INTRODUCTION
Genetic selection for higher growth rate, and providing balanced diet and superior health care measures led to a higher body weight in commercial broilers. The birds that are being reared in open-sided poultry houses may lead to stress due to constant dynamic variations in temperature and humidity in the environment, which causes adverse effects on performance (Niu et al., 2009), meat quality and immune responses (Thompson and Scott, 1970). Further, heat causes a major stressor for chicken due to their high metabolic rate, and body temperature and lack of sweat glands (Sahin and Kucuk, 2003). High environmental temperature in the tropical countries causes heavy financial losses to poultry due to reduced feed intake and decreasing feed conversion efficiency (Panda et al., 2008). However, theses adversities in the tropics can be minimized by feeding diet supplemented with organic selenium (Se).
Se is a trace mineral that is essential to keep good health for animals and humans. Deficiency of Se results in exudative diathesis and pancreatic degeneration (Toghyani et al., 2008), and it plays an important role in immune function and production of immunoglobulin (Spallholz et al., 1973). Supplementation of Se in chicken diet increases antibody titers to Newcastle disease (ND) (Hegazy and Adachi, 2000). Further, Se influences immune responses through its incorporation into selenoproteins such as the amino acid selenocysteine (Hoffmann, 2007).
The biological systems are continuously influenced to free radicals and reactive oxygen species (ROS), which are produced as a part of the normal cell metabolism (Tappel and Tappel, 2004). Furthermore, in a several physiological and pathological states, excess amounts of ROS are generated (Fridivich, 1978), which damages the cell phospholipid membranes and other macromolecules (Wiseman and Halliwell, 1996). Utilization of appropriate antioxidants will help the biological system by scavenging reactive oxygen, which intern reduces lipid peroxidation (LP) and increase activity of antioxidant defense system (Nunes et al., 2005).
The ideal Se concentration in the diet is essential for the normal development and functioning of the immune system (Koski and Marilyn, 2003) and for maintaining the systemic defense in broilers reared in tropics. However, information about the beneficial effects of supplementation of Se in maize and soybean meal-based diets on performance of broiler reared in tropics is scanty. Therefore, an experiment was conducted to study the effects of supplementing Se on performance, slaughter parameters, stress indices, and immune responses in commercial broilers.
MATERIALS AND METHODS
Experimental birds, management, and diets
Day-old commercial female broiler chicks (n = 240) were distributed randomly into eight replicates of five treatments with six chicks in each replicate (pen). On day one, chicks were wing banded and housed in wire-floored stainless steel battery brooders. The brooder temperature was maintained at 35±0.5°C until 7 d of age and gradually decreased to 27°C by 21 d of age, after which, chicks were maintained at room temperature (20 to 27°C). Birds were vaccinated against Newcastle (7th and 28th d) and infectious bursal diseases (15th d) as per the standard vaccination schedule. The experimental protocol was approved by the institute animal ethics committee.
Maize and soybean meal-based diets were prepared to contain 2,944, 3,049 and 3,150 kcal ME/kg during the prestarter (1 to 11 d), starter (12 to 21 d), and finisher (> 22 d of age) phases, with respective crude protein contents of 23, 21, and 19 g/100 g feed (Table 1). The maize and soybean meal were analyzed for amino acid concentrations (Llames and Fontaine, 1994). The analyzed amino acid composition was utilized for formulating the experimental diets. Further, the diets were supplemented with organic Se (MinPlex Selenium, Bentoli AgriNutrition, Homestead, Florida, USA) to arrive five graded concentrations in diet I (basal diet without organic Se); diet II (diet I+organic Se at 100 μg/kg diet); diet III (diet I+organic Se at 200 μg/kg diet); diet IV (diet I+organic Se at 300 μg/kg diet); diet V (diet I+organic Se at 400 μg/kg diet), and each diet was allotted at random to eight replicates and fed ad libitum from 1 to 42 d of age. The weighed amount of organic Se was incorporated in the premix for various diets in order to attain the desired concentration and finally the premix was thoroughly mixed with the respective diets using stainless steel double cone blenders with 300 rpm for 3 min.
Productive performance and carcass parameters
Body weight and feed intake were recorded at 21 and 42 d of age, and feed efficiency per pen was calculated as body weight gain per unit feed intake.
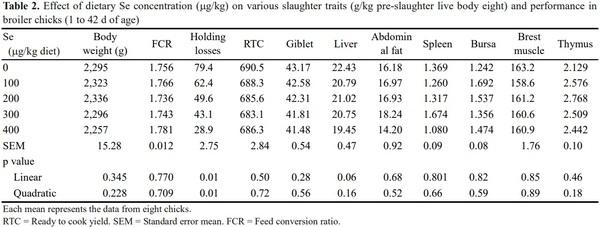
At 42 d of age, eight birds from each treatment weighing near to the mean body weight (±5%) of the respective group were selected and removed from feed for 12 h to record body weight loss during pre-slaughter holding period. The birds were slaughtered by cervical dislocation to study carcass traits. Ready to cook yield (including giblet) and relative weight of breast, abdominal fat, and liver were recorded and expressed as grams per kilogram pre-slaughter live body mass of the respective bird.
Stress parameters
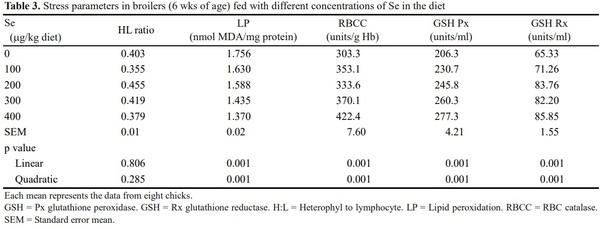
Stress responses in terms of LP, activities of certain antioxidant enzymes, and HL ratio in peripheral blood smears were studied. Approximately 4 ml of blood was drawn from the brachial vein of eight birds per treatment on 35th d of age into a centrifuge tube containing citrate buffer (citrate 0.8 g/100 ml and 1.5 ml/10 ml blood) for erythrocyte separation and enzyme estimation. Blood samples were centrifuged at 500xg for 15 min at 4°C to separate the buffy coat and RBC pellet. The RBC were washed thrice with PBS (pH 7.4). The RBC pellet was mixed with an equal volume of PBS, and then lysed using distilled water (RBC to distilled water, 1:20) and the same procedure was applied for quantifying the LP (Ohkawa et al., 1979), activity of RBCC (Berg Meyer, 1983). The degree of peroxidation was expressed as nanomoles malonyl dialdehyde (MDA)/milligram protein and the RBCC activity as units per gram of hemoglobulin, respectively. The hemolysate was also analyzed for the activities of GSH Rx (Cohen et al., 1970) and GSH Px (Paglia and Valantine, 1967), which were expressed as units/milliliter. At 42 d of age, blood smears (eight birds from each treatment) were prepared and stained with Giemsa, and 100 cells were counted to calculate HL ratio.
Immune responses
The effect of supplementing Se on humoral (ND virus) and cell-mediated (LPR) immune responses were studied. Blood samples were collected from one bird from each pen (eight birds from each treatment) on the 40th d of age to study the LPR and antibody titres against NDV.
In vitro LPR
In vitro LPR was determined using the mitogen Concanavalin A (Con A) and determined as LPR between the proliferation of mitogen-stimulated and unstimulated cells. The LPR was measured using 3-(4, 5-dimethylthiazol2-yl)-2, 5-diphenyl tetrazolium bromide (MTT) salt (Bounous et al., 1992). At 40 d of age, approximately 2 ml blood was collected from the brachial vein of the bird from each replicate and transferred to a centrifuge tube containing 5 mg heparin disodium salt. The unclotted blood sample was layered gently over histopaque 1077 (Sigma, Mumbai, India) and centrifuged at 500xg for 20 min at 4°C. The cellular band at the interface was collected and transferred to another tube and washed three times with RPMI 1640 medium (AL 028A, Himedia, India). Numbers of viable cells were determined by using the trypan blue dye exclusion method, and cell concentration was adjusted to 1x107 cells/ml of RPMI 1640 medium. A sample of cells (105 purified lymphocytes) was used to measure lymphocyte proliferation by adding 10 μl of suspension to each well of a 96-well flat bottom sterile tissue culture plate. Con A (0.9 μg in 150 μl RPMI/well) was added to those cells to be stimulated. A similar volume of RPMI was added to the unstimulated cultures. The plate was incubated at 37°C and 5% CO2 concentration for 69 h in a humid atmosphere, then 20 μl MTT (10 mg/ml) was added to each well, and the plate was incubated for 3 h. At 72 h, 100 μl of 1 M HCl-isopropanol (4%) was added to each well and mixed thoroughly with a micropipette to dissolve the formazan crystals, which gave a deep purple color. The color intensity was measured in an enzyme-linked immunosorbent assay (ELISA) reader (V. 200.1, μ Quant; Biotek Instruments, Inc., USA) at 550 nm. The LPR was calculated as (OD of Con A well-OD of without Con A well)/OD of unstimulated cells.
Antibody titre to ND vaccine
Broilers were vaccinated against ND by ocular route at the 7th and 28th d of age with Lsota strain (ND Lasota Vac500; Indivax Pvt., Ltd., Hyderabad, India). At 40 d of age, blood was collected and a serum was separated. Antibody titres against ND vaccine were measured with ELISA (96- 6547; Synbiotics, San Diego, CA, USA).
Statistical analysis
The data were subjected to regression analysis considering the levels of Se as independent variable and various parameters (production, slaughter, stress, and immune responses) as dependent variables to know the relation (linear or nonlinear) between independent and dependant variables using SPSS software (SPSS Version 12; South Asia Pvt., Ltd., Bangaluru, India).
RESULTS AND DISCUSSION
Productive performance and carcass traits
The results of the present study showed the body weight gain, feed efficiency and slaughter traits (relative weight of ready to cook, giblet, liver, abdominal fat, spleen, bursa, breast muscle and thymus) were not affected (p> 0.05) with Se supplementation (Table 2). The similar results have been reported, wherein Se supplementation revealed insignificant effects on body weight gain and feed efficiency (Downs et al., 2000; Choct et al., 2004; Payne and Southern, 2005; Perić et al., 2009) in broilers. On the contrary, it has been reported that the improved body weight and feed efficiency in chicken reared under the heat stress, when fed diet supplemented with Se (Christine et al., 2002; Niu et al., 2009; Ibrahim et al., 2011). Similarly, increased feed efficiency and body weight gain is reported in broilers chicken fed diet supplemented Se-enriched yeast (Krstic et al., 2012). The insignificant difference in body weight gain, feed efficiency and other slaughter parameters in the present study might be due to feeding the balanced diet with adequate nutritional practice (Lesson and Summer, 2001).
Stress parameters
LP decreased linearly (p< 0.01) with each increment of Se in diet (Table 3). On the contrary, the activities of glutathione peroxidase (GSH Px), glutathione reductase (GSH Rx) and red blood cell (RBC) catalase (RBCC) increased linearly (p< 0.01) with increase in Se concentration in diet. However, variation in concentration of Se in diet did not influence (p> 0.05) the heterophyl to lymphocyte (HL) ratio in broilers. It is well established that the LP, a process involved in degradation of structural components of cell membranes (Kuhn and Borchert, 2002; Georgieva et al., 2011) and ROS accumulation is known to induce LP and oxidative stress (Devi et al., 2000). Whereas, the RBCC, GSH Px and GSH Rx protects the biological cells from the adversities of ROS (Georgieva et al., 2011). GSH Px and GSH Rx are well known for catalyzing the reduction of hydrogen peroxide and organic hydroperoxides, thus protecting cells from oxidative damage (Papp et al., 2007). In the present study, decreased LP and increased activities of the above antioxidant enzymes suggest that Se supplementation progressively reduced oxidative stress with gradient increase in dietary Se concentration (Cave et al., 2006) in broiler. The cell-mediated immune response (LPR) increased significantly with the concentration of Se in broiler diet. Similar findings are also reported in domestic animals (Finch and Turner, 1996), humans (McKenzie et al., 1998) and broilers (Singh et al., 2006) fed diets with Se supplementation.
Immune responses
Selenium is an important for optimum immune response, and it influences the innate and acquired immune systems. Effect of dietary Se concentration on antibody titres to ND vaccine and lymphocyte proliferation ratio (LPR) in commercial broilers is presented in Table 4. Antibody titres against ND vaccine was not affected (p> 0.05) by feeding the diet containing varying concentration of Se, which might be due to ideal ambient temperature during finisher phase. Contrary, in vitro cell-mediated immune response (LPR) increased linearly (p< 0.001) with increasing the concentration of Se (up to 400 μg/kg) in broiler diet. The immune modulator effect of Se might have mediated through reduction of stress in chicken as evident by increased antioxidant (GSH Px, GSH Rx, and RBCC) and decreased LP in the present study. Similarly, it has also been reported that Se or vitamin E greatly influences the humoral immune response up to 30 wks of age (Marsh et al., 1981), but not during the later phase in broilers. Similarly, Zhang et al. (2012) reported that the selenium supplementation in the chicken diets improved the immunological parameters.
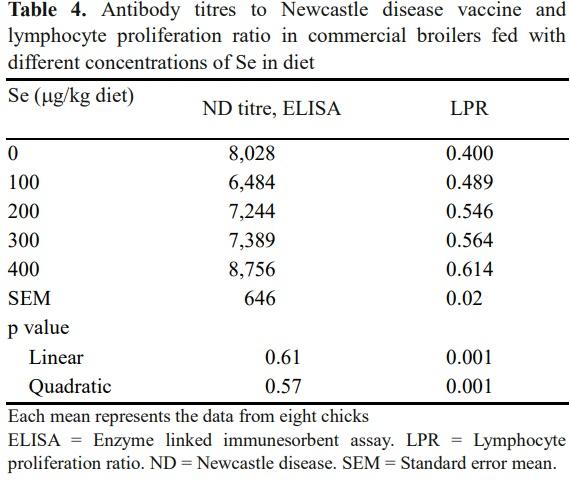
Further, they reported that the deficiency in Se in the diets results in increased levels of oxidative stress and its related diseases. However, the lack of response in humoral immunity in the present study might be due to the study of the parameter was carried out at the latter phase of the broiler growth.
CONCLUSIONS
Based on the results, it is concluded that supplementation of Se did not influence body weight and feed efficiency. However, Se supplementation (up to 400 μg/kg diet) increased anti-oxidant status (reduced LP and increased activities of GSH Px, GSH Rx, and RBCC) and LPR.
This article was originally published in Asian-Australasian Journal of Animal Sciences, Vol. 26, No. 2: 247-252 February 2013. http://dx.doi.org/10.5713/ajas.2012.12299. This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/). 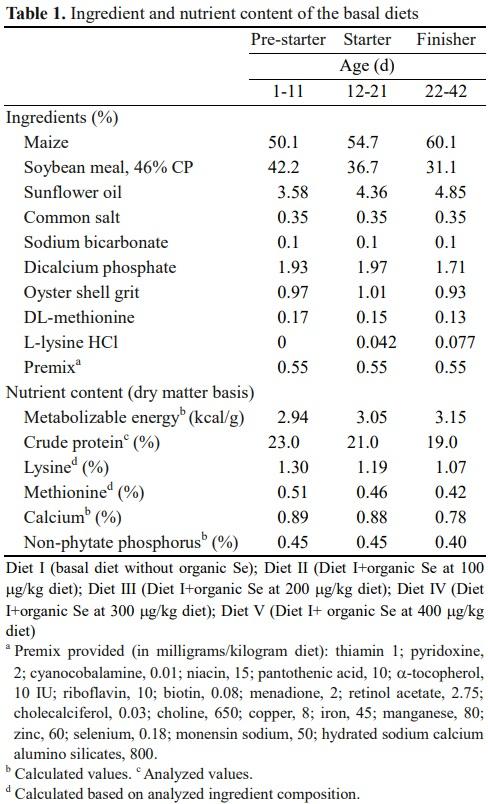












.jpg&w=3840&q=75)