coccidiosis in broiler chicken
Coccidiosis in Poultry- A Review
Coccidiosis causes a great economic loss in the poultry industry due to high rate of morbidity and mortality, sub optimal growth and conversion efficiency and loss of egg production (1). Mortality is mostly marked in caecal form of the disease in young chicks whereas morbidity is well documented in the intestinal form of the disease in adult birds.
Etiology
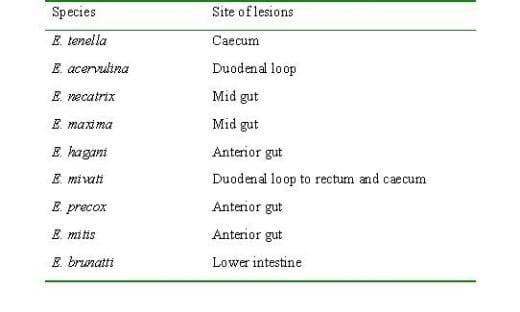
The disease is caused by a protozoan parasite known as Eimeria. The numbers of Eimeria species have been recorded from poultry and they are affecting a particular part of the intestinal tract (2). They are listed as below:
Transmission and predisposing factors
The disease is transmitted through horizontal course. Birds pick up the infection through ingestion of oocyst contaminated feeds and drinking water. Soil, houses and utensils are also the source of infection as these may have been contaminated previously by droppings of other young infected birds or by adult birds. Oocysts remain viable in litter for many months. In this way they can contaminate a farm from year to year. Oocysts are killed by freezing, extreme dryness and high temperatures. The number of oocysts eaten, strain of coccidia, environmental factors, site of development within the host, age of the bird and nutritional status of the host are act as influencing factor for the development of the coccidiosis (3&4).
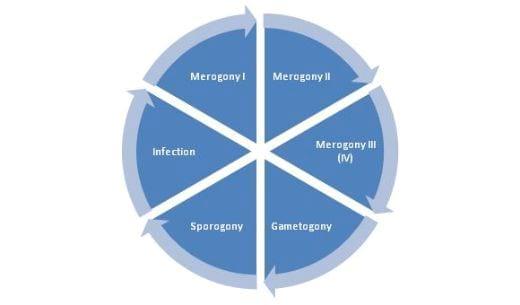
Life cycle
Stages of coccidia in chickens appear both within the host as well as outside. The oocysts are excreted in the droppings of the infected birds. Under proper conditions of temperature and moisture the oocyst undergo development to form a sporulated oocyst (Sporogony) which is capable of infecting other chickens. About 24 to 72 hours are required for the formation of sporocysts.
Clinical signs (6&7)
The disease is appeared in two forms i.e. caecal and intestinal form. Caecal form is generally observed in young chicks whereas adult birds usually suffer from intestinal form of coccidiosis.
Following clinical signs are noted in young chicks:
Depression and droopiness.
Stop feeding, huddle together, having ruffled feathers and dropped wings.
Watery diarrhoea and by the fourth day blood begins to appear in the droppings.
The greatest amount of blood appears by day five or six and by the eighth or ninth day the bird is either dead or on the way to recovery.
Severe haemorrhagic diarrhoea may produce anaemia.
Comb and wattles become pale.
Mortality is highest between the fourth and sixth days.
Birds that recover may develop a chronic illness as a result of a persistent caecal core. However, the core usually detaches itself by eight to ten days and is shed in the droppings.
Clinical signs in adult birds:
Gradual loss of appetite.
Ruffled feathers.
Progressive emaciation and birds disincline to move.
Comb and wattles become pale.
Chocolate coloured droppings are characteristic.

yellowish discoloration of its beak, comb and legs (8)
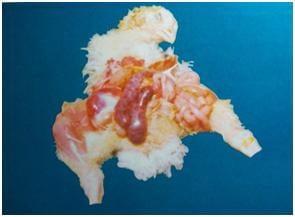
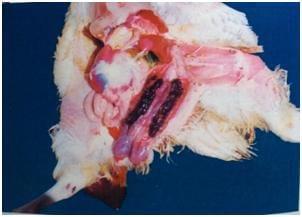
Necropsy findings (5)
The type and location of lesions in the gut indicates the species of Eimeria.
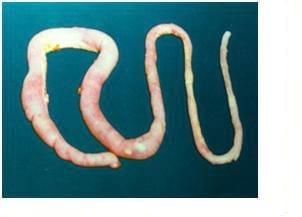
Eimeria acervulina affects the upper part of the small intestine. Affected parts of the intestine are grossly distended and small red spots and white bands may be noted on it.
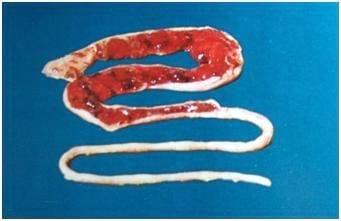
Eimeria maxima affects the entire small intestine. The intestines look watery, and in later stages have blood and mucus. The intestine may look thickened and ballooned with red pinpoint lesions.
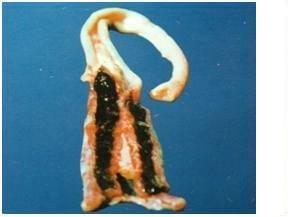
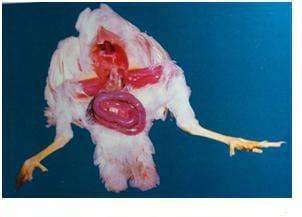
Eimeria tenella affects the blind sacs (ceca) of the gut. Haemorrhagic lesions may be noted in the wall of the caecum. Caecum may be filled with blood and pus and turn into a solid core.
 |  |
 |  |
 |  |
Diagnosis is based on the following points:
Droppings may be chocolate or blood stained.
Microscopic examination of characteristic eggs passed with droppings.
Microscopic examination of the scrapings from different sites of the intestinal mucosa and caecum.
Post-martem examination to reveal characteristic gross lesions.
Treatment and control
Management has always been important to control the coccidiosis in poultry. As coccidial oocysts are ubiquitous and easily disseminated in the poultry house environment and have such a large reproduction potential, it is very difficult to keep chickens coccidia free, especially under current intensive rearing conditions. Since species-specific immunity develops rapidly, the management focuses on reducing the number of coccidia to keep infection at a minimum until immunity is established. Hygiene, anticoccidial drugs and vaccines all play major roles.
Hygiene
It is necessary to maintain good hygiene and sanitation in the farm as disinfectants are not effective against coccidia. Following point should be considered to maintain good hygiene:
Put waterers and feeders at a height level with the backs of the birds, so they cannot defecate or scratch litter into them.
Clean the pens and remove infected droppings.
Prevent the access of infected droppings to the non-infected birds.
Keep older birds away from chicks, since old birds are carriers.
Avoid moisture and humidity in litters.
Keep the litter dry by frequent turning of litter to reduce the sporulation of the oocysts.
Avoid over-crowding in the house.
Anticoccidial drugs
The effective use of anticoccidial feed additives over the past 50 years has played a major role in the growth of the poultry industry and has allowed the increased availability of high quality, affordable poultry products to the consumer.
These anticoccidial can be classified as:

Acute infection of coccidia is generally treated with anticoccidials, but the economic losses are mainly because of subacute infection. Prophylactic use is preferred because most of the damage occurs before signs become apparent, and delayed treatment may not benefit the entire flock. Only a few of the prophylactic drugs are also effective therapeutically. Water medication is generally preferred over feed medication for treatment. Antibiotics and increased levels of vitamins A and K are sometimes used in the ration to improve rate of recovery and prevent secondary infections.
Continuous use of anticoccidial drugs may result in selection for and survival of drug-resistant strains of coccidia. While there is little cross-resistance to anticoccidials with different modes of action, there is widespread resistance to most drugs. Drug resistance is a genetic phenomenon, and once established in a line of coccidian remains for many years. To overcome this problem, shuttle programme and frequent rotation of drugs may be practised.
Shuttle programme:
The use of one anticoccidial in the starter and another in the grower ration is called a shuttle programme. The shuttle programme is meant to improve coccidiosis control, for example, intensive use of the ionophores drugs for many years has produced strains of coccidia that have reduced sensitivity for the ionophores. It is a common practice to use another drug such as nicarbazin or halofuginone in either the starter or grower ration to strengthen the programme of control against coccidiosis and to take some pressure away from the ionophores. The use of shuttle programme has been found to reduce drug resistance (9&10).
Rotation of drugs:
It is very useful to make changes in the use of anticoccidial drugs. Rotation of drugs may improve productivity. This is because the new drug will be effective against the build up of coccidia that had reduced sensitivity against the previous product that had been used for a long time.
For the poultry sector Montajat Veterinary Pharmaceuticals is providing anticoccidials for the effective control of coccidiosis in poultry to ensure optimal production. Some of the better-defined and more common agents are described briefly below.
1. Sulfonamides
Sulfonamides were the first effective anticoccidials used. Sulphonamides can be either coccidiostatic or coccidiocidal (11). The structure of sulphonamide is similar to para-aminobenzoic acid (PABA), which is required by bacteria or protozoa in the synthesis of folate (folic acid). They interfere in the early phase of folate synthesis. Sulphonamides have most activity against the asexual stages and lesser activity against the sexual stages of coccidia. The sulphonamides used for poultry coccidiosis are discussed below:
• Sulfadimidine, 16% solution: Given as 8-10 ml/liter of water for 4 days for 100 birds.
• Sulfadimethyl pyrimidine, 12.5% solution: Given as 30 ml/4 liters of water for 2 days followed by half the dose for 3 days.
• Sulfaquinoxaline, 10.32% solutions:
Caecal coccidiosis: given as 50 ml/10 liters of water in the following schedule (3:2:3 basis)
3 days medicated water
2 days plain water
3 days medicated water
Intestinal coccidiosis: given as 50 ml/15 liters of water in the following schedule
3 days medicated water
2 days plain water
3 days medicated water
2 days plain water
3 days medicated water
2. Quinolones:
Buquinolate, decoquinate and neouinate are quinolone anticoccidials. They act on the sporozoite stage and inhibit coccidial respiration by disrupting electron transport in the mitochondrial system. Their main use is in shuttle programs and withdrawal feeds. Quinolines have low toxicity and broad-spectrum activity. Rapid emergence of resistance to these compounds has been their major weakness. Decoquinate is fed at 0.003% for prevention of coccidiosis in broilers. It should be fed for at least 28 days when development of coccidiosis is likely.
3. Clopidol:
It is active against the sporozoite stage, allowing host cell penetration but not parasitic development. It also has activity against second generation schizogony, gametogony and sporulation (12). Sporozoites can resume development after the medication is removed. It is given at 0.0125-0.0250% for prevention of coccidiosis. A 5 days withdrawal is required if the 0.0250% level is used. It has a broad species spectrum, but development of resistance is a problem.
4. Robenidine:
It is a guanidine derivative and active against the first generation schizont of coccidia by preventing formation of merozoites. It is both coccidiostatic and coccidiocidal, but drug resistance has been a problem. It is fed at 33 ppm for the prevention of coccidiosis. A 5-day withdrawal period is needed to avoid altering flavour of the poultry meat.
5. Amprolium (MONCOXTM oral liquid, 25%)
Amprolium is a vitamin B1 or thiamine structure analogue. It acts on the first-generation schizont to prevent merozoite production and has some activity against sexual stages and the sporulation oocyst. It is often combined with other agents to increase activity. It competitively inhibits the active transport of thiamine in parasites. It is used for the treatment and control of caecal and intestinal coccidiosis in poultry.
Dosage and administration
Curative: 1 ml/liter of drinking water, once daily for 5-7 days.
Prophylaxis: Use half the treatment dose for 5 days.
6. Nitrobenzamides:
Dinitolamide is a nitrobenzamide anticoccidial agent. It acts primarily on the first generation schizont and inhibit sporulation of oocysts (13). It is coccidiostatic if given for 6 days but is coccidiocidal if given for longer periods (12). It is fed at 0.0125% to prevent coccidiosis in chickens.
7. Nicarbazin:
It was the first product to have truly broad-spectrum activity and is still in common use. While not completely understood, the mode of action is thought to be via inhibition of succinate-linked nadide reduction and the energy-dependent trans-hydrogenase, and the accumulation of calcium in the presence of ATP. Nicarbazin is fed at 125 ppm for the prevention of coccidiosis. Nicarbazin must not be used in layers, and a 4-day withdrawal period is required in broilers. Nicarbazin is usually restricted in use to the starting period because of potential growth suppressing effect and to cooler months of the year because of its potential to enhance the effects of heat distress (14).
8. Halofuginone:
It is an alkaloid originally isolated from the plant Dichroa febrifuga. It is active against the asexual stages of coccidia. It has both coccidiostatic and coccidiocidal effects. Halofuginone is fed at 0.03% (3 ppm) for the prevention of coccidiosis. Do not feed to layers and a 4 day withdrawal period is required.
9. Toltrazuril (COXURILTM oral liquid, 2.5%)
It is a triazinone antiprotozoal drug and is used as a coccidiocidal for the prevention and treatment of coccidiosis in poultry. It is active against both asexual and sexual stages of coccidia by inhibiting nuclear division of schizonts and microgamonts and the well forming bodies of the macrogamonts.
Dosage and administration:
Curative: 7 mg/kg bwt per day; 1 ml/L of drinking water continuously over 48 hours, or at 3ml/L given for 8 hours per day, on 2 consecutive days. Repeat one medication after 5 days.
Prophylaxis: Broilers: Three medications, on day 9 to 10, on day 16 to 17 and on day 23 to 24 of life.
10. Diclazuril (DICLACOXTM)
It is a triazinone antiprotozoal drug that is indicated for the control of caecal and intestinal Coccidiosis.
Dosage and administration
200 grams/ ton of feed (0.0001%), chickens are fed from day 1 to slaughter age.
Ionophores
Polyether ionophores form complexes with various ions, principally sodium, potassium and calcium and transport these into and through biological membrane and disrupting osmotic balance. The ionophores affect both extra- and intracellular stages of the parasite. Their principal activity is exerted during the early, asexual stages of parasite development. Some of these are mentioned below:
11. Monensin (MONENCOXTM)
It is monovalent polyether ionophore that is a fermentation product of Streptomyces cinnamonensis. It is given for the prevention of coccidiosis in broilers. Do not feed to layers or chickens over 16 weeks of age. No withdrawal is required.
Dosage and administration
500 to 600 gm per ton of feed (100 to 120 ppm)
12. Lasalocid:
lasalocid is a divalent polyether ionophore that is a fermentation product of Streptomyces lasaliensis. It is fed at 0.0075-0.0125% (75-125 ppm) for the prevention of coccidiosis.
13. Salinomycin (SALINOCOXTM)
It is monovalent polyether ionophore that is a fermentation product of Streptomyces albus. It is active against sporozoites and early and late asexual stages of chicken coccidia (Conway et al, 1993). It is given for the prevention of coccidiosis in broilers. Do not feed to layers. No withdrawal is required.
Dosage and administration
500 gm per ton of finished feed
14. Maduramicin (MADURAJATTM)
Maduramicin is a monovalent monoglycoside polyether ionophores that is a fermentation product of Actinomadura yumaense. It is used for the prevention of coccidiosis in broilers and fed at 5 ppm. A 5 day withdrawal period is required.
Dosage and administration
500 gm per ton of feed, equivalent to 5 ppm in the final feed
Vaccination
Live, virulent vaccines have been utilized for the last 50 years, and live attenuated vaccines have become available over the last two decades. The attenuated strains have been selected for rapid passage through the host. Consequently, they have low reproductive potential and have lost their virulence, but still have strong immunogenicity. Importantly, they cause no post-vaccinal decrease in weight gain, and are therefore suitable for use in broiler flocks (15, 16&17).
References:
1. Williams, R. B. (2005) Intercurrent coccidiosis and necrotic enteritis of chickens: Rational, integrated disease management by maintenance of gut integrity. Avian Pathol. 34:159-180.
2. McDougald, L. R. (1998) Intestinal protozoa important to poultry. Poult. Sci. 77:1156-1158.
3. Narciri, M., Yvore, P. And Conan, L. (1983) Influence of contamination of environment and breeding condition of development of coccidian in chicken. Annals Research Veterinaire. 13:117-121.
4. McDougald, L. R. (2003). Coccidiosis. Pages 974-991 in Diseases of Poultry. Y. M. Saif, ed. Iowa State University Press, Ames.
5. Vegad, J. L. (2004) Poultry diseases. A guide for farmers and poultry professionals. 1st edition, IBDC, India.
6. Pellergly, L.P. (1974) Coccidia and Coccidiosis. IInd Ed. Verlag Paul Purey Berlin and Hamburg.
7. Soulsby, E.J. (1982) Helminths, Arthropods and Protozoa of domesticated animals. Seventh Edition. pp 594-605.
8. Vegad, J. L. (2007) A colour atlas of Poultry diseases. An aid to farmers and poultry professionals. 1st edition, IBDC, India.
9. Williams, R. B. (1998) Epidemiological aspects of the use of live anticoccidial vaccines for chickens. Int. J. Parasitol. 28:1089-1098.
10. Reid, W. M. (1975) Progress in the control of coccidiosis with anticoccidials and planned immunization. Am. J. Vet. Res. 36:593-596.
11. Mathis, G. F. and McDougald, L. R. (1984) Effectiveness of therapeutic anticoccidials drug against recently isolated coccidia. Poult. Sci. 63:1149-1153.
12. Long, P. L. (1993) Avian coccidiosis. In J.P. Kreier, ed., Parasitic protozoa, vol 4, 2nd ed. pp 1-88 San Diego: Academic press.
13. Mathis, G. F. and McDougald, L. R. (1981) Experimental development of resistance to amprolium or dinitolamide in Eimeria acervulina and its effect on inhibition of sporulation of oocysts. J. Parasitol. 67:956-957.
14. McDougald, L. R. (1993) Chemotherapy of coccidiosis. Proc 6th Int. coccid. conf. Guelph, Canada, pp. 45-47.
15. Shirley, M. W., A. C. Bushell, J. E. Bushell, V. McDonald, and B. Roberts. (1995) A live attenuated vaccine for control of avian coccidiosis: trials in broiler breeders and replacement layer flocks in the United Kingdom. Vet. Rec. 137:453-457.
16. Chapman, D. (2000) Practical use of vaccines for the control of coccidiosis in the chicken. World's Poultry Science Journal. 56:7-12.
17. Chapman, D. (2002) Sustainable coccidiosis control in poultry production: The role of live vaccines. Int. J. Parasitol. 32:617- 629.

I am a doctor in Pakistan related to poultry feed and I mostly check poultry farms. I have been facing coccidiosis problem from 4 to 5 months in the field. There is mostly litter dry and climate in pakistan is mostly dry. But my poultry farmers face the coccidiosis problem in three or four times during 42 days of flock. This causes great economic losses to them. Mostly I use sulfaquinoxaline or toltrazuril salt in water and clopidol in feed to control and treat coccidiosis but sometimes coccidiosis attack is sever and no medicine works. I am working as technical advisor in poultry farms from last 7 years but didnot see so much coccidiosis in my 7 years. I am facing very difficulty even in hot environment and dry littler conditions to control coccidiosis.
Can you give me your valuable suggestions?
THank u so much for this informative article about coccidiosis in poultry. i have experienced that shuttle programme is highly effective to control the coccidiosis.
Thank u doctors for the nice piece of informative article about coccidiosis in poultry .Have u got any idea of HOMEOPATHIC medicine which can act like a anti-coccidial medicine ? If U know pls ,can furnish it , I feel the homeo will be more affordable and easily available to the poor farmers at village condition .

Dear Mr Khalil,
Weve successfully managed flocks by feeding only ionophores particularly salinomycin, sacox. Weve done this experiment on flocks of 55000 broilers each consistently for 6 batches, may change type of ionophore used for next 2-3 flocks to shift back again on Sacox.
















.jpg&w=3840&q=75)