Effect of Diets with Different Inclusion Levels of Distillers Dried Grain with Solubles Combined with Lysine and Methionine Supplementation on the Lipid Peroxidation and Glutathione Status of Chickens
Published: February 18, 2014
By: Mónika HEINCINGER1, Krisztián BALOGH1,2, Hedvig FÉBEL3, Márta ERDÉLYI1
and Miklós MÉZES1*
1Department of Nutrition, Faculty of Agricultural and Environmental Sciences, Szent István University, Páter K. u. 1, H-2103 Gödöllo, Hungary;
2Research Group of Animal Breeding and Hygiene, Faculty of Animal Science, University of Kaposvár, Kaposvár, Hungary;
3Research Institute for Animal Breeding and Nutrition, Herceghalom, Hungary
2Research Group of Animal Breeding and Hygiene, Faculty of Animal Science, University of Kaposvár, Kaposvár, Hungary;
3Research Institute for Animal Breeding and Nutrition, Herceghalom, Hungary
Acta Veterinaria Hungarica 59 (2), pp. 195–204 (2011)
DOI: 10.1556/AVet.2011.005
First published online 5 April 2011
DOI: 10.1556/AVet.2011.005
First published online 5 April 2011
To study the possible effects of different inclusion levels of distillers dried grain with solubles (DDGS) on the lipid peroxidation and glutathione redox status of chickens, 200 three-week-old Ross 308 cockerels were assigned to four treatment groups of 50 birds each. The groups were fed a control and three experimental, isocaloric and isonitrogenous grower diets containing 15, 20 and 25% DDGS, respectively, combined with lysine (Lys) and methionine (Met) supplementation until 6 weeks of age. It was found that DDGS inclusion increased the ether extract content of the diets which resulted in higher reduced glutathione (GSH) content and elevated glutathione peroxidase activity (GSHPx) in the liver. However, DDGS addition with Lys and Met supplementation did not influence the malondialdehyde content of the blood and the liver. The oleic acid proportion of the diet showed a close positive correlation with GSH content of the liver. A smaller ratio of methionine and cysteine in the diet with DDGS resulted in significantly higher liver GSH content. GSHPx activity increased parallel with the elevated GSH content of the liver homogenate, suggesting that the enzyme is activated by the actual supply of its co-substrate. In conclusion, the results show that DDGS, even at a high inclusion level combined with Lys and Met supplementation, has no initiative effect on lipid peroxidation in the blood and liver of broiler chickens.
Key words: Chicken, distillers dried grain with solubles, DDGS, glutathione, glutathione peroxidase, malondialdehyde
Glutathione (γ-glutamylcysteinylglycine) is a tripeptide that exists either in reduced (thiol, GSH) or oxidised (disulphide, GSSG) form (DeLeve and Kaplowitz, 1990). GSH plays a key role in biological antioxidant defence against oxidative stress (Daba and Abdel Rahman, 1998). It is synthesised from its constituent amino acids, and the liver plays a central role in its whole-body homeostasis (Wang et al., 1997). The synthesis of GSH is affected by the actual methionine and cysteine supply as it has been demonstrated in poultry (Németh et al., 2004) and rats (Wang et al., 1997). The availability of GSH depends on the flux of GSH and cysteine between sites of biosynthesis and sites of utilisation (Giustarini et al., 2009). Cysteine transport to the cells is more effective as compared to that of cystine (Segal and Crawhall, 1968); therefore, the availability of cysteine in the blood circulation has primary importance for glutathione synthesis. In neuronal and astroglial cells it was found that the replacement of cysteine by cystine during a re-feeding period resulted in a lower intracellular amount of glutathione (Kranich et al., 1996). When feeding a low-protein diet, the formation of GSH depends on the level of homocysteine which is converted by transsulphuration into cysteine and then to GSH (Deminice et al., 2009). As regards the other amino acids, an extremely high dietary methionine level has been shown to cause oxidative stress in rabbits (Yalcinkaya-Demirsöz et al., 2009) and it also depresses the specific activity of glutathione peroxidase (GSHPx) in mouse erythrocytes (He and Yasumoto, 1992). The antioxidant defence of erythrocytes is essential for erythrocyte lifespan, and it partly depends on the glutathione redox system, namely the amount of GSH and the activity of GSHPx, as it has been demonstrated in mammalian species (Kurata et al., 1993). Excess methionine has been shown to accelerate the senescence of erythrocytes (He and Yasumoto, 1992). Additionally, hepatic GSHPx activity and GSH level were found to have a close and statistically significant negative correlation with maximum lifespan in vertebrates (Lopez-Torres et al., 1993).
Glutathione (γ-glutamylcysteinylglycine) is a tripeptide that exists either in reduced (thiol, GSH) or oxidised (disulphide, GSSG) form (DeLeve and Kaplowitz, 1990). GSH plays a key role in biological antioxidant defence against oxidative stress (Daba and Abdel Rahman, 1998). It is synthesised from its constituent amino acids, and the liver plays a central role in its whole-body homeostasis (Wang et al., 1997). The synthesis of GSH is affected by the actual methionine and cysteine supply as it has been demonstrated in poultry (Németh et al., 2004) and rats (Wang et al., 1997). The availability of GSH depends on the flux of GSH and cysteine between sites of biosynthesis and sites of utilisation (Giustarini et al., 2009). Cysteine transport to the cells is more effective as compared to that of cystine (Segal and Crawhall, 1968); therefore, the availability of cysteine in the blood circulation has primary importance for glutathione synthesis. In neuronal and astroglial cells it was found that the replacement of cysteine by cystine during a re-feeding period resulted in a lower intracellular amount of glutathione (Kranich et al., 1996). When feeding a low-protein diet, the formation of GSH depends on the level of homocysteine which is converted by transsulphuration into cysteine and then to GSH (Deminice et al., 2009). As regards the other amino acids, an extremely high dietary methionine level has been shown to cause oxidative stress in rabbits (Yalcinkaya-Demirsöz et al., 2009) and it also depresses the specific activity of glutathione peroxidase (GSHPx) in mouse erythrocytes (He and Yasumoto, 1992). The antioxidant defence of erythrocytes is essential for erythrocyte lifespan, and it partly depends on the glutathione redox system, namely the amount of GSH and the activity of GSHPx, as it has been demonstrated in mammalian species (Kurata et al., 1993). Excess methionine has been shown to accelerate the senescence of erythrocytes (He and Yasumoto, 1992). Additionally, hepatic GSHPx activity and GSH level were found to have a close and statistically significant negative correlation with maximum lifespan in vertebrates (Lopez-Torres et al., 1993).
The objective of this experiment was to study the effect of different DDGS inclusion levels combined with lysine and methionine supplementation on lipid peroxidation in broiler chickens, as measured by its meta-stable end-product, malondialdehyde (Tappel and Dillard, 1981) and some key redox parameters such as GSH content and GSHPx activity in the blood and liver.
Materials and methods
Experimental birds and diets
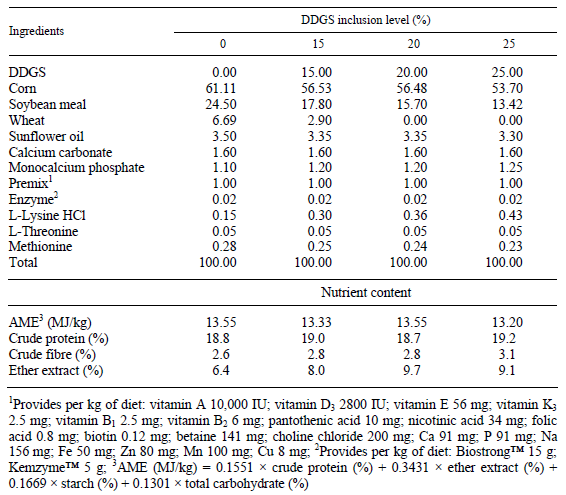
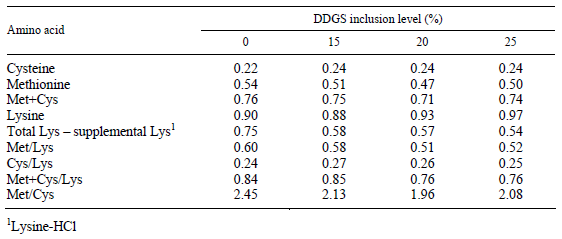
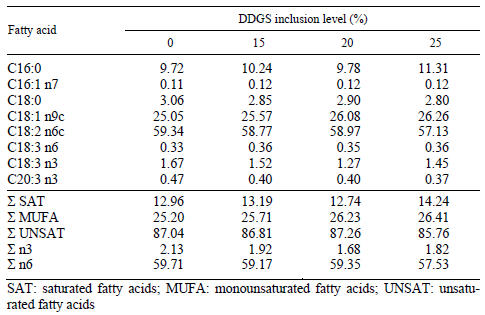
Cockerel chicks of the Ross 308 genotype (n = 200) were reared up to 3 weeks of age on deep litter and fed a commercial diet. At 3 weeks of age, the chicks were randomly divided into four different groups (50 birds in each) and fed a control and three different isocaloric and isonitrogenous grower diets containing different rates of corn-based distillers grain with solubles (DDGS) and amino acids until six weeks of age. Amino acid (lysine-HCl and methionine) supplementation was calculated according to the measured amino acid composition of DDGS (Lys: 0.6%; Met: 0.56% and Cys: 0.52%) as compared to the amino acid content of other feed components, corn, soybean meal and wheat (Lys: 2.95, 6.09 and 2.70%; Met: 2.11, 1.39 and 1.55%; Cys: 2.26, 1.57 and 2.24%) which were replaced in the experimental diets. The composition and nutrient contents of the experimental diets are shown in Table 1. The analysed amino acid composition and calculated essential amino acid ratios of the diets are presented in Table 2 and the fatty acid composition of the experimental diets is shown in Table 3.
The experimental protocol met the standard criteria of the Scientific Ethics Committee for Animal Experiments of Szent István University.
Sample collection
Blood and liver samples were taken from six randomly selected birds in each group at the end of the feeding trial at 6 weeks of age. Blood was taken from the wing vein into tubes containing disodium EDTA (0.2 M/L; 0.05 ml/ml blood), then the chickens were slaughtered by decapitation, and the liver was removed immediately after exsanguination.
Blood was centrifuged (2500 g for 10 min at +4 °C) within 1 h after sampling. The plasma was separated and red blood cell (1:9 v/v) haemolysates were made with redistilled water and by freezing and thawing. Plasma and red blood cell (RBC) haemolysate samples were stored at –70 °C until analysed. Liver samples were taken from the right lobe and stored at –70 °C until the analysis. Immediately before analysis, liver samples were homogenised with a ninefold amount of cold (+4 °C) isotonic saline (0.65% w/v NaCl) in an Ultra-Turrax homogeniser (Donau Lab AG, Linz, Austria).
Table 1 Composition and nutrient content of the experimental diets (%)
Chemical and biochemical analyses
Chemical composition (crude protein, ether extract, crude fibre, starch and total carbohydrates) of the experimental diets was measured according to Hungarian National Standard methods (Hungarian Feed Code, 2004). Amino acid content of the diets was measured by ion-exchange amino acid analysis of total hydrolysates of the complete feeds by HPLC with pre-column derivatisation (Csapó et al., 2008). Total lipids were extracted from the diets by the method of Folch et al. (1957) and saponified and methylated using boron trifluoride as methylating agent (Morrison and Smith, 1964). The fatty acid composition of methyl esters was analysed with a gas chromatograph (Shimadzu 2010, Kyoto, Japan) equipped with a SP-2380-type fused silica capillary column (30 m, 0.25 mm i.d., 0.25 μm film, Supelco Inc., Bellefonte, USA). Malondialdehyde content of the blood plasma, RBC haemolysate and crude liver homogenate was determined using the 2-thiobarbituric acid assay of Placer et al. (1966), while reduced glutathione (GSH) content and glutathione peroxidase (GSHPx) activity of the blood plasma, RBC haemolysate and 10,000 g supernatant fraction of liver homogenate were measured according to Sedlak and Lindsay (1968) and Lawrence and Burk (1976), respectively.
Table 2 Measured amino acid composition (%) of the experimental diets
Statistical analysis
The results were evaluated by the comparison of means and calculated by ANOVA and coefficient of correlation with linear correlation analysis using the SPSS 15.0 software (SPSS, 2006).
Table 3 Ratio of main fatty acids in the experimental diets (g/100 g fatty acid)
Results
There was no significant difference between the groups in the malondialdehyde content of the different tissues (Table 4).
Table 4 Malondialdehyde (MDA) and reduced glutathione (GSH) content and glutathione peroxidase (GSHPx) activity of blood plasma, red blood cell haemolysates and liver homogenate
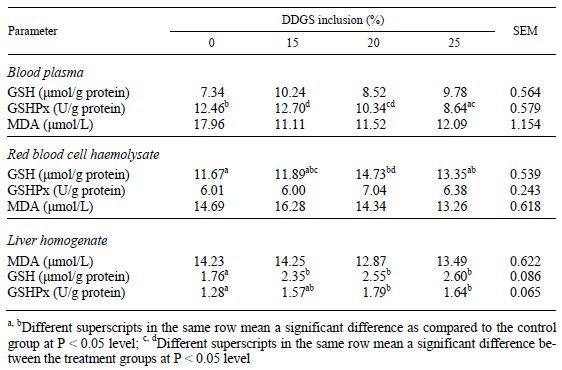
GSH content of the blood plasma did not differ significantly among the treatment groups. GSH content of the RBC haemolysates was significantly higher in the groups fed a diet containing 20% DDGS. GSH content of the liver homogenate showed a dose-dependent increase. GSHPx activity in the blood plasma was lower at 20 and 25% DDGS inclusion rates as compared to the control. There was no significant difference between the treatment groups in the GSHPx activity of RBC haemolysates. GSHPx activity of the 10,000 g supernatant fraction of the liver homogenate was higher in each group fed a DDGScontaining diet with amino acid supplementation as compared to the control.
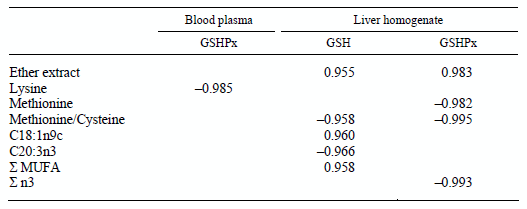
A significant positive correlation was found between ether extract content of the diets and GSH level as well as GSHPx activity in the liver homogenate (Table 5). However, Met/Cys ratio of the diet had a close negative correlation with the measured glutathione redox status parameters of the liver. GSHPx activity in the liver homogenate was in a significant negative correlation with Met content of diet. Lys content of experimental diets had a close negative significant correlation with GSHPx activity of blood plasma. There was a close negative significant correlation between GSHPx activity and the proportion of total n3 fatty acids of the diets. GSH content of the liver showed a close positive correlation with the proportion of oleic acid (C18:1n9) and total monounsaturated fatty acids in the diet, while it was in a close negative correlation with the dietary ratio of eicosatrienoic acid (C20:3n3).
Table 5 Significant correlation (P < 0.05) between nutrient content and fatty acid composition of the diets and the parameters of glutathione redox status
Discussion
The results of the present study show that the inclusion of DDGS, even at high (up to 25%) rates, did not cause an increased formation of lipid peroxidation end-products, namely malondialdehyde, in the blood and liver of broiler chickens. It was found that higher DDGS inclusion levels slightly increased the total ratio of MUFA; however, the proportion of total n3 and n6 fatty acids decreased with increasing DDGS inclusion rate. Different changes observed in the fatty acid composition of experimental diets did not cause a higher rate of lipid peroxidation, as was measured by MDA concentration in blood and liver. These results mean that a higher oxidative susceptibility of blood and liver, similar to that found in broiler chicken meat when DDGS was fed (Loar et al., 2009), should not be expected.
Glutathione synthesis is affected by different factors such as age (Wang et al., 1997) and amino acid, mainly Met and Cys, supply (Singer, 1975; Németh et al., 2004). However, according to our results it also depends on the supply of other amino acids such as Lys, as well as on the ratio between Met and Cys. One of the two key amino acids for GSH synthesis, Cys, was slightly higher in the diets containing DDGS, which supports the above hypothesis, and confirms the significantly higher GSH content in the liver, the main site of GSH synthesis (Wang et al., 1997). A similar effect of additional amino acids on glutathione synthesis has been found in yeasts as well (Wen et al., 2006). However, the Met/Cys ratio of the diet has a close negative correlation with GSH synthesis in the liver, which means that the ratio between Met and Cys has a primary effect on GSH synthesis. Additionally, the ether extract content of the diets had a close positive correlation with liver GSH content, which means that the actual energy supply is also important in GSH synthesis, besides the amino acid supply. However, the fatty acid composition of fats also has an effect on GSH synthesis, possibly because of their different rate of transformation into energy. The correlation coefficients showed that the dietary proportion of some fatty acids, such as oleic (C18:1 n9) acid, had a positive, while eicosatrienoic (C20:3 n3) acid had a negative effect on GSH synthesis in the liver. In addition, the ratio of total MUFAs in the diet was found to have a positive effect on GSH concentration in the liver.
GSHPx activity increased parallel with the elevated GSH content of the liver homogenate, which is in agreement with the findings of our previous studies (Németh et al., 2004; Balogh et al., 2007). This finding supports the hypothesis that GSHPx is an allosteric enzyme (Perona et al., 1978), which is activated by the actual supply of its co-substrate, GSH. Ether extract content of the diets has a close positive correlation with GSHPx activity, which was also found in rats fed diets with adequate fat content according to the actual requirement (Kuratko and Pence, 1991). Total n-3 fatty acids had a close negative correlation with GSHPx activity in the liver. This finding was contrary to the result of Crosby et al. (1996) who found that saturated fatty acids had no or negative, while unsaturated fatty acids had a positive effect on GSHPx activity. Furthermore, results of present study do not support the findings of Venkatraman et al. (1998) who found that unsaturated fatty acids of the n-6 group had more pronounced effect as compared to the n-3 group. In contrast to the liver, no significant changes in GSHPx activity were found in the red blood cell haemolysate, even at a higher GSH content; moreover, GSHPx activity was significantly decreased in the blood plasma without marked changes in GSH concentration. These contradictory results might be partly explained by the different kinetic properties of GSHPx isoenzymes (Brigelius-Flohé, 1999). The close negative correlation found between Met/Cys ratio in the diet and GSH and GSHPx in the liver homogenate may also support these findings.
In conclusion, the results of the present study show that DDGS, even at a high inclusion level, has no initiating effect on lipid peroxidation in the blood and liver of broiler chickens. However, the higher ether extract content of the DDGS-containing diets resulted in lower GSHPx activity in the blood plasma and significantly higher GSH content and GSHPx activity in the liver.
References
Balogh, K., Weber, M., Erdélyi, M. and Mézes, M. (2007): Investigation of lipid peroxide and glutathione redox status of chicken concerning on high dietary selenium intake. Acta Biol. Hung. 58, 269–279.
Boler, D. D., Gabriel, S. R., Yang, H., Balsbaugh, R., Mahan, D. C., Brewer, M. S., McKeith, F. K. and Killefer, J. (2009): Effect of different dietary levels of natural-source vitamin E in growing-finishing pigs on pork quality and shelf life. Meat Sci. 83, 723–730.
Brigelius-Flohé, R. (1999): Tissue-specific functions of individual glutathione peroxidases. Free Radic. Biol. Med. 27, 951–965.
Calkins, C. R., de Mello, A. S. Jr. and Senaratne, L. S. (2009): Effects of distillers grains on beef carcass quality and palatability. J. Anim. Sci. 87 (E-Suppl. 2), 328–329. [Abstract]
Crosby, A. J., Wahle, K. W. and Duthie, G. G. (1996): Modulation of glutathione peroxidase activity in human vascular endothelial cells by fatty acids and the cytokine interleukin-1 beta. Biochim. Biophys. Acta 1303, 187–192.
Csapó, J., Varga, V., Lóki, K., Albert, C. and Salamon, S. (2008): The influence of extrusion on loss and racemization of amino acids. Amino Acids 34, 287–292.
Daba, M. M. and Abdel Rahman, M. S. (1998): Hepatoprotective activity of thymoquinone in isolated rat hepatocytes. Toxicol. Lett. 95, 23–29.
DeLeve, L. D. and Kaplowitz, N. (1990): Importance and regulation of hepatic glutathione. Semin. Liver Dis. 10, 251–266.
Deminice, R., Portari, G. V., Marchini, J. S., Vannucchi, H. and Jordao, A. A. (2009): Effects of a low-protein diet on plasma amino acid and homocysteine levels and oxidative status in rats. Ann. Nutr. Metab. 54, 202–207.
Folch, J. M., Lees, M. and Sloane-Stanley, G. H. (1957): A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226, 495–509.
Giustarini, D., Dalle-Donne, I., Milzani, A. and Rossi, R. (2009): Oxidative stress induces a reversible flux of cysteine from tissues to blood in vivo in the rat. FEBS J. 276, 4946–4958.
He, P. and Yasumoto, K. (1992): Effect of ingestion of excess methionine diet on aging of erythrocytes in mice. J. Nutr. Sci. Vitaminol. (Tokyo) 38, 57–68.
Hungarian Feed Code (2004): Determination of nutrients of feedingstuffs [in Hungarian]. Vol. II/II. OMMI (National Institute for Agricultural Quality Control), Budapest.
Kranich, O., Hamprecht, B. and Dringen, R. (1996): Different preferences in the utilization of amino acids for glutathione synthesis in cultured neurons and astroglial cells derived from rat brain. Neurosci. Lett. 219, 211–214.
Kurata, M., Suzuki, M. and Agar, N. S. (1993): Antioxidant systems and erythrocyte life-span in mammals. Comp. Biochem. Physiol. 106B, 477–487.
Kuratko, C. and Pence, B. C. (1991): Changes in colonic antioxidant status in rats during long-term feeding of different high fat diets. J. Nutr. 121, 1562–1569.
Lawrence, R. A. and Burk, R. F. (1976): Glutathione peroxidase activity in selenium deficient rat liver. Biochem. Biophys. Res. Commun. 71, 952–956.
Loar, R. E., Schilling, M. W. and Corzo, A. (2009): Organoleptic and compositional effects of feeding distillers dried grains with solubles on broiler meat. Poult. Sci. 88 (Suppl. 1), 149 (Abstract M53).
Lopez-Torres, M., Perez-Campo, R., Rojas, C., Cadenas, S. and Barja, G. (1993): Maximum life span in vertebrates: relationship with liver antioxidant enzymes, glutathione system, ascorbate, urate, sensitivity to peroxidation, true malondialdehyde, in vivo H2O2, and basal and maximum aerobic capacity. Mech. Ageing Dev. 70, 177–199.
Lumpkins, B. S., Batal, A. B. and Dale, N. M. (2004): Evaluation of distillers dried grains with solubles as a feed ingredient for broilers. Poult. Sci. 83, 1891–1896.
Morrison, W. R. and Smith, L. M. (1964): Preparation of fatty acid methyl esters and dimethyl acetals from lipids with boron trifluoride-methanol. J. Lipid Res. 5, 600–608.
Németh, K., Mézes, M., Gaál, T., Bartos, Á., Balogh, K. and Husvéth, F. (2004): Effect of supplementation with methionine and different fat sources on the glutathione redox system of growing chickens. Acta Vet. Hung. 52, 369–378.
Parsons, C. M. and Baker, D. H. (1983): Distillers dried grains with solubles as a protein source for the chick. Poult. Sci. 62, 2445–2451.
Perona, G., Guidi, G. C., Piga, A., Cellerino, R., Menna, R. and Zatti, M. (1978): In vivo and in vitro variations of human erythrocyte glutathione peroxidase activity as result of cells ageing, selenium availability and peroxide activation. Br. J. Haematol. 39, 399–408.
Placer, Z. A., Cushman, L. L. and Johnson, B. C. (1966): Estimation of product of lipid peroxidation (malonyl dialdehyde) in biochemical systems. Anal. Biochem. 16, 359–364.
Sedlak, I. and Lindsay, R. H. (1968): Estimation of total protein-bound and non-protein sulfhydryl groups in tissues with Ellmann’s reagent. Anal. Biochem. 25, 192–205.
Segal, S. and Crawhall, J. C. (1968): Characteristics of cystine and cysteine transport in rat kidney cortex slices. Proc. Natl Acad. Sci. USA 59, 231–237.
Singer, T. P. (1975): Oxidative metabolism of cysteine and cystine in animal tissues. In: Greenberg, G. M. (ed.) Metabolic Pathways. Vol. 7: Metabolism of Sulfur Compounds. 3rd edition. Academic Press, New York. pp. 535–546.
SPSS (2006): User’s Guide to 15.0.1 software. SPSS Inc., Michigan.
Tappel, A. L. and Dillard, C. J. (1981): In vivo lipid peroxidation: Measurement via exhaled pentane and protection by vitamin E. Fed. Proc. 40, 174–178.
Yalcinkaya-Demirsöz, S., Depboylu, B., Dogru-Abbsoglu, S., Unlücerci, Y. and Uysal, M. (2009): Effects of high methionine diet on oxidative stress in serum apo-B containing lipoproteins, heart, and aorta in rabbits. Ann. Clin. Lab. Sci. 39, 386–391.
Venkatraman, J. T., Angkeow, P., Satsangi, N. and Fernandes, G. (1998): Effects of dietary n-6 and n-3 lipids on antioxidant defense system in livers of exercised rats. J. Am. Coll. Nutr. 17, 586–594.
Waldroup, P. W., Owen, J. A., Ramsey, B. E. and Whelchel, D. L. (1981): The use of high levels of distillers dried grains plus solubles in broiler diets. Poult. Sci. 60, 1479–1484.
Wang, S. T., Chen, H. W., Sheen, L. Y. and Lii, C. K. (1997): Methionine and cysteine affect glutathione level, glutathione-related enzyme activities and the expression of glutathione-Stransferase isoenzymes in rat hepatocytes. J. Nutr. 127, 2135–2141.
Wang, Z., Cerrate, S., Coto, C., Yan, F. and Waldroup, P. W. (2007): Utilization of distillers dried grains with solubles (DDGS) in broiler diets using a standardized nutrient matrix. Int. J. Poult. Sci. 6, 470–477.
Wen, S., Zhang, T. and Tianwei Tan, T. (2006): Maximizing production of glutathione by amino acid modulation and high-cell-density fed-batch culture of Saccharomyces cerevisiae. Process Biochem. 41, 2424–2428.
Wood, J. D., Whittington, F. M. and Hallett, K. G. (2009): By-product feeding effects on pork quality and carcass traits. J. Anim. Sci. 87 (E-Suppl. 2), 329 [Abstract].
Authors:
Join to be able to comment.
Once you join Engormix, you will be able to participate in all content and forums.
* Required information
Would you like to discuss another topic? Create a new post to engage with experts in the community.
Create a post







