Effect of Bacillus Subtilis-based Direct-fed Microbials on Immune Status in Broiler Chickens Raised on Fresh or Used Litter
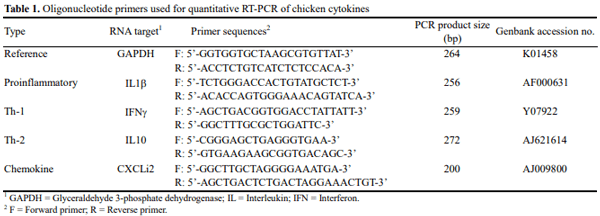
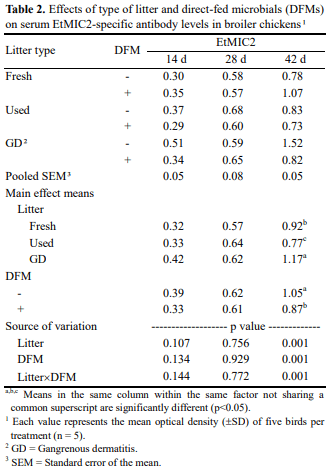
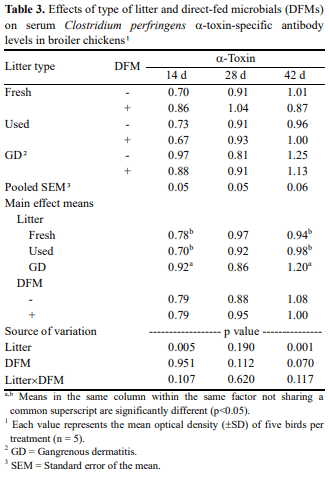
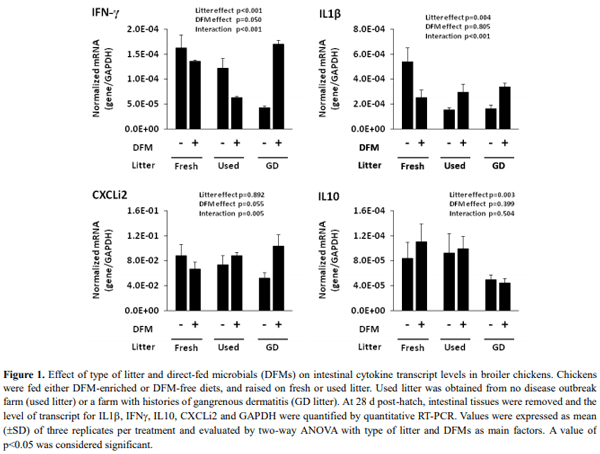
Type of dietary direct-fed microbials (DFMs) or poultry litter could directly influence the composition of gut microbiota. Gut microbiota plays an important role in shaping the developing immune system and maintaining the homeostasis of the mature immune system in mammal and chickens. The present study was carried out to investigate the interaction among litter, DFMs and immunity in broiler chickens exposed to a field-simulated environment. Immune status of broiler chickens was assessed by serum antibodies against Eimeria spp. and Clostridium spp. and intestinal cytokine mRNA expression. The current experimental design had a 3x2 factorial arrangement of treatments with three types of litter, i.e., fresh litter or used litter that was obtained from a farm with no disease outbreak (used litter) or a farm with history of a gangrenous dermatitis outbreak (GD litter), and two dietary treatments with or without DFMs. It was found that either DFM addition or type of litter significantly affected anticoccidial antibody levels of broiler chickens at d 42. In general, dietary DFMs increased the anticoccidial antibodies in the fresh-litter raised chickens, but lowered the levels in the GD-litter raised chickens. Serum antibodies against Clostridium perfringens α-toxin were significantly (p<0.05) higher in chickens raised on GD litter compared with those raised on fresh litter. Cytokine mRNA expression was significantly (p<0.05) altered by either the type of litter or DFMs. Of interest, dietary DFMs lowered interferon-γ, interleukin 1beta, and CXCLi2 cytokine mRNA expression in chickens raised on fresh litter but increased them in GD-litter raised chickens. In conclusion, dietary DFMs modulate various immune parameters of broiler chickens, but the DFM-mediated effects were dependent upon the type of litter on which chickens were raised.
(Key Words: Direct-fed Microbials, Cytokines, Broiler Chickens, Used Litter, Immunity).
Applegate, T. J., V. Klose, T. Steiner, A. Ganner, and G. Schatzmayr. 2010. Probiotics and phytogenics for poultry: Myth or reality? J. Appl. Poult. Res. 19:194-210.
Bolan, N. S., A. A. Szogi, T. Chuasavathi, B. Seshadri, M. J. Rothrock, and P. Panneerselvam. 2010. Uses and management of poultry litter. World’s Poult. Sci. J. 66:673-698.
Callaway, T. R., T. S. Edrington, R. C. Anderson, R. B. Harvey, K. J. Genovese, C. N. Kennedy, D. W. Venn, and D. J. Nisbet. 2008. Probiotics, prebiotics and competitive exclusion for prophylaxis against bacterial disease. Anim. Health Res. Rev. 9:217-225.
Chinivasagam, H. N., M. Redding, G. Runge, and P. J. Blackall. 2010. Presence and incidence of food-borne pathogens in Australian chicken litter. Br. Poult. Sci. 51:311-318.
Cressman, M. D., Z. Yu, M. C. Nelson, S. J. Moeller, M. S. Lilburn, and H. N. Zerby. 2010. Interrelations between the microbiotas in the litter and in the intestines of commercial broiler chickens. Appl. Environ. Microbiol. 76:6572-6582.
Dumas, M. D., S. W. Polson, D. Ritter, J. Ravel, J. Gelb, R. Morgan, and K. E. Wommack. 2011. Impacts of poultry house environment on poultry litter bacterial community composition. PLoS ONE 6:e24785.
FAO/WHO. 2002. Guidelines for the evaluation of probiotics in food. Joint FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food. Ontario, Canada.
Flint, J. F. and M. R. Garner. 2009. Feeding beneficial bacteria: A natural solution for increasing efficiency and decreasing pathogens in animal agriculture. J. Appl. Poult. Res. 18:367- 378.
Hong, Y. H., H. S. Lillehoj, E. P. Lillehoj, and S. H. Lee. 2006. Changes in immune-related gene expression and intestinal lymphocyte subpopulations following Eimeria maxima infection of chickens. Vet. Immunol. Immunopathol. 114:259- 272.
Lee, K. W., H. S. Lillehoj, S. I. Jang, G. Li, S. H. Lee, E. P. Lillehoj, and G. R. Siragusa. 2010a. Effect of Bacillus-based direct-fed microbials on Eimeria maxima infection in broiler chickens. Comp. Immunol. Microbiol. Infect. Dis. 33:e105- e110.
Lee, K. W., S. H. Lee, H. S. Lillehoj, G. X. Li, S. I. Jang, U. S. Babu, M. S. Park, D. K. Kim, E. P. Lillehoj, A. P. Neumann, T. G. Rehberger, and G. R. Siragusa. 2010b. Effects of direct-fed microbials on growth performance, gut morphometry, and immune characteristics in broiler chickens. Poult. Sci. 89:203- 216.
Lee, K. W., H. S. Lillehoj, and G. R. Siragusa. 2010c. Direct-fed microbials and their impact on the intestinal microflora and immune system of chickens. J. Poult. Sci. 47:106-114.
Lee, K. W., H. S. Lillehoj, S. H. Lee, S. I. Jang, G. D. Ritter, D. A. Bautista, and E. P. Lillehoj. 2011. Impact of fresh or used litter on the post-hatch immune system of commercial broilers. Avian Dis. 55:539-544.
Lee, K. W., H. S. Lillehoj, M. S. Park, S. I. Jang, G. D. Ritter, Y. H. Hong, W. Jeong, H. Y. Jeoung, D. J. An, and E. P. Lillehoj. 2012a. Clostridium perfringens ?-toxin and NetB toxin antibodies and their possible role in protection against necrotic enteritis and gangrenous dermatitis in broiler chickens. Avian Dis. 56:230-233.
Lee, K. W., H. S. Lillehoj, S. I. Jang, M. Pagès, D. A. Bautista, C. R. Pope, G. D. Ritter, E. P. Lillehoj, A. P. Neumann, and G. R. Siragusa. 2012b. Effects of in ovo vaccination and anticoccidials on the distribution of Eimeria spp. in poultry litter and serum antibody titers against coccidia in broiler chickens raised on the used litters. Res. Vet. Sci. 93:177-182.
Li, G., H. S. Lillehoj, K. W. Lee, S. I. Jang, P. Marc, C. G. Cyril, G. D. Ritter, D. A. Bautista, K. Phillips, A. P. Neumann, T. G. Rehberger, and G. R. Siragusa. 2010a. An outbreak of gangrenous dermatitis in commercial broiler chickens. Avian Pathol. 39:247-253.
Li, G., H. S. Lillehoj, K. W. Lee, S. H. Lee, M. S. Park, S. I. Jang, G. R. Bauchan, C. G. Gay, G. D. Ritter, D. A. Bautista, and G. R. Siragusa. 2010b. Immunopathology and cytokine responses in commercial broiler chickens with gangrenous dermatitis. Avian Pathol. 39:255-264.
Lillehoj, H. S., X. Ding, R. A. Dalloul, T. Sato, A. Yasuda, and E. P. Lillehoj. 2005. Embryo vaccination against Eimeria tenella and E. acervulina infections using recombinant proteins and cytokine adjuvants. J. Parasitol. 91:666-673.
Muller, P. Y., H. Janovjak, A. R. Miserez, and Z. Dobbie. 2002. Processing of gene expression data generated by quantitative real-time RT-PCR. Biotechniques 32:1372-1379.
Neumann, A. P., J. A. Benson, K. W. Lee, G. D. Ritter, D. A. Bautista, H. S. Lillehoj, and G. R. Siragusa. 2011a. Microbiological effects of Bacillus-based DFM supplementation in broilers raised on used litter from commercial farms with different disease histories. Poult. Sci. 90 (E-Suppl. 1):120-121(Abst.).
Neumann, A. P., J. A. Benson, K. W. Lee, G. D. Ritter, D. A. Bautista, H. S. Lillehoj, and G. R. Siragusa. 2011b. Intestinal microbiota of broiler chickens administered a Bacillus based DFM and raised on used litter from commercial farms with different disease histories. 2011 Congress on Gastrointestinal Function. Chicago, IL. April 18-20.
Park, S. S., H. S. Lillehoj, P. C. Allen, D. W. Park, S. Fitzcoy, D. A. Bautista, and E. P. Lillehoj. 2008. Immunopathology and cytokine responses in broiler chickens coinfected with Eimeria maxima and Clostridium perfringens with the use of an animal model of necrotic enteritis. Avian Dis. 52:14–22.
Ritter, G. 2006. Proposed pathogenesis of gangrenous dermatitis in chickens and attempts at experimental reproduction. In: Proc. from the 41st National Meeting on Poultry Health and Processing, Ocean City, MD.
Shanmugasundaram, R., M. S. Lilburn, and R. K. Selvaraj. 2012. Effect of recycled litter on immune cells in the cecal tonsils of chickens. Poult. Sci. 91:95-100.
Torok, V. A., R. J. Hughes, K. Ophel-Keller, M. Ali, and R. MacAlpine. 2009. Influence of different litter materials on cecal microbiota colonization in broiler chickens. Poult. Sci. 88:2474-2481.
Volkova, V. V., R. H. Bailey, and R. W. Wills. 2009. Salmonella in broiler litter and properties of soil at farm location. PLOS ONE 4:e6403.











.jpg&w=3840&q=75)