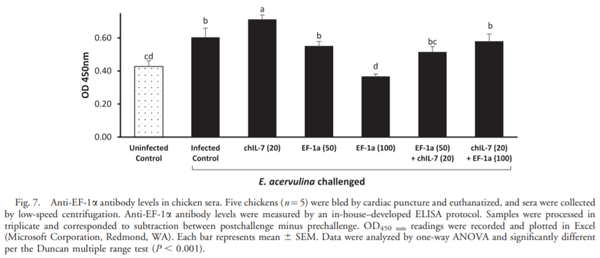
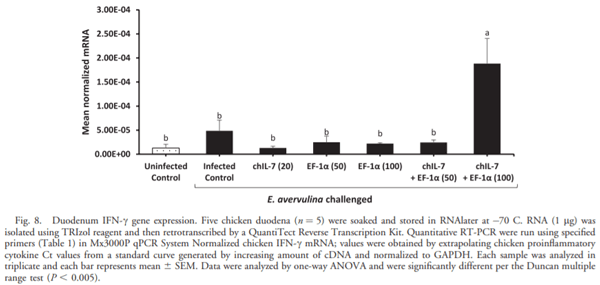
The current study was undertaken to assess the vaccine efficacy of Eimeria tenella EF-1a/chicken IL-7 (chIL-7) DNA vaccine when administered with Montanide Gel 01 adjuvant against live Eimeria acervulina challenge in commercial broiler chickens. The criteria used for the evaluation of vaccine efficacy were weight gain, duodenal lesion scores, oocyst counts, humoral antibody response, and duodenal proinflammatory cytokine gene expression. Chickens vaccinated with EF-1a (100 lg)/chIL-7 (20 lg) in Gel 01 PR adjuvant showed body weight gain similar to the uninfected control and higher oocyst shedding, a lower gut lesion score, and higher proinflammatory cytokine gene expression than did the infected controls. Moreover, chickens vaccinated with chIL-7 (20 lg) in Gel 01 PR adjuvant shed fewer oocysts with reduced gut lesion scores and produced higher levels of anti-EF-1a serum antibody than did the infected control. Chickens vaccinated with EF-1a (50 lg)/chIL-7 (20 lg) in Gel 01 PR adjuvant showed higher weight gains than did the infected control and shed significantly fewer oocysts than the infected control. Furthermore, chickens vaccinated with EF-1a (100 lg) in Gel 01 PR adjuvant demonstrated the lowest anti-EF-1a serum antibody levels. This study demonstrated the beneficial effects of using EF-1a and/or host cytokine chIL-7 DNA vaccine together with Gel 01 PR adjuvant to improve T-cell-mediated effector function in broiler chickens challenged with live E. acervulina.

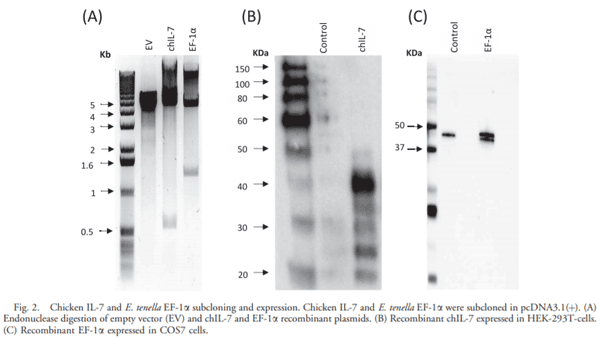
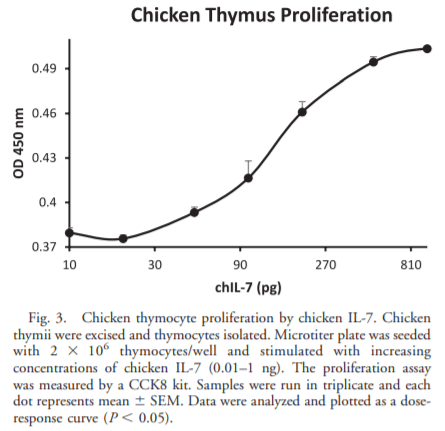
- Armitage R. J., Namen A. E., Sassenfeld H. M., Grabstein K. H.. Regulation of human T-cell proliferation by IL-7. J. Immunol. 144:938–1990.
- Atayde V. D., Aslan H., Townsend S., Hassani K., Kamhawi S., Olivier M. Exosome secretion by the parasitic protozoan Leishmania within the sand fly midgut. Cell Rep. 13:957–967. 2015.
- Bela´kova´ J., Horynova´ M., Krupka M., Weigl E., Raska M. DNA vaccines: are they still just a powerful tool for the future? Arch. Immunol. Ther. Exp. (Warsz) 55:387–398. 2007.
- Bhadra R., Guan H., Khan I. A.. Absence of both IL-7 and IL-15 severely impairs the development of CD8 T-cell response against Toxoplasma gondii. PLoS One 26:e10842. 2010.
- Bivas-Benita M., Ottenhoff T. H., Junginger H. E., Borchard G. Pulmonary DNA vaccination: concepts, possibilities and perspectives. J. Control. Release 107:1–29. 2005.
- Bradley L. M., Haynes L., Swain S. L.. IL-7: maintaining T-cell memory and achieving homeostasis. Trends Immunol. 26:172–176. 2005.
- Chapman H. D., Barta J. R., Blake D., Gruber A., Jenkins M., Smith N. C., Suo X., Tomley F. M. A selective review of advances in coccidiosis research. Adv. Parasitol. 83:93–171. 2013.
- Chapman H. D., Roberts B., Shirley M. W., Williams R. B. Guidelines for evaluating the efficacy and safety of live anticoccidial vaccines and obtaining approval for their use in chickens and turkeys. Avian Pathol. 34:279–290. 2005.
- Chen J., Li Z. Y., Petersen E., Liu W. G., Zhu X. Q.. Co-administration of interleukins 7 and 15 with DNA vaccine improves protective immunity against Toxoplasma gondii. Exp. Parasitol. 162:18–23. 2016.
- Cherif M. S., Shuaibu M. N., Kurosaki T., Helegbe G. K., Kikuchi M., Yanagi T., Tsuboi T., Sasaki H., Hirayama K. Immunogenicity of novel nanoparticle-coated MSP-1 C-terminus malaria DNA vaccine using different routes of administration. Vaccine 29:9038–9050. 2011.
- Condeelis, J. Elongation factor 1-a, translation and the cytoskel-eton. Trends Biochem. Sci. 20:169–170. 1995.
- Costa R. M., Nogueira F., de Sousa K. P., Vitorino R., Silva M. S. Immunoproteomic analysis of Plasmodium falciparum antigens using sera from patients with clinical history of imported malaria. Malar. J. 12:100. 2013.
- Cui D., Zhang J., Zuo Y., Huo S., Zhang Y., Wang L., Li X., Zhong F. Recombinant chicken interleukin-7 as a potent adjuvant increases the immunogenicity and protection of inactivated infectious bursal disease vaccine. Vet. Res. 49:10. 2018.
- Dalloul R. A., Lillehoj H. S.. Poultry coccidiosis: recent advancements in control measures and vaccine development. Expert Rev. Vaccines 5:143–163. 2006.
- Del Cacho E., Gallego M., Lee S. H., Lillehoj H. S., Quilez J., Lillehoj E. P., Sa´nchez-Acedo C. Induction of protective immunity against Eimeria tenella infection using antigen-loaded dendritic cells (DC) and DC-derived exosomes. Vaccine 29:3818–3825. 2011.
- Del Cacho E., Gallego M., Lee S. H., Lillehoj H. S., Quilez J., Lillehoj E. P., Sa´nchez-Acedo C. Induction of protective immunity against Eimeria tenella, Eimeria maxima, and Eimeria acervulina infections using dendritic cell-derived exosomes. Infect. Immun. 80:1909–1916. 2012.
- Del Cacho E., Gallego M., Lillehoj H. S., Quilez J., Lillehoj E. P., Sa´nchez-Acedo C. Induction of protective immunity against experimental Eimeria tenella infection using serum exosomes. Vet. Parasitol. 224:1–6. 2016.
- Derventzi A., Rattan S. I., Clark B. F. Phorbol ester PMA stimulates protein synthesis and increases the levels of active elongation factors EF-1 alpha and EF-2 in ageing human fibroblasts. Mech. Ageing Dev. 69:193–1993.
- Deville S., Ben Arous J., Ionkoff G., Bertrand F., Kukushkin S., Baybikov T., Borisov V., Dupuis L. Load reduction in live PRRS vaccines using oil and polymer adjuvants. Procedia in Vaccinology 6:134–140. 2012.
- Dharmawardhane S., Demma M., Yang F., Condeelis J. Compart-mentalization and actin binding properties of ABP-50: the elongation factor-1 alpha of Dictyostelium. Cell Motil. Cytoskeleton 20:279–288. 1991.
- Di Giacomo S., Quattrocchi V., Zamorano P. Use of adjuvants to enhance the immune response induced by a DNA vaccine against bovine herpesvirus-1. Viral Immunol. 28:343–346. 2015.
- Ding X., Lillehoj H. S., Quiroz M. A., Bevensee E., Lillehoj E. P. Protective immunity against Eimeria acervulina following in ovo immuni-zation with a recombinant subunit vaccine and cytokine genes. Infect. Immun. 72:6939–6944. 2004.
- Gadde U., Rathinam T., Lillehoj H. S. Passive immunization with hyperimmune egg-yolk IgY as prophylaxis and therapy for poultry diseases—a review. Anim. Health Res. Rev. 16:163–176. 2015.
- Gazzinelli R. T., Denkers E. Y. Protozoan encounters with toll-like receptor signaling pathways: implications for host parasitism. Nat. Rev. Immunol. 6:895–906. 2006.
- Gurunathan S., Stobie L., Prussin C., Sacks D. L., Glaichenhaus N., Iwasaki A., Fowell D. J., Locksley R. M., Chang J. T., Wu C. Y., Seder R. A. Requirements for the maintenance of Th1 immunity in vivo following DNA vaccination: a potential immunoregulatory role for CD8þ T-cells. J. Immunol. 165:915–924. 2000.
- Hoft D. F., Eickhoff C. S., Giddings O. K., Vasconcelos J. R., Rodrigues M. M. Trans-sialidase recombinant protein mixed with CpG motif-containing oligodeoxynucleotide induces protective mucosal and systemic Trypanosoma cruzi immunity involving CD8þ CTL and B cell-mediated cross-priming. J. Immunol. 179:6889–6900. 2007.
- Hong Y. H., Lillehoj H. S., Lee S. H., Dalloul R. A., Lillehoj E. P.. Analysis of chicken cytokine and chemokine gene expression following Eimeria acervulina and Eimeria tenella infections. Vet. Immunol. Immuno-pathol. 114:209–223. 2006.
- Huo S., Wang L., Zhang Y., Zhang J., Zuo Y., Xu J., Cui D., Li X., Zhong F. Molecular cloning of chicken IL-7 and characterization of its antiviral activity against IBDV in vivo. Poult. Sci. 95:2647–2654. 2016.
- Huo S., Zuo Y., Li N., Li X., Zhang Y., Wang L., Liu H., Zhang J., Cui D., He P., Xu J., Li Y., Zhu X., Zhong F. Chicken IL-7 as a potent adjuvant enhances IBDV VP2 DNA vaccine immunogenicity and protective efficacy. Vet. Microbiol. 193:145–155. 2016.
- Jang S. I., Lillehoj H. S., Lee S. H., Lee K. W., Lillehoj E. P., Bertrand F., Dupuis L., Deville S. Montanidee IMS 1313 N VG PR nanoparticle adjuvant enhances antigen-specific immune responses to profilin following mucosal vaccination against Eimeria acervulina. Vet. Parasitol. 182:163–170. 2011.
- Jang S. I., Lillehoj H. S., Lee S. H., Lee K. W., Lillehoj E. P., Bertrand F., Dupuis L., Deville S. Mucosal immunity against Eimeria acervulina infection in broiler chickens following oral immunization with profilin in Montanidee adjuvants. Exp. Parasitol. 129:36–41. 2011.
- Johnson J., Reid W. M. Anticoccidial drugs: lesion scoring techniques in battery and floor-pen experiments with chickens. Exp. Parasitol. 28:30–36. 1970.
- Kasper L. H., Matsuura T., Fonseka S., Arruda J., Channon J. Y., Khan I. A.. Induction of cd T-cells during acute murine infection with Toxoplasma gondii. J. Immunol. 157:5521–5527. 1996.
- Kasper L. H., Matsuura T., Khan I. A. IL-7 stimulates protective immunity in mice against the intracellular pathogen, Toxoplasma gondii. J. Immunol. 155:4798–4804. 1995.
- Khan I. A., Matsuura T., Kasper L. H. Interleukin-12 enhances murine survival against acute toxoplasmosis. Infect. Immun. 62:1639–1642. 1994.
- Laky K., Lefran¸cois L., Lingenheld E. G., Ishikawa H., Lewis J. M., Olson S., Suzuki K., Tigelaar R. E., Puddington L. Enterocytes expression of interleukin 7 induces development of cd T-cells in Peyer’s patches. J. Exp. Med. 191:1569–1580. 2000.
- Li X., Galliher-Beckley A., Nietfeld J. C., Faaberg K. S., Shi J. Montanide Gel 01 ST adjuvant enhances PRRS modified live vaccine efficacy by regulating porcine humoral and cellular immune response. World J. Vaccines 3:1–9. 2013.
- Lillehoj H. S.. Role of T lymphocytes and cytokines in coccidiosis. Int. J. Parasitol. 28:1071–1081. 1998.
- Lillehoj H. S., Min W., Dalloul R. A. Recent progress on the cytokine regulation of intestinal immune responses to Eimeria sp. Poult. Sci. 83:611–623. 2004.
- Lin R. Q., Lillehoj H. S., Lee S. K., Oh S., Panebra A., Lillehoj E. P. Vaccination with Eimeria tenella elongation factor-1a recombinant protein induces protective immunity against E. tenella and E. maxima infections. Vet. Parasitol. 243:79–84. 2017.
- Matsubayashi M., Minoura C., Kimura S., Tani H., Furuya M., Lillehoj H. S., Matsuda H., Takenaka S., Hatta T., Tsuji N., Sasai K. Identification of Eimeria acervulina conoid antigen using chicken mono-clonal antibody. Parasitol. Res. 115:4123–4128. 2016.
- Matsubayashi M., Teramoto-Kimata I., Uni S, Lillehoj H. S., Matsuda H., Furuya M., Tani H., Sasai K. Elongation factor-1a is a novel protein associated with host cell invasion and a potential protective antigen of Cryptosporidium parvum. J. Biol. Chem. 288:34111–34120. 2013.
- McDonald V., Shirley M. W. Past and future: vaccination against Eimeria. Parasitology 136:1477–1489. 2009.
- Nandan D., Reiner N. E. Leishmania donovani engages in regulatory interference by targeting macrophage protein tyrosine phospha-tase SHP-1. Clin. Immunol. 114:266–277. 2005.
- Park J. H., Adoro S., Lucas P. J., Sarafova S. D., Alag A. S., Doan L. L., Erman B., Liu X., Ellmeier W., Bosselut R., Feigenbaum L., Singer A. ‘Coreceptor tuning’: cytokine signals transcriptionally tailor CD8 coreceptor expression to the self-specificity of the TCR. Nat. Immunol. 8:1049–1059. 2007.
- Park J. H., Yu Q., Erman B., Appelbaum J. S., Montoya-Durango D., Grimes H. L., Singer A. Suppression of IL7Ralpha transcription by IL-7 and other pro-survival cytokines: a novel mechanism for maximizing IL-7-dependent T-cell survival. Immunity 21:289–302. 2004.
- Piedra-Quintero Z. L., Apodaca-Medina A. I., Beltra´n-Lopez´ E., Leon´-Sicairos C. R., Cha´vez-Ontiveros J., Rendon´-Maldonado J. G., Ortiz-Navarrete V. F., Lopez´-Moreno H. S. Immunoproteomic identification of p29 antigen as the elongation factor-1a of Leishmania mexicana. Vector Borne Zoonotic Dis. 15:449–452. 2015.
- Shirley M. W., Blake D., White S. E., Sheriff R., Smith A. L.. Integrating genetics and genomics to identify new leads for the control of Eimeria sp. Parasitology 128(Suppl. 1):S33–S42. 2004.
- Shirley M. W., Lillehoj H. S. The long view: a selective review of 40 years of coccidiosis research. Avian Pathol. 41:111–121. 2012.
- Shirley M. W., Smith A. L., Tomley F. M. The biology of avian Eimeria with an emphasis on their control by vaccination. Adv. Parasitol. 60:285–330. 2005.
- Sieling P. A., Sakimura L., Uyemura K., Yamamura M., Oliveros J., Nickoloff B. J., Rea T. H., Modlin R. L. IL-7 in the cell-mediated immune response to a human pathogen. J. Immunol. 154:2775–2783. 1995.
- Siles-Lucas M., Sa´nchez-Ovejero C., Gonza´lez-Sa´nchez M., Gon-za´lez E., Falcon´-Perez´ J. M., Boufana B., Fratini F., Casulli A., Manzano-Roma´n R. Isolation and characterization of exosomes derived from fertile sheep hydatid cysts. Vet. Parasitol. 236:22–33. 2017.
- Silverman J. M., Reiner N. E. Leishmania exosomes deliver preemptive strikes to create an environment permissive for early infection. Front. Cell. Infect. Microbiol. 1:26. 2012.
- Singh V., Gowthaman U., Jain S., Parihar P., Banskar S., Gupta P., Gupta U. D., Agrewala J. N. Co-administration of interleukins 7 and 15 with Bacilli Calmette-Guerin´ mounts enduring T-cell memory response against Mycobacterium tuberculosis. J. Infect. Dis. 202:480–489. 2010.
- Skarin H., Ringqvist E., Hellman U., Svard¨ S. G. Elongation factor 1-a is released into the culture medium during growth of Giardia intestinalis trophozoites. Exp. Parasitol. 127:804–810. 2011.
- Slobin L. I. The role of eukaryotic factor Tu in protein synthesis. The measurement of the elongation factor Tu content of rabbit reticulocytes and other mammalian cells by a sensitive radioimmunoassay. Eur. J. Biochem. 110:555–563. 1980.
- Tomley F. M. Characterization of rhoptry proteins of Eimeria tenella sporozoites: antigenic diversity of rhoptry epitopes within species of the genus Eimeria and among three asexual generations of a single species, E. tenella. Infect. Immun. 62:4656–4658. 1994.
- Tomley F. M., Bumstead J. M., Billington K. J., Dunn P. P. Molecular cloning and characterization of a novel acidic microneme protein (Etmic-2) from the apicomplexan protozoan parasite, Eimeria tenella. Mol. Biochem. Parasitol. 79:195–206. 1996.
- Tomley F. M., Clarke L. E., Kawazoe U., Dijkema R., Kok J. J. Sequence of the gene encoding an immunodominant microneme protein of Eimeria tenella. Mol. Biochem. Parasitol. 49:277–288. 1991.
- Vermeulen A. N., Kok J. J., van den Boogaart P., Dijkema R., Claessens J. A. Eimeria refractile body proteins contain two potentially functional characteristics: transhydrogenase and carbohydrate transport. FEMS Microbiol. Lett. 110:223–229. 1993.
- Vialle R., Dupuis L., Deville S., Bertrand F., Gaucheron J., Aucouturier J. Microgel particulate adjuvant: characterization and mecha-nism of action. Procedia in Vaccinology 2:12–16. 2010.
- Wallach M. The importance of transmission-blocking immunity in the control of infections by apicomplexan parasites. Int. J. Parasitol. 27:1159–1167. 1997.
- Wallach M. G., Mencher D., Yarus S., Pillemer G., Halabi A., Pugatsch T. Eimeria maxima: identification of gametocyte protein antigens. Exp. Parasitol. 68:49–56. 1989.
- Wang S., Wang Y., Sun X., Zhang Z., Liu T., Gadahi J. A., Hassan I. A., Xu L., Yan R., Song X., Li X. Protective immunity against acute toxoplasmosis in BALB/c mice induced by a DNA vaccine encoding Toxoplasma gondii elongation factor 1-a. BMC Infect. Dis. 15:448. 2015.
- Williams R. B. A compartmentalized model for the estimation of the cost of coccidiosis to the world’s chicken production industry. Int. J. Parasitol. 29:1209–1229. 1999.
- Xue Q., Zhao Z., Liu H., Chen K., Xue Y., Wang L. First comparison of adjuvant for trivalent inactivated Haemophilus parasuis serovars 4, 5 and 12 vaccines against Glasser’s¨ disease. Vet. Immunol. Immunopathol. 168:153–158. 2015.
- Yang F., Demma M., Warren V., Dharmawardhane S., Condeelis J. Identification of an actin-binding protein from Dictyostelium as elongation factor 1a. Nature 347:494–496. 1990.
- Yu X., Harris S. L., Levine A. J. The regulation of exosome secretion: a novel function of the p53 protein. Cancer Res. 66:4795–4801. 2006.
- Zhao Z., Liu H., Zhang K., Xue Q., Chen K., Xue Y. Minimum dose, antigen content, and immunization duration of a trivalent vaccine of inactivated Haemophilus parasuis serovars 4, 5 and 12 against Glasser¨ disease in pigs. Can. J. Vet. Res. 80:287–293. 2016.









.jpg&w=3840&q=75)