Effect dietary glutamine and vitamin E supplementation on performance, some blood antioxidant indices in broiler chickens under continuous heat stress temperature
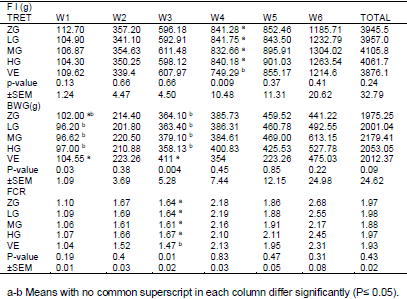
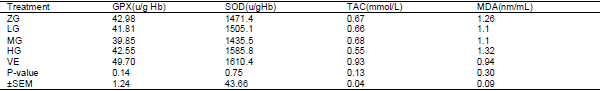
Effects of different levels of 0.0 glutamine (ZG), 0.25 (LG), 0.50 (MG) and 1 percent (HG) along with 100 mg/kg VE were investigated on performance and blood antioxidant status of broiler chickens under heat stress condition. There was significant differences were observed between the treatments for FI during week 4 the experimental period. No significant difference were observed between the treatments for FI during the whole the experimental periods. There was significant differences were observed between the treatments for BWG during the week 1and 3 the experimental period, wasn’t significant differences between the treatments for BWG during the whole the experimental period. Meanwhile during the whole period FI of VE birds were lower than that of ZG, LG and HG ones. For whole the experimental period, BWG of MG and VE fed birds was higher than that of ZG and LG fed birds. There was significant difference between the treatments for FCR during the week 3 experimental period; there wasn’t significant difference between the treatments for FCR during the whole experimental period. Meanwhile, during the whole period FCR of VE and MG birds were lower than that of ZG, LG and HG ones. There wasn't significant differences were observed between the treatments for TAC, MDA, SOD and GPx between the treatments for during experiment period. It was concluded that although dietary Glutamine and VE consumption can improve the performance of broiler chickens during the weekly period under heat stress.
Keywords: antioxidant status, broiler chickens, heat stress, performance, Glutamine.
Bartell SM and Batal AB. 2007. The effect of supplemental glutamine on growth performance, development of the gastrointestinal tract and humoral immune response of broilers. J Poult Sci, 86:1940–1947.
Chance BHS and Boveris A. 1979. Hydro peroxide Metabolism in Mammalin Organs. Physiol Rev, 59: 527-605.
De Colnago GL, Jensen LS and Long PL. 1984. Effects of selenium and vitamin E on the development of immunity to coccidiosis in chickens. Poult. Sci. 63:1136–1143.
Dai SF, Gaoa F, Song SX, Zhanga WH , Xub XL and Zhoub GH. 2011. Effects of Dietary Glutamine and Gamma-Amino Butyric Acid on Performance, Carcass Characteristics and Serum Parameters in Broilers Under Circular Heat Stress. J Anim Feed Sci Tech,168:51– 60.
Dale NM and Fuller HL. 1980. Effect of diet composition on feed intake and growth of chicks under heat stress. II .Constantvs. cycling temperatures. Poult., Sci., 59: 1434-1441.
De Winne A and Dirinck P. 1996. Studies on vitamin E and meat quality. 2. Effect of feeding high vitamin E levels on chicken meat quality. J Agric Food Chem., 44: 1691–1696.
Deaton JW, Reece FN and McNaughton JL. 1978. The effect of temperature during the growing period on broiler performance. Poult. Sci., 57: 1070-1074.
De-lian M, Chao S, Jun-hu Y, Gong-she Y and FANG J. 2009. Effect of Aspartate and Glutamine on a Part of the Fatty Traits and the Levels of Adipogenesis Genes mRNA Expression of Broiler. Sci Agricul Sin., 7; 2513-2522.
Donkoh A. 1989. Ambient temperature: a factor affecting performance and physiological response of broiler chickens nt. J. Biometeorol.,33; 259-265.
Duthie GG, Wahle KWJ and James WPT. 1989. Oxidants, Antioxidant's and Cardiovascular Disease.Nutr.Res.Rev.2:52-62.
Ebadiasl G. 2011. Effects of supplemental glutamine and glutamate on growth performance, gastrointestinal development, jejunum morphology and clostridium perfringens count in caecum of broilers, UPPSALA, 07; 1-26.
Edens FW and Siegel HS. 1975. Adrenal responses in high and low ACTH response lines of chicken during acute heat stress. Gen Comp Endocr, 25:64–73.
Ewan RC. 1993. Vitaminas. Pages 457–469 in Dukes: Fisiologia dos Animais Dome´sticos. M. J. Swenson, and W. O. Reece,11th ed. Guanabara Koogan, Rio de Janeiro, Brazil.
Freeman BA and Crapo JD. 1982. Biology of disease: freeradicals and tissue injury. Lab. Invet., 47: 412-426.
Fuller HL and Mora G. 1973. Effect of diet composition on heat increment, feed intake, and growth of chicks subjected to heat stress. Poult. Sci., 52: 2029 (Abs).
Geraert PA, Padilha JCF and Guillaumin S. 1996. Metabolic and endocrine changes induced by chronic heat exposure in broiler chickens: Growth performance, body composition and energy retention. Br J Nutr., 75:195–204.
Gomez E, Duque P, Diaz E, Facal N, Antolin I, Hidalgo C and Diez C. 2002. Effects of acetoacetate and D-β-hydroxybutyrate on bovine in vitro embryo development in serum-free medium.Theriogenology, 57: 1551-1562.
Haggendal l, jonsson L Johansson G , Bjustrom S , Carlsten J and Thoren-Tolling K. 1987 . Catecholamine-mduced free radicals in myocaidial cell necioss on expet-pen - mental stress in pigs Aca Physiol Scand 131 , 447 452 .
Hai LD and Zhang ZY. 2000. The effect of thermal environment on the digestion of broilers. J. Anim. Physiol. Anim. Nutr., 83: 57-64.
Hillman PE, Scott NR and Van Tienhoven A. 1987.Physiological And Adaptations To Hot and Cold Environments. In: Stress Physiology In Livestock, V.III. Poultry , Mk. Yousef , CRC Press Inc.,43-54.
Hosseini-Mansoub N, Chekani-Azar S, Tehrani AA, Lotfi AR and Khosravi Manesh M. 2010. Influence of dietary vitamin E and zinc on performance, oxidative stability and some blood measures of broiler chicken sreared under heat stress (35°C). J. Agrobiol., 27:103-110.
Howlider MAR and Rose SP. 1987. Temperature and growth of broilers. World’s Poult. Sci. J., 43:228-237.
Hurwitz S, Weiselberg M, Eisner U, Bartov I, Riesenfeld G, Sharvit M, Niv A and Bornstein S. 1980. The energy requirements and performance of growing chickens and turkeys as affected by environmental temperature. Poult. Sci.,59:2290-2299.
Kaufmann Y, Spring P and Klimberg VS. 2008. Oral glutamine prevents DMBA-induced mammary carcinogenesis via upregulation of glutathione production. Nutrition 24:462-469.
Kitt J, Miller PS, Lewis AJ and Fischer RL. 2002. Effects of glutamine on growth performance and small intestine villus height in weanling pigs. Pages .in Nebraska Swine Rep. Univ. Nebraska, London. 29-32 .
Kolahi S, Hejazi J, Mohtadinia J, Jalili M and Farzin H. 2011. The evaluation of concurrent supplementation with vitamin E and omega-3 fatty acids on plasma lipid per oxidation and antioxidant levels in patients with rheumatoid arthritis.Internet J. Rheumatol. 7:1cf7.
Kul M, Vurucu S, Demirkaya E, Tunc T, Aydinoz I, Meral C, Kesik V and Alpay F. 2008. Enteral Glutamine and/or Arginine Supplementation Have Favorable Effects on Oxidative Stress Parameters in Neonatal Rat Intestine. J Pediatr Gastr Nutr, 49:85-89.
Leshchinsky TV and Klasing KC. 2001. Relationship between the level of dietary vitamin E and the immune response of broiler chickens. Poult. Sci. 80:1590–1599.
Maiorka A, Silva AVF, Santin E, Borges SA, Boleli IC and Macari M. 2000. Influência da suplementação de glutamina sobre o desempenho e o desenvolvimento de vilos e criptas do intestino delgado de frangos. Arquivo Brasileiro de Medicina Veterinária e Zootecnia; 52:487-490.
McDowell LR,. 989. Vitamins in animal nutrition. In: Mc Dowell, L.R. (Ed.), Comparative Aspects to HumanNutrition: Vitamin E. Academic Press, London, 93–131.
Mok, E, Constantin. B, Favreau F, Neveux N, Magaud C, Delwail A and Hankard R. 2008. L-Glutamine administration reduces oxidized glutathione and MAP kinase signaling in dystrophic muscle of mdx mice. Pediatr. Res. 63:28-273.
Murakami AE, Franco JRG, Sakamoto MI, Natali MRM and Souza LMG. 2007. Supplementation of glutamine and vitamin E on the morphometry of the intestinal mucosa in broiler chickens, Poultry Science,86; 488–495.
Naziro glu M, sahin K, simsek H, Aydilek N and Ertas ON. 2000. The effect of food withdrawal and darkening on lipidperoxidation of laying hens in high ambient temperatures.Dtsch. Tierarztl. Wschr., 107, 199–202.
NRC. 1994. Nutrient requirements of poultry. 9th. Revised ed.,National Academy Press, Washington, DC.
Peter G and Elia M. 1997. The use of glutamine in the treatment of gastrointestinal disorders in man.J Nutr, 13, 743–747.
Puthpongsiriporn U, Scheideler SE, Sell JL and Beck MM. 2001. Effects of vitamin E and C supplementation on performance, in vitro lymphocyte proliferation and antioxidant status of laying hens during heat stress. Poult. Sci, 80: 1190-1200.
Rymer C and Givens DI. 2005. N-3 fatty acid enrichment of edible tissue of poultry: A review. Lipids 40: 121–130.
Sahin K, Sahin N, Onderci M, Yaralioglu S and Kucuk O. 2001. Protective role of supplemental vitamin E on lipid peroxidation, vitamins E, A and some mineral concentrations of broilers reared under heat stress. Vet. Med. – Czech, 46, 140–144.
Sahin, K, Kucuk, O, Sahin, N and Gursu MF. 2002. Optimal dietary concentration of vitamin E for alleviating the effect of heat stress on performance, thyroid status ACTH and some serum metabolite and mineral concentrations in broilers. J. Vet.Med.-Czech 47:110-116.
Sakamoto MI, Murakami AE, Silveira TGV, Fernandes JIM and Oliveira CAL. 2006. Influence of Glutamine and Vitamin E on the Performance and the Immune Responses of Broiler Chickens. Brazil j Poult Sci 8:243-249.
SAS 2002. SAS Users Guide: Statistics. SASInstitute Inc., Cary, NC, USA.
Shu G, Fu WL, Jiang1 QY, Gao P, Sun FW, Xu PW, Zhou GX and Zhu XT. 2007. Effects of glycyl-glutamine on the growth performance and lipid deposition in yue-huang broilers. J Anim Feed Sci, 16:232-240.
Singal PK, Kapur N, Dhillon KS, Beamish RE and Dahalla NS. 1982.Role Of Free Radical In Catecholamine Induces Cardiomyopathy,Can.J.Physiol.Oharmacol.60:1390-1397.
Singleton KD and Wischmeyer PE. 2008. Glutamine attenuates inflammation and NF-kappa B activation via Cullin-1 deneddylation. Biochem. Biophys. Res. Commun. 373:445-449.
Soltan MA. 2009. Influence dietary supplementation on growth performance, small intestinal morphology, immune response and some blood parameters broiler chickens, International of Poultry Science , 1; 60-68.
Souba WW. 1993. glutamine and cancer.ann.surg., 218;715-728.
Teeter RG, Smith MO and Owens FN. 1985.Chronic Heat Stress and Respiratory Alkalosis: Occurence and Treatment in Broiler Chicks. Poult. Sci., 64: 1060-1064.
Tengerdy RP and Nockels CF. 1973. The effect of vitamin E on egg production, hatchability and humoral immune response of chickens. Poult. Sci., 52: 778-783.
Thomas OP, Farran M and Tamplin CB. 1992. Broiler nutrition update. in: Proc. Maryland Nutr.Conf., College Park, MD. 45-53.
Villar PG, Diaz CA, Avila GE, Guinzberg R, Pablos JL and Pina E. 2002. Effects of dietary Supplementation with vitamin C or vitamin E on growth performance in broilers. America J Vete Rese. 63 (5): 573-576.
Wallis IR and Balnave D. 1984. The influence of environmental temperature, age and sex on the digestibility of amino acids in growing broiler chickens. Br. Poult. Sci. 25:401-407.
Whittow GC. 1994. Regulation of body temperature. In: Sturkie’s Avian Physiology. Academic Press, San Diego, California, 344-379.
Windmueller HG and Spaeth AE. 1980. Respiratory fuels and nitrogen metabolism in vivo in small intestine of fed rats. J Biol Chem, 255: 107–112.
Wu G. 1998. Intestinal mucosal amino acid catabolism. Journal of Nutrition 128:1249-1252.
Yahav S, Straschnow A, Plavnik I and Hurwitz S. 1997. Blood system response of chickens to changes in environmental temperature. PoultSci 76: 627–633.
Yi GF, Allee GL, Frank JW, Spencer JD and Touchette KJ. 2001. Impact of glutamine, menhaden fish meal and spray-dried plasma on the growth and intestinal morphology of broilers . J Poult Sci, 80: (Supplement. 1), 201.
Zuprizal ML, Chagneau AM and Geraert PA. 1993 .Influence of ambient temperature on true digestibility of protein and amino acids of rapeseed and soybean meals in broilers. Poult. Sci., 72:289-295.
Yardibi H and Turkay G. 2008. The Effects of Vitamin E on the Antioxidant System, Egg Production, and Egg Quality in Heat Stressed Laying Hens. Turk. J. Vet. Anim. Sci, 32: 319-325.










.jpg&w=3840&q=75)