INTRODUCTION
In studies with birds which objective is to evaluate the immune responses and in which nutrients are manipulated to enhance bird immunity, not only performance responses are used. As the immune system is very complex, and some parameters are influenced by related sub-systems, immune responses are also used. Cell-mediated immune response is one of those methodologies applied in this kind of research as an auxiliary tool.
Cutaneous basophil hipersensitivity (CBH) test consists of an intradermal injection of phytohemagglutinin-P (PHA-P) in the skin between the 3rd and 4th interdigital space of chickens, where the thymus-dependent reaction mediated by T cells is easy to analyze. This test can be performed in birds of up to two weeks of age, according Stadecker et al., (1977) but other authors (Boa-Amponsen et al., 2000; Sakamoto et al. 2006) have applied it in older birds.
Corrier & Deloach (1990) used this method to evaluate the interdigital skin space between the toes of chickens as a skin-test site for eliciting CBH responses after the administration of dexamethasone. The authors concluded that the CBH test with PHA-P is a simple and fast method to evaluate of cell-mediated immunity in chickens. Sakamoto et al. (2006) also used this method in 36-day-old chickens, whereas Boa-Amponsen et al. (2000) used 90-day-old birds.
In 1959, the mitogenic properties of PHA-P were discovered. These properties are characterized by T-cell stimulation in several animal species, such as rats, mice, guinea-pigs, and birds. In humans, this method is applied in hypersensitivity tests (“late-phase reactions”; Goto et al., 1978). PHA-P is a glycoprotein of non-immune origin present in many varieties of beans, such as Phaseoulus vulgaris (SIGMA-Aldrich®), particularly in red kidney beans.
Vitamin E is one of the most frequent studied nutrients presenting immunomodulating properties, as it is a natural antioxidant of cell membranes and improves the phagocytic activity of macrophages (Konjufca et al., 2004), among another biological actions. Research on the effect of that vitamin on the immune response of poultry have yielded controversial results. For instance, vitamin levels that have some effect on immune parameters usually do not affect others, such as skin cell response. Sakamoto et al. (2006) observed higher cell proliferation feeding broilers with 10mg/kg vitamin E as compared to 500 mg/kg. Leshchinsky & Klasing (2001) did not observe any effect of 0 to 200mg/kg of vitamin E dietary levels on the CBH test, and Boa-Amponsem et al. (2000) found lower reaction using 300mg/kg vitamin E as compared to 10mg/kg in rooster diets.
The aim of the present study was to study the effects of vitamin E levels (levels similar to those recommended under practical situations) on the cell-mediated immunity of broilers submitted to vaccine challenge, and to validate the CBH test as a method in this type of experiment.
MATERIALS AND METHODS
Birds, housing, and treatments
Two hundred and sixteen one-day-old Ross 308 male chicks were used in the experiment. Birds were housed in metal battery cages (at 0.72m2 /cage during the starter phase and 0.84 m2 /cage during the grower phase) placed in a room with temperature control in the experimental facilities of the Laboratório de Ensino Zootécnico (LEZO) of the Federal University of Rio Grande do Sul, Brazil. Birds were offered the experimental diets and water ad libitum from day one. The lighting program used was 24 hours of artificial light during the entire experimental period, which lasted 36 days.
The experiment was carried out according to a completely randomized design, with three dietary vitamin E levels and six replicates of 12 birds/replicate from day one to day 29, when this number was adjusted for 10 birds/replicate.
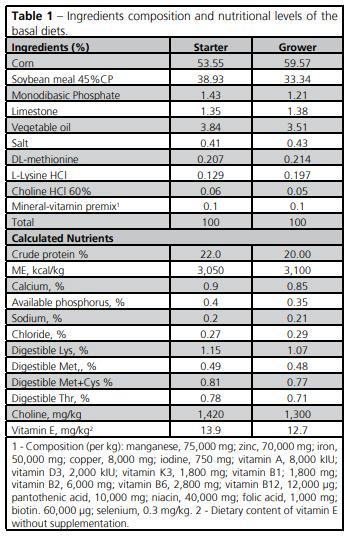
The experimental diets were manufactured from a basal diet, which was formulated using the Brazilian Tables for Poultry and Swine (Rostagno, 2005). Three vitamin E levels (30, 65 and 100mg/kg) were added to the basal diet to establish the treatments (Table 1). Vitamin E was added as 50% -tocopherol (BASF®). The experimental diets were analyzed for dry matter and crude protein content (Animal Nutrition Laboratory of UFRGS). Vitamin E content in the experimental diets was analyzed by a commercial lab (CBO Assessoria e Análises, Campinas, SP, Brazil). Dry matter, crude protein, and vitamin E content were within the expected range. Diet composition is shown in Table 1.
Immune stimulation
At three days of age, chicks were vaccinated against coccidiosis (Livacox Q®, Merial) at a dose of 10mL/4L of water distributed in 110 mL in each drinker placed in each cage. The floor of the cages was lined with paper for 16 days after vaccination to allow re-infestation of birds with the excreted oocysts.
At 14 and 30 days of age, birds were vaccinated by eyedrop against Newcastle Disease (NCD) (New Vac LS®, Fort Dodge). When birds were 35 days old, phytohemagglutinin (PHA-P) was applied at dose of 100µg/bird in three birds/replicate. The cell reaction caused by the PHA-P injection was evaluated as cutaneous basophil hipersensitivity (CBH) according to the methodology described by Corrier & DeLoach (1990). PHA-P was purchased from SIGMA Aldrich® (PHA-P code number L8754 – lectin from Phaseolus vulgaris), and 15mg of the lyophilized powder were diluted in 15mL of phosphate buffered saline solution (PBS) in order to obtain a dose of 100µg/0.1mL per bird. The inoculation was made in the interdigital space between the third and fourth toes of right foot of three birds/replicate, by intradermal injection. In the same interdigital space of the left foot (in the same bird), 0.1mL PBS was injected as control.
Measurements
The thickness of interdigital spaces were measured before the injection and six, 12 and 24 hours afterwards using a digital caliper (Electronic Digital Caliper CE, with 0.01mm precision). The results were used to calculate the following:
(1) response = post-PHA-P injection thickness of the right foot – pre-PHA-P injection thickness of the right foot (mm)
(2) PBS control response = post-PBS injection thickness of the right foot – pre-PBS injection thickness of the left foot (mm)
Therefore, cell reaction at each evaluation time was calculated as:
CBH = (1) – (2)
Statistical analysis
Experimental data normality was verified, and then data were submitted to analysis of variance, using SAS (2000) software package. Means were compared using least-square means test.
RESULTS AND DISCUSSION
Figure 1 shows that the vitamin E dietary level of 65mg/kg provided the longer lasting cell-mediated immune response (p≤0.08), as 24h post-PHA-P injection, the skin in the interdigital space of birds fed that vitamin level was still very thick.
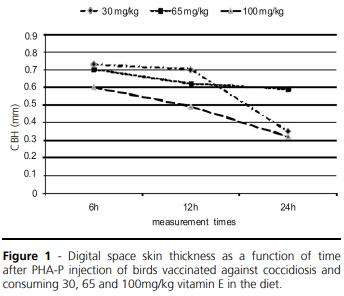
The measured response is called cell-mediated response, which function is to eliminate intracellular antigens, which requires the release of cytokines by T “helper” lymphocytes or Th1 (Erf. 2004). This response, along with humoral immunity and innate immunity, comprises the sub-systems of immune system. Opposite to the innate system, which is nonspecific and has several negative effects on the body, such as fever and anorexia (Humphrey & Klasing, 2004), the cell-mediated immune response is able of recognizing a large number of intracellular antigens through of T-cell receptors and to eliminate them. Cytokines are also secreted during cell-mediated immune response: cell surface molecules are expressed, sending out activation signals to the other cells of the innate and adaptive systems (Erf. 2004).
Cell-mediated immune responses usually do not affect broiler performance, as observed by Konjufca et al. (2004) and Sakamoto et al. (2006). On the other hand, Koustsos et al. (2007) suggest that, due to the variability of results obtained in CBH tests, skin thickness increase at the site of PHA-P injection could also reveal innate immune reaction processes and not only cell-mediated reactions. Those authors have made biopsies and cell identification of lesions caused by PHA-P, and observed that the increase of skin thickness at the site of injection can be caused by a high influx of macrophages and heterophils, and not only of basophils and lymphocytes. This suggests that there are innate immune reactions or interactions between various types of leucocytes and not merely a single cell-mediated reaction. Corrier & DeLoach (1990) also reported diffuse infiltration of neutrophils and basophils, as well as of lymphocytes and monocytes in their histopathological study in similar lesions. They also observed maximum cellular reaction 12h post-PHA-P injection, and that this reaction remained constant or slightly declined 24h post-injection.
Koustsos et al. (2007) studied the influence of carotenoids, especially of lutein, on the cutaneous basophil responses in chickens and found local cell reactions up to 48h post-PHA-P injection. The authors believe that because lutein can activate lymphocyte multiplication and is also able to reduce the production of free radicals, it may interfere in the local reaction to PHA-P. In the present experiment, the cell types present in the local lesions were not studied, but as vitamin E acts as antioxidant similarly to lutein, the dietary vitamin E level of 65 mg/kg could contribute for the longer lasting cell reactions found.
When testing vitamin E levels associated or not to glutamine in broiler diets, Sakamoto et al. (2006) observed higher cell proliferation 12h post-PHA-P injection in birds consuming 10mg/kg than in those consuming 500mg/kg. On the other hand, Leshchinsky & Klasing (2001), using 0 to 200mg/kg vitamin E in broiler diets, did not observe any effects on CBH. BoaAmponsem et al. (2000) obtained lower CBH with 300mg/kg vitamin E than with 10mg/kg in rooster diets. The authors emphasized that the CBH method can lead to variable responses because of the difficulty to do a perfect application (intradermal injection) of the PHA-P effective dose. This could explain the high coefficients of variation found for this response.
CONCLUSIONS
The results obtained for the group of birds consuming 65mg/kg of vitamin E showed that this level is effective for the enhancement of the immune response as evaluated by the CBH test in birds challenged with the vaccine against coccidiosis. Under practical conditions, this level of vitamin E can be used by poultry nutritionists. Despite the variability of responses, the CBH test is useful in studies on nutrition related to immunity.
This article was originally published in Revista Brasileira de Ciencia Avícola. vol.13 no.1 Campinas Jan./Mar. 2011. http://dx.doi.org/10.1590/S1516-635X2011000100008. This is an Open Access article licensed under a Creative Commons Attribution License.










.jpg&w=3840&q=75)