I. INTRODUCTION
In order to improve production, nutritionists endeavor to increase digestibility, availability of nutrients in the diet and to obtain more product consumed per unit feed, and therefore they consider total nutrient intake and retention as well as waste output. Although poultry is highly efficient in converting feed to egg or body gain, the presence of components such as antinutritional factors and indigestible nutrients in the feed may reduce production efficiency. These components in the feeds reduce nutrient digestibility and availability by either chemically or physically binding nutrients or by direct toxic action the animal (Nelson, 1971; Adeola and Cowieson, 2011). Availability of phosphorus (P) for monogastric animals in feed ingredients of plant origin is much lower than the P content of those ingredients, because of the greater portion of their total P is present as phytate. Phytate or phytic acid is both an antinutritional factor and an indigestible nutrient (Adeola and Cowieson, 2011). The P contained in the molecule is very low availability to monogastric animals, especially poultry. Because of its chelating ability with essential mineral elements such as zinc, manganese, iron, calcium, potassium, and proteins, phytate reduces the availability of these nutrients through the formation of indigestible complexes (Nelson, 1967).
Recently advances in enzyme technology have led to the development enzyme products to be used as feed additives. Many fungi, bacteria, and yeasts naturally produce the enzyme phytase and some of them already on the market. Nelson et al. (1971) found that when the preparation of phytase made from Aspergillusficuum was added to acorn-soybean diet containing low P, phytate phosphorus was almost completely hydrolyzed by the chickens. Recently, heightened environmental concerns over phosphates in animal waste have renewed the interest phytase (Sharpley et al., 2004; Toprak and Yilmaz, 2016) and many research with broiler(Yan et al., 2003) and quail (Sacakli et al., 2006; Saima et al., 2014) were conducted demonstrating enhancements of P availability by phytase addition to diets. Newkirk and Classen (1992) suggested that when phytase and xylanase enzymes were added to diets containing different levels of total P (0.4, 0.5, 0.6, 0.7%) in the laying hens, phytase reduced the feed efficiency and interacted with xylanase to influence egg production. Konca and Bahtiyarca (1996), Bahtiyarca and Parlat (1997), Sacakli et al. (2006) showed that when the diets have low P were supplemented with phytase, body weight gain, feed conversion ratio was increased and also bone ash and strength were comparable with the control diet has adequate P. On the other hand, with addition of phytase to poultry diets sometimes revealed controversy results (Adeola and Cowieson, 2011). Therefore the objective of the present study was to determine the effects of different levels of phytase addition to a diet containing low P on the performance, shell quality and bone characteristics in laying Japanese quail.
II. MATERIALS AND METHODS
In the experiment, a total of 162 7-week-old female Japanese quail were used. They were selected from 300 female quail on the basis of body weight. They fed a starter diet containing 23% crude protein, 12.5 MJ ME/kg, 0.80% Ca, 0.45% available P, 1.3% lysine, 0.50% methionine and 0.75% methionine+ cysteine and fed test diets until 7 weeks of age. The experimental diets were prepared based on corn-soybean meal containing adequate level (control, 0.33%) and low level (basal, 0.15%) of available P and the basal diet and supplemented with 0; 500; 1000; 1500 and 2000 units/kg of the phytase (total six groups). Diets compositions are presented in Table 1.
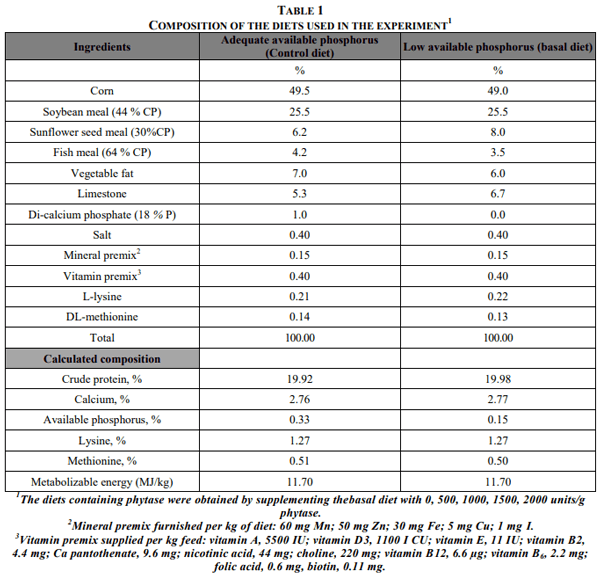
Phytase enzyme (Finase™, produced A. niger, Alko Biotechnology, Ltd., Rajamâki, Finland). Phytase preparation has 500.000 phytase units/g (a phytase unit is defined as the amount that liberates 1 nanomole of phosphorus from sodium phytate in one minute under standard conditions). It is recommended that the phytase enzyme should be added at a level of 250.000-1.000.000 phytase units/kg feed (Anonymous, 1993 a, b). The research was carried out for five periods and each period 28 days (total 140 days) in randomized plot designs with three replicates of nine female quail each. (6 groupx9 quailx3 replicate=total 162 quail). The quail were housed in groups of nine birds in laying wire battery cages that were manufactured in Konya. Feed in mash form and water supplied ad libitum and continual lighting was provided during growing and laying periods.
Body weights of female quail were determined in groups of nine quail at the start and end of the experiment. Egg numbers and given feed were recorded daily and average egg production and feed consumption for every 28 days periods were calculated from these data. Egg weight was determined for all eggs collected at the last three days of every 28 days periods. Egg mass was found to be multiplying daily egg number by average egg weight for every period and feed conversion ratio was found by dividing of average daily feed consumption by egg mass. Shell parameters were measured for eggs collected from quail at 70, 71, 72thdays (1st period) and at the last three days (2iid period) of the experiment. The surface area of the egg was calculated according to the equation of Carter (1975). The breaking strength of the eggs were determined with Test Instrument of Biological Materials using the procedure described by Ögüt and Aydin (1992). After the eggs were broken, their contents were removed and shell washed with tap water. Five shells from each replicate were selected at random. Two pieces of shell were taken from the equator of the egg and shell membrane was removed and thickness was measured using a micrometer. After the pieces of shell were combined with main shell, they were dried in oven for 24 hours at 105, and air-cooled and weighed. The weight of the shell with membrane was expressed as a percentage of whole egg weight and to calculate shell weight per unit surface area, the weight of shell plus membrane was divided by surface area.
To determine carcass weight all quail in group were sacrificed, defeathered, eviscerated (except heart and lungs) at termination of the experiment and were subsequently weighed per group all quail. Right tibiae of birds were cut, kept in a refrigerator for 24 hours and then cleaned from soft tissues to determine of tibia weight and tibia ash. Tibiae were dried in oven for 24 hours at 105 °C. Fat-free dried tibia weight and percentage tibia ash were determined according to the method of Said and Sullivan (1985). Manure was collected during last five days of the experiment and combined and dried in oven for 24 hours at 105 °C, and air-cooled. Dried manure was a shed in a muffle furnace for 6 hours at 550 °C and percentage of manure ash was calculated.
Statistical analysis was carried out using the statistical software SPSS. Data for all measurement were subjected to ANOVA procedure appropriate for the completely randomized design. Linear, quadratic and cubic effects were observed, however, in the tables only linear effects are given due to similar probability (P) values. Duncan's multiple range tests was used to separate differences between the means. All percentage values were transformed according to the equation below before analysis of variance was performed (Winer, 1971):
Transformed value = 2 x arcsin√(%/100)
However, the percentage values are shown in the tables and figures for easy interpretation. Probability significance levels were accepted as P<0.05.
III. RESULTS
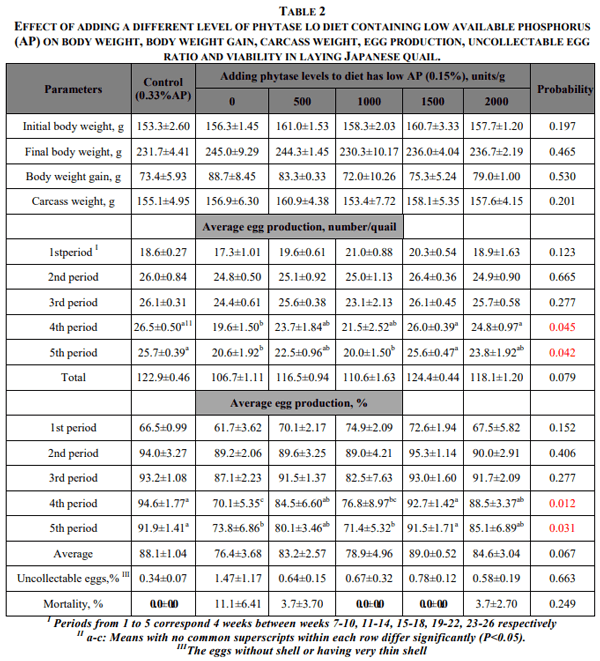
Dietary treatments did not significant effect on final body weight, body weight gain, and carcass weight of quail. Although for the first three periods of the experiment, there were no significant responses to dietary treatments with regard to egg number or percentage of egg production, egg number of the group fed low AP was significantly lower in period 4 and, egg numbers of the groups fed low AP+0 and 1000 units/g phytase were significantly lower (P<0.005) than that of control and 1500 units/g phytase group in period 5. In addition, the percentage egg production of the same groups were significantly lower (P<0.05) than that of the control and 1500 units/g phytase groups in periods 4 and 5 (Table 2). Egg number and egg yield of quail were generally increased; when the diet has low AP was supplemented with different levels of phytase. But an increase in egg production was not linear with increasing dietary phytase, especially egg production of quail receiving low AP+1500 units phytase/g was similar to those of control group, even slightly higher than the control group (Table 2). Dietary phosphorus and different phytase levels had no significant effect on percentage uncollectable egg (very thin shell or no shell) and mortality. However, mortality rate of the quail fed diet containing low AP was relatively higher when compared to control group.
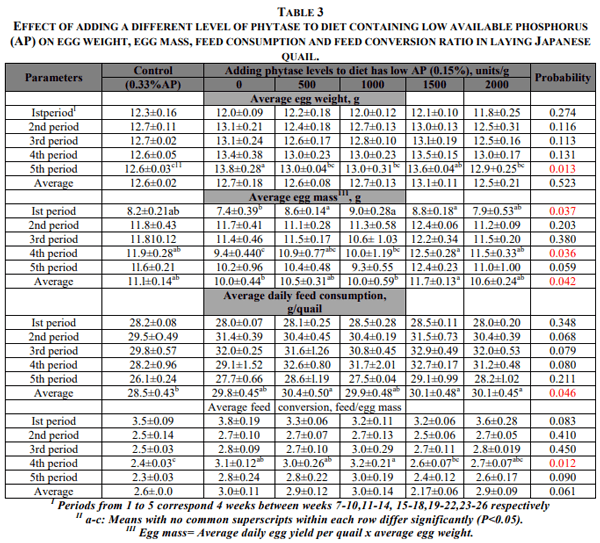
The quail fed the diets containing low AP+0 and 1500 phytase levels had egg weight in period 5 significantly higher than that of C group. This effect was not seen in other periods and on average egg weight. But there were significant differences in egg mass among the dietary treatments. The egg mass of the group fed low AP was significantly higher than the control group in period 4. When the diet has low AP was supplemented with different levels of phytase, egg mass was improved. Egg mass of the groups receiving 500, 100, 1500 units phytase in period 1 and 1500, 200 units phytase in period 4, and average egg mass of the group receiving 1500 units phytase were significantly higher (P<0.05) than the group fed low AP+0 unit phytase. Egg mass of the experiment groups were very close to those of control group (Table 3).
Dietary treatments had no significant effect on feed consumption in different periods of the experiment, but average daily feed consumption during the experiment of the quail fed low AP+500, 1500 and 2000 units phytase were significantly higher (P<0.05) than that of C group. However, feed conversion (feed/egg mass) of the quail fed low AP supplemented with phytase generally tended to slightly high. However, the groups fed low AP+0; 500 and 1500 units phytase had feed conversion significantly higher (P<0.05) than control in period 4. The group fed 1500 units phytase was very close to those of control group in feed conversion (Table 3).
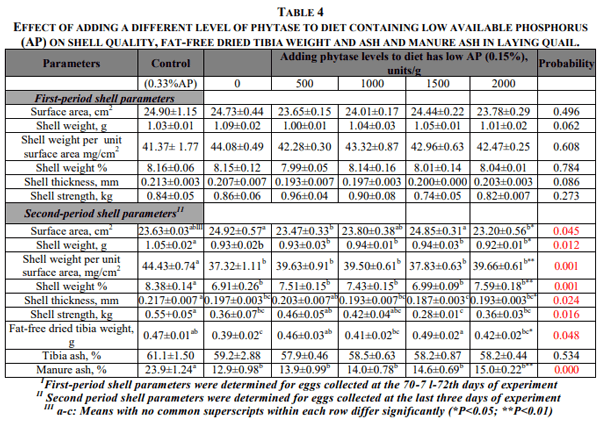
Although dietary treatments had no significant effect on the shell quality parameters determined at the first period of the experiment, they had a significant effect on the various shell quality parameters at the second period of the experiment. Eggshell surface area of the groups fed with low AP+500 and 2000 units phytase were close to those of group C, however, significantly lower than those of fed low AP+0 and 1500 units phytase. The groups fed low AP and low AP supplemented different phytase had shell weight (P<0.05) and shell weight per unit area (P<0.01) significantly lower than that of group C (Table 4). When the diet has low AP was supplemented with 500 units phytase, the shell thickness of other groups was significantly lower (P<0.05) than that of group C. In addition, while the groups receiving low AP+0, 1500 and 2000 phytase units had shell strength significantly lower (P<0.05) than that of C group. The groups receiving 500 and 1000 phytase units had shell strength comparable to those of C diet. The result of the experiment shows that the response of the laying quail as age increased was different with respect to various shell quality parameters when the diet has low AP was supplemented with phytase.
Fat-free dried tibia weight of the group fed low AP was significantly lower than that of group C (P<0.05). However, when phytase added to diet has low AP, fat-free dried tibia weight was improved and bone weights of the groups supplementing phytase were very close to that of group C. In addition, the groups fed low AP+500 and 1500 phytase units had bone weight significantly higher than those of group fed low AP. Percentage of bone ash of the groups fed low AP with and without were comparable to those of group C. Manure ash content of the groups fed diets has low AP supplemented with or without phytase was significantly lower (P<0.05) compared to group C.
IV. DISCUSSION
Results of the present experiment showed that egg production (as number and percentage), egg mass, average daily feed consumption, fat-free dried tibia weight and viability improved when added phytase level to diet has low AP. These parameters were comparable to those of control group fed diet has adequate AP. Final live weight, carcass weight, body weight, body weight gain, feed conversion ratio, uncollectable eggs ratio, some egg shell quality parameters measured in eggs collected at the end of the first period were not affected by treatments. Similar findings were observed by Sacakli et al., (2006) who reported that BW, BWG, FC, FCR, and carcass were not affected by the phytase addition (300 phytase unit) to low AP content (2 g/kg) quail diets in growing period. On the other hand, many trial resulted lower body weight, feed intake and feed efficiency by a low level AP diets and phytase addition to diets improved broiler performance (Denbow et al., 1995; Qian et al., 1996).
These findings were similar results of the meat type poultry and agree with previous reports of Simons and Versteegh (1991). They reported that laying hens fed diets with low AP supplemented with phytase had performance as high as those of fed diet with adequate AP. Results of the experiment that was conducted at Penn State University with enzymes aims to reduce fecal phosphorus levels of commercial pullets and hens, also showed that body weight, production and egg parameters equal to the control can be maintained with diet has low P added phytase (Patterson, 1996). In addition, Patterson (1996) narrated from Van der Klis et al. (1996) that egg production, egg weight and feed/egg ratio can be improved from a negative control diet (3.3 g/kg total P) with addition phytase at 200 unit/kg. Another report (van der Klis et al. 1991) showed that most portion of dietary phytate phosphorus was hydrolysed by addition phytase at 250 and 500 units/kg feed in WL-laying hens. However, Newkirk and Classen (1992) reported that in two lines of leghorn hens, egg production decreased with addition phytase to diets have different levels of total P. However in this study, with regard to egg production, egg mass, average daily feed consumption, the responses of laying quail to the increasing phytase levels were increased, but this increase was not linear.
These findings indicate that in order to improve availability of phosphorus addition phytase to diet has high phytate phosphorus is an effective application in laying quail as broiler chicks and laying hens.
As increase age, low dietary phosphorus resulted in a decrease in many of shell parameters and while adding increasing levels of phytase to diet containing low AP was improved some shell parameters, some of them were not affected. No data concerning the effect of phytase on shell quality was found in the literature.
Fat-free dried tibia bone weight of quail were significantly increased by addition phytase at 500 and 1500 units/g feed when compared with quail fed low AP and, quail fed diet with and without phytase had % tibia ash comparable to those quail fed adequate AP. These findings suggest that utilization of phosphorus was increased by addition phytase. An increase in body weight gain, feed efficiency, fat-free tibia weight, bone ash and strength due to adding phytase to diet has low AP were also seen by Konca and Bahtiyarca (1996) and Bahtiyarca and Parlat (1997) in young Japanese quail.
The performance and bone parameters of the group fed with 1500 phytase units were very close values to those of control group, even higher than the control group in some characters. However, any of phytase level did not prevent a decrease in shell weight measured at the second period and a decrease in shell thickness and strength with 1500 and 2000 unit phytase were not prevented. The difference in source and activity phytase of enzymes, species of bird and inclusion rate were marked difficult interpretation of results obtained from the different experiment. The results of the present experiment indicated that with regard to performance and bone parameters, the responses of laying quail to the diet has low AP supplemented with increasing phytase levels were improved but this increase was not linear, and the responses of quail fed with 1500 phytase units were very close to or better than those fed adequate AP, but some shell parameters were impaired. Manure P content was decreased by lower AP consumption with feed. This result is expected because it has been known that when poultry fed diet containing low AP, phosphorus level of manure was significantly decreased.
V. CONCLUSION
Low dietary AP concentration may reduce production parameters in quail. The phytase addition to quail diets may help to improve production parameters. Tibia weight may decrease with low AP diets. Manure P content may be decreased via lower AP content in diets.
This article was originally published in International Journal of Environmental & Agriculture Research (IJOEAR). Vol-3, Issue-12, December- 2017.














.jpg&w=3840&q=75)