A Review of Prevention and Control Methods of Salmonella species in Swine Production the Role of Dietary Non-Nutritional Additives
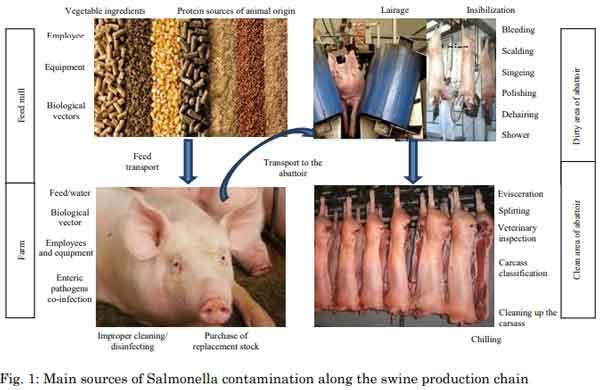
The control of Salmonella spp. is still an important issue in pork production. Contamination happens at any stage of the production chain and no stand-alone measure is efficient enough to eliminate this pathogen. In order to minimize Salmonella contamination, the farm-to-fork approach is currently used, in which specific measures are implemented in each sector of the production chain. The already known hygiene measures in the abattoir are important to reduce the risk of carcass contamination; however, pre-slaughter conditions have been shown to be high risk factors at slaughter and during processing. Salmonella spread at slaughter can be traced back to the pig herd and therefore, there has been increasing focus on the pre-harvest phase. Numerous studies have identified risk factor for Salmonella infection in pigs and reviews studies have presented interesting and important comparative analyses on this subject. The use of dietary additives for pig and their effects on Salmonella infection have been studied during the last years. Particularly interesting is the use of dietary non-nutritional additives, such as prebiotics, probiotics, phytogenics and essential oils and organic acids. Although, very promising, much research is needed in this field in order to identify the most efficient products and/or compounds and reveal how they act on the bacterium metabolism, aiming to improve the control of Salmonella contamination along the swine production chain. In this review, we surveyed the literature to present a compilation of the scientific knowledge currently available about potential uses of dietary non-nutritional additives to prevent and/or control Salmonella infection in swine populations.
Key words: On-farm control, dietary non-nutritional additives, pig, Salmonella, slaughter.
Agarwal, S., M.A. Reynolds, S. Pou, D.E. Peterson, J.A. Charon and J.B. Suzuki, 1991. The effect of sanguinarine on human peripheral blood neutrophil viability and functions. Oral Microbiol. Immunol., 6: 51-61.
Altenhoefer, A., S. Oswald, U. Sonnenborn, C. Enders, J. Schulze, J. Hacker and T.A. Oelschlaeger, 2004. The probiotic Escherichia coli strain Nissle 1917 interferes with invasion of human intestinal epithelial cells by different enteroinvasive bacterial pathogens. FEMS Immunol. Med. Microbiol., 40: 223-229.
Althouse, C., S. Patterson, P. Fedorka-Cray and R.E. Isaacson, 2003. Type 1 fimbriae of Salmonella enterica serovar Typhimurium bind to enterocytes and contribute to colonization of swine in vivo. Infect. Immunity, 71: 6446-6452.
Anonymous, 2009. Annual report on zoonoses in Denmark 2008. National Food Institute, Technical University of Denmark, Soborg, Denmark.
Aperce, C.C., T.E. Burkey, B. KuKanich, B.A. Crozier-Dodson, S.S. Dritz and J.E. Minton, 2010. Interaction of Bacillus species and Salmonella enterica serovar Typhimurium in immune or inflammatory signaling from swine intestinal epithelial cells. J. Anim. Sci., 88: 1649-1656.
Arguello, H., P. Rubio, A. Jaramillo, V. Barrios, M. Garcia and A. Carvajal, 2011. Evaluation of cleaning and disinfection procedures against Salmonella enterica at swine farms, transport and lairage facilities. Proceedings of the 9th International Conference on the Epidemiology and Control of Biological, Chemical and Physical Hazards in Pigs and Pork, June 19-22, 2011, Maastricht, The Netherlands, pp: 254-257.
Arguello, H., A. Alvarez-Ordonez, A. Carvajal, P. Rubio and M. Prieto, 2013a. Role of slaughtering in Salmonella spreading and control in pork production. J. Food Prot., 76: 899-911.
Arguello, H., A. Carvajal, S. Costillas and P. Rubio, 2013b. Effect of the addition of organic acids in drinking water or feed during part of the finishing period on the prevalence of Salmonella in finishing pigs. Foodborne Pathogens Dis., 10: 842-849.
Badia, R., M.T. Brufau, A.M. Guerrero-Zamora, R. Lizardo and I. Dobrescu et al., 2012. β-Galactomannan and Saccharomyces cerevisiae var. boulardii modulate the immune response against Salmonella enterica serovar Typhimurium in porcine intestinal epithelial and dendritic cells. Clin. Vaccine Immunol., 19: 368-376.
Badia, R., R. Lizardo, P. Martinez and J. Brufau, 2013. Oligosaccharide structure determines prebiotic role of β-galactomannan against Salmonella enterica ser. Typhimurium in vitro. Gut Microbes, 4: 72-75.
Bahnson, P.B., P.J. Fedorka-Cray, S.R. Ladely and N.E. Mateus-Pinilla, 2006. Herd-level risk factors for Salmonella enterica sub sp. enterica in U.S. market pigs. Prev. Vet. Med., 76: 249-262.
Bailey, M., 2009. The mucosal immune system: Recent developments and future directions in the pig. Dev. Comp. Immunol., 33: 375-383.
Baptista, F.M., J. Dahl and L.R. Nielsen, 2010. Factors influencing Salmonella carcass prevalence in Danish pig abattoirs. Prevent. Vet. Med., 95: 231-238.
Bayoumi, M.A. and M.W. Griffiths, 2010. Probiotics down-regulate genes in Salmonella enterica serovar Typhimurium pathogenicity islands 1 and 2. J. Food Protect., 73: 452-460.
Bearson, B.L., L. Wilson and J.W. Foster, 1998. A low pH-inducible, PhoPQ-dependent acid tolerance response protects Salmonella typhimurium against inorganic acid stress. J. Bacteriol., 180: 2409-2417.
Bello, F.D., J. Walter, C. Hertel and W.P. Hammes, 2001. In vitro study of prebiotic properties of levan-type exopolysaccharides from lactobacilli and non-digestible carbohydrates using denaturing gradient gel electrophoresis. Syst. Applied Microbiol., 24: 232-237.
Beloeil, P.A., P. Fravalo, C. Fablet, J.P. Jolly and E. Eveno et al., 2004. Risk factors for Salmonella enterica subsp. enterica shedding by market-age pigs in French farrow-to-finish herds. Prev. Vet. Med., 63: 103-120.
Beloeil, P.A., C. Chauvin, K. Proux, C. Fablet, F. Madec and A. Alioum, 2007. Risk factors for Salmonella seroconversion of fattening pigs in farrow-to-finish herds. Vet. Res., 38: 835-848. Berchieri, Jr. A., 2000. Salmoneloses aviarias. In: Doencas das Aves, Berchieri, Jr. A. and M. Macari (Eds.). Fundacao APINCO de Ciencia e Tecnologia Avicolas (FACTA), Campinas, Brazil, pp: 185-196.
Berends, B.R., F. Van Knapen, J.M. Snijders and D.A. Mossel, 1997. Identification and quantification of risk factors regarding Salmonella spp. on pork carcasses. Int. J. Food Microbiol., 20: 199-206.
Bertrand, S., K. Dierick, K. Heylen, T. De Baere and B. Pochetet al. 2010. Lessons learned from the management of a national outbreak of Salmonella ohio linked to pork meat processing and distribution. J. Food Protect., 73: 529-534.
Blanchard, P. and K. Kjeldsen, 2003. Time to hit Salmonella in pigs. Pig J., 52: 182-194.
Blank, R., B. Muller-Siegwardt and S. Wolffram, 2010. Sanguinarine does not influence availability or metabolism of tryptophan in pigs. Livestock Sci., 134: 24-26.
Borch, E., T. Nesbakken and H. Christensen, 1996. Hazard identification in swine slaughter with respect to foodborne bacteria. Int. J. Food Microbiol., 30: 9-25.
Boughton, C., J. Egan, G. Kelly, B. Markey and N. Leonard, 2007. Rapid infection of pigs following exposure to environments contaminated with different levels of Salmonella typhimurium. Foodborne Pathog. Dis., 4: 33-40.
Boyen, F., F. Haesebrouck, D. Maes, F. Van Immerseel, R. Ducatelle and F. Pasmans, 2008. Non-typhoidal Salmonella infections in pigs: A closer look at epidemiology, pathogenesis and control. Vet. Microbiol., 130: 1-19.
Brufau, J., 2003. Animal Feeding in Europe: Challenges and Opportunities. In: Nutritional Biotechnology in the Feed and Food Industries, Lyons, T.P. and K.A. Jacques (Eds.). Nottingham University Press, Nottingham, UK.
Brustolin, J.C. A. Dal Pisol, J. Steffens, G. Toniazzo, E. Valduga, M. Di Luccio and R.L. Cansian, 2014. Decontamination of pig carcasses using water pressure and lactic acid. Braz. Arch. Biol. Technol, 57: 954-961.
Burt, S., 2004. Essential oils: Their antibacterial properties and potential applications in foods: A review. Int. J. Food Microbiol., 94: 223-253.
Bysted, D., 2003. Effect of feeding strategy on Salmonella in Danish sows and weaners. Proceedings of the 5th International Symposium on the Epidemiology and Control of Foodborne Pathogens in Pork, October 1-4, 2003, Heraklion, Crete, Greece, pp: 138.
CDC., 2014. Outbreak of salmonellosis associated with consumption of pulled pork at a church festival-Hamilton County, Ohio, 2010. Morbidity Mortality Weekly Rep., 62: 1045-1047.
Callaway, T.R., J.A. Carroll, J.D. Arthington, C. Pratt and T.S. Edrington et al., 2008. Citrus products decrease growth of E. coli O157: H7 and Salmonella typhimurium in pure culture and in fermentation with mixed ruminal microorganisms in vitro. Foodborne. Pathog. Dis., 5: 621-627.
Carrique-Mas, J.J., S. Bedford and R.H. Davies, 2007. Organic acid and formaldehyde treatment of animal feeds to control Salmonella: Efficacy and masking during culture. J. Applied Microbiol., 1013: 88-96.
Casey, P.G., G.D. Casey, G.E. Gardiner, M. Tangney and C. Stanton et al., 2004. Isolation and characterization of anti-Salmonella lactic acid bacteria from the porcine gastrointestinal tract. Lett. Applied Microbiol., 39: 431-438.
Chang, C.H., Y.S. Chen, M.T. Chiou and C.H. Su et al., 2013. Application of Scutellariae radix, Gardeniaefructus and probiotics to prevent Salmonella enterica serovar Choleraesuis infection in swine. Evidence Based Complement. Altern. Med. 10.1155/2013/568528
Charles, S.D., A.S. Abraham, E.T. Trigo, G.F. Jones and T.L. Settje, 2000. Reduced shedding and clinical signs of Salmonella Typhimurium in nursery pigs vaccinated with a Salmonella Choleraesuis vaccine. J. Swine Health Prod., 8: 107-112.
Chaturvedi, M.M., A. Kumar, B.G. Darnay, G.B.N. Chainy, S. Agarwal and B.B. Aggarwal, 1997. Sanguinarine (Pseudochelerythrine) is a potent inhibitor of NF-κB activation, IκBα phosphorylation and degradation. J. Biol. Chem., 272: 30129-30134.
Christensen, J. and M. Rudemo, 1998. Multiple change-point analysis applied to the monitoring of Salmonella prevalence in Danish pigs and pork. Prev. Vet. Med., 36: 131-143.
Colombo, M.L. and E. Bosisio, 1996. Pharmacological activities of Chelidonium majus L. (Papaveraceae). Pharmacol. Res., 33: 127-134.
Cosentino, S., C.I.G. Tuberoso, B. Pisano, M. Satta, V. Mascia, E. Arzedi and F. Palmas, 1999. In-vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Applied Microbiol., 29: 130-135.
Creus, E., J.F. Perez, B. Peralta, F. Baucells and E. Mateu, 2007. Effect of acidified feed on the prevalence of Salmonella in market-age pigs. Zoonoses Public Health, 54: 314-319.
Crump, J.A., P.M. Griffin and F.J. Angulo, 2002. Bacterial contamination of animal feed and its relationship to human foodborne illness. Clin. Infect. Dis., 35: 859-865.
Da Silva, L.E., V. Dias, A. Ferronatto, P. Guerra and L. Berno et al., 2012. Longitudinal dissemination of Salmonella enterica clonal groups through the slaughter process of Salmonella-positive pig batches. J. Food Prot., 75: 1580-1588.
Davies, P.R., W.E. Morrow, F.T. Jones, J. Deen, P.J Fedorka-Cray and I.T. Harris, 1997a. Prevalence of Salmonella in finishing swine raised in different production systems in North Carolina, USA. Epidemiol. Infect., 119: 237-244.
Davies, P.R., W.E. Morrow, F.T. Jones, J. Deen, P.J Fedorka-Cray and J.T. Gray 1997b. Risk of shedding Salmonella organisms by market-age hogs in a barn with open-flush gutters. J. Am. Vet. Med. Assoc., 210: 386-389.
Davies, P.R., H.S. Hurd, J.A. Funk, P.J. Fedorka-Cray and F.T. Jones, 2004. The role of contaminated feed in the epidemiology and control of Salmonella enterica in pork production. Foodborne Pathog. Dis., 1: 202-215.
Davies, R.H. and C. Wray, 1995. Observations on disinfection regimens used on Salmonella enteritidis infected poultry units. Poult. Sci., 74: 638-647.
Davies, R.H. and C. Wray, 1996. Studies of contamination of three broiler breeder houses with Salmonella enteritidis before and after cleansing and disinfection. Avian Dis., 40: 626-633.
Davies, R.H. and M.H. Hilton, 2000. Salmonella in Animal Feed. In: Salmonella in Domestic Animals. Wray, C. and A. Wray (Eds.)., CABI Publishing, Wallingford, Oxfordshire, England. Davies, R.H. and A. Cook, 2008. Why has the UK pig industry more Salmonella than other European pig industries and how can we lift our position? Feed Compd., 28: 33-35.
De Busser, E.V., J. Dewulf, N. Nollet, K. Houf and K. Schwarzer et al., 2009. Effect of organic acids in drinking water during the last 2 weeks prior to slaughter on Salmonella shedding by slaughter pigs and contamination of carcasses. Zoonoses Public Health, 56: 129-136.
De Busser, E.V., D. Maes, K. Houf, J. Dewulf, H. Imberechts, S. Bertrand and L. De Zutter, 2011. Detection and characterization of Salmonella in lairage, on pig carcasses and intestines in five slaughterhouses. Int. J. Food Microbiol., 145: 279-286.
De Busser, E.V., L. Zutter, J. Dewulf, K. Houf and D. Maes, 2013. Salmonella control in live pigs and at slaughter. Vet. J., 196: 20-27.
De Ridder, L., D. Maes, J. Dewulf, F. Pasmans and F. Boyen et al., 2013. Evaluation of three intervention strategies to reduce the transmission of Salmonella Typhimurium in pigs. Vet. J., 197: 613-618.
Delhalle, L., L. De Sadeleer, K. Bollaerts, F. Farnir and C. Saegerman et al., 2008. Risk factors for Salmonella and hygiene indicators in the 10 largest Belgian pig slaughterhouses. J. Food Protec., 71: 1320-1329.
Denagamage, T., A.M. O'Connor, J. Sargeant, A. Rajic and J.D. McKean, 2007. Vaccination against Salmonella and the association with measures of Salmonella prevalence in live and slaughtered swine-A systematic review. Proceedings of the 7th International Symposium on the Epidemiology and Control of Foodborne Pathogens in Pork, May 9-11, 2007, Verona, Italy, pp: 283-286.
Dougan, G., V. John, S. Palmer and P. Mastroeni, 2011. Immunity to salmonellosis. Immunol. Rev., 240: 196-210.
EFSA., 2008. Scientific opinion of the panel on biological hazards on a request from the health and consumer protection, directorate general, European commission on microbiological risk assessment in feeding stuffs for food producing animals. EFSA J., Vol. 720.
EFSA., 2010a. Scientific opinion on a quantitative microbiological risk assessment of Salmonella in slaughter and breeder pigs. EFSA J., Vol. 8.
EFSA., 2010b. The community summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in the European Union in 2008. EFSA J., Vol. 8.
EFSA., 2011. Analysis of the baseline survey on the prevalence of Salmonella in holdings with breeding pigs, in the EU, 2008-Part B: Factors associated with Salmonella pen positivity. EFSA J., Vol. 9.
EFSA., 2012. The European Union summary report on trends and sources of zoonoses, zoonotic agents and foodborne outbreaks in 2010. EFSA J., Vol. 10. 10.2903/j.efsa.2012.2597
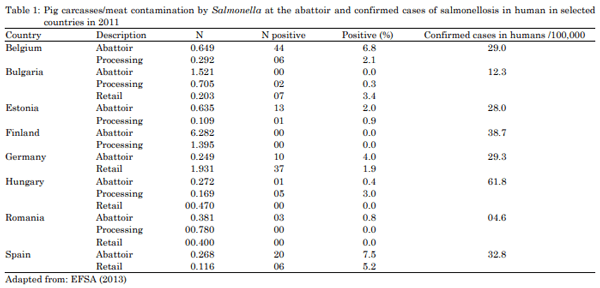
EFSA., 2013. The European union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2011. EFSA J., Vol. 11.
Edel, W., P.A.M. Guinee, M. van Schothorst and E.H. Kampelmacher, 1967. Salmonella infections in pigs fattened with pellets and unpelleted meal. Zentralblatt Veterinarmedizin Reihe B, 14: 393-401.
Ekperigin, H.E. and K.V. Nagaraja, 1998. Microbial food borne pathogens. Salmonella. Vet.
Clin. North Am. Food Anim. Pract., 14: 17-29.
European Commission, 2011. A strategic vision for European standards: Moving forward to enhance and accelerate the sustainable growth of the European economy by 2020. http://eur- lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52011DC0311.
Farzan, A., R.M. Friendship, C.E. Dewey, K. Warriner, C. Poppe and K. Klotins, 2006. Prevalence of Salmonella spp. on Canadian pig farms using liquid or dry-feeding. Prev. Vet. Med., 73: 241-254.
Farzan, A. and R.M. Friendship, 2010. A clinical field trial to evaluate the efficacy of vaccination in controlling Salmonella infection and the association of Salmonella-shedding and weight gain in pigs. Can. J. Vet. Res., 74: 258-263.
Farzan, A., R.M. Friendship, C.E. Dewey, C. Poppe and J. Funk, 2010. Evaluation of the risk factors for shedding Salmonella with or without antimicrobial resistance in swine using multinomial regression method. Zoonoses Public Health, 57: 85-93.
Fosse, J., H. Seegers and C. Magras, 2009. Prevalence and risk factors for bacterial food-borne zoonotic hazards in slaughter pigs: A review. Zoonoses Public Health, 56: 429-454.
Franz, C., R. Bauer, R. Carle, D. Tedesco, A. Tubaro and K. Zitterl-Eglseer, 2005. Study on the assessments of plants/herbs, plant/herb extracts and their naturally or synthetically produced components as additives for use in animal production. CFT/EFSA/FEEDAP., pp: 5-169. http://www.efsa.europa.eu/sites/default/files/scientific_output/files/main_documents/070828.pdf Freitas, J., 2011. Evolucion epidemiologica de serovares prevalentes en la cadena productiva de aves e modelo de banco de cepas brasileiro. Proceedings of the Brasil-Seminario Internacional sobre Salmonellosis Aviar, June 28-30, 2011, Rio de Janeiro, Brasil.
Friendship, R.M., A. Mounchili, S. McEwen and A. Rajic, 2009. Critical review of on-farm intervention strat e g i e s ag ai n s t Salmonella . BPEX/ZNCP, Ontario. http://development.bpex.org.uk/downloads/298614/292327/Critical%20review%20of%20onfar m%20intervention%20strategies%20against%20Salmonella.pdf.
Funk, J.A., P.R. Davies and W. Gebreyes, 2001. Risk factors associated with Salmonella enterica prevalence in three-site swine production systems in North Carolina, USA. Berl Munch Tierarztl Wochenschr, 114: 335-338.
Funk, J. and W.A. Gebreyes, 2004. Risk factors associated with Salmonella prevalence on swine farms. J. Swine Health Prod., 12: 246-251.
Funk, J.A., T.E. Wittum, J.T. LeJeune, P.J. Rajala-Schultz, A. Bowman and A. Mack, 2007. Evaluation of stocking density and subtherapeutic chlortetracycline on Salmonella enterica subsp. enterica shedding in growing swine. Vet. Microbiol., 124: 202-208.
Gaggia, F., P. Mattarelli and B. Biavati, 2010. Probiotics and prebiotics in animal feeding for safe food production. Int. J. Food. Microbiol., 141: S15-S28.
Garcia-Feliz, C., A. Carvajal, J.A. Collazos and P. Rubio, 2009. Herd-level risk factors for faecal shedding of Salmonella enterica in Spanish fattening pigs. Prev. Vet. Med., 91: 130-136.
Gedek, B.R., 1999. Adherence of Escherichia coli serogroup 0 157 and the Salmonella typhimurium
mutant DT 104 to the surface of Saccharomyces boulardii. Mycoses, 42: 261-264.
Gibert, E.C. and R.C.M. Jaime, 2010. Salmonelosis en explotaciones porcinas, 2a parte: Dinamica de la transmision en las explotaciones porcina. Suis, 68: 40-48.
Gibson, G.R. and M.B. Roberfroid, 1995. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr., 125: 1401-1412.
Godsey, B.H., K.A. Skjolaas and J.E. Minton, 2007. Pre-exposure to Bacillus licheniformis reduces interleukin 8 response of swine intestinal epithelial cells to Salmonella enterica serovar Typhimurium. Proceedings of the ADSA, PSA, AMPA and ASAS Joint Meeting, July 8-12, 2007, Midwest American Society of Animal Science, San Antonio, TX., USA., pp: 108.
Goldbach, S.G. and L. Alban, 2006. A cost-benefit analysis of Salmonella-control strategies in Danish pork production. Prev. Vet. Med., 77: 1-14.
Gourbeyre, P., S. Denery and M. Bodinier, 2011. Probiotics, prebiotics and synbiotics: Impact on the gut immune system and allergic reactions. J. Leukocyte Biol., 89: 685-695.
Greig, J.D. and A. Ravel, 2009. Analysis of foodborne outbreak data reported internationally for source attribution. Int. J. Food Microbiol., 130: 77-87.
Grilli, E., M.R. Messina, A. Tedeschi and A. Piva, 2010. Feeding a microencapsulated blend of organic acids and nature identical compounds to weaning pigs improved growth performance and intestinal metabolism. Livestock Sci., 133: 173-175.
Grimont, P.A.D. and F.X. Weill, 2007. Antigenic Formulae of the Salmonella serovars. 9th Edn., WHO Collaborating Centre for Reference and Research on Salmonella. Institute Pasteur, Paris, France.
Guibourdenche, M., P. Roggentin, M. Mikoleit, P.I. Fields, J. Bockemuhl, P.A.D. Grimont and F.X. Weill, 2010. Supplement 2003-2007 (No. 47) to the white-kauffmann-le minor scheme. Res. Microbiol., 161: 26-29.
Hafez, M., K. Hayes, M. Goldrick, G. Warhurst, R. Grencis and I.S. Roberts, 2009. The K5 capsule of Escherichia coli strain Nissle 1917 is important in mediating interactions with intestinal epithelial cells and chemokine induction. Infect. Immunity, 77: 2995-3003.
Hald, T., A. Wingstrand, M. Swanenburg, A. von Altrock and B.M. Thorberg, 2003. The occurrence and epidemiology of Salmonella in European pig slaughterhouses. Epidemiol. Infect., 131: 1187-1203.
Hamilton, D., G. Holds, M. Lorimer, A. Kiermeier, C. Kidd, J. Slade and A. Pointon, 2010. Slaughterfloor decontamination of pork carcases with hot water or acidified sodium chlorite- A comparison in two Australian abattoirs. Zoonoses Public Health, 57: 16-22.
Hammer, K.A., C.F. Carson and T.V. Riley, 1999. Antimicrobial activity of essential oils and other plant extracts. J. Applied Microbiol., 86: 985-990.
Hautekiet, V., V. Geert, V. Marc and G. Rony, 2008. Development of a sanitary risk index for Salmonella seroprevalence in Belgian pig farms. Prev. Vet. Med., 86: 75-92.
Henao, O.L., E. Scallan, B. Mahon and R.M. Hoekstra, 2010. Methods for monitoring trends in the incidence of foodborne diseases: Foodborne diseases active surveillance network 1996-2008. Foodborne Pathog. Dis., 7: 1421-1426.
Himathongkham, S., M.D.G. Pereira and H. Riemann, 1996. Heat destruction of Salmonella in poultry feed: Effect of time, temperature and moisture. Avian Dis., 40: 72-77.
Hirsh, D.C., 2003. Salmonella. In: Microbiologia Veterinaria, Hirsh, D.C. and Y.C. Zee (Eds.).
Guanabara-Koogan, Rio de Janeiro, ISBN: 9788527707848, pp: 69-73.
Hotes, S., N. Kemper, I. Traulsen, G. Rave and J. Krieter, 2010. Risk factors for Salmonella infection in fattening pigs-an evaluation of blood and meat juice samples. Zoonoses Public Health, 57: 30-38.
Hotes, S., I. Traulsen and J. Krieter, 2011. Salmonella control measures with special focus on vaccination and logistic slaughter procedures. Transbound. Emerg. Dis., 58: 434-444.
Hurd, H.S., J.D. McKean, I.V. Wesley and L.A. Karriker, 2001. The effect of lairage on Salmonella isolation from market swine. J. Food Protect., 64: 939-944.
Hurd, H.S., J.D. McKean, R.W. Griffith, I.V. Wesley and M.H. Rostagno, 2002. Salmonella enterica infections in market swine with and without transport and holding. Applied Environ. Microbiol., 68: 2376-2381.
Jones, F.T. and K.E. Richardson, 2004. Salmonella in commercially manufactured feeds. Poult. Sci., 83: 384-391.
Jones, F.T., 2011. A review of practical Salmonella control measures in animal feed. J. Applied Poul. Res., 20: 102-113.
Kantas, D., V.G. Papatsiros, P.D. Tassis, L.V. Athanasiou and E.D. Tzika, 2015. The effect of a natural feed additive (Macleaya cordata), containing sanguinarine, on the performance and health status of weaning pigs. Anim. Sci. J., 86: 92-98.
Kasbohrer, A., D. Protz, R. Helmuth, K. Nockler, T. Blaha, F.J. Conraths and L. Geue, 2000. Salmonella in slaughter pigs of German origin: An epidemiological study. Eur. J. Epidemiol., 16: 141-146.
Kich, J.D., N. Mores, I.A. Piffer, A. Coldebella, A. Amaral, L. Ramminger and M. Cardoso, 2005. [Factors associated with seroprevalence of Salmonella in commercial pig herds]. Ciencia Rural, 35: 398-405, (In Portuguese).
Kich, J.D., A. Coldebella, N. Mores, M.G. Nogueira and M. Cardoso et al., 2011. Prevalence, distribution and molecular characterization of Salmonella recovered from swine finishing herds and a slaughter facility in Santa Catarina, Brazil. Int. J. Food Microbiol., 151: 307-313.
Kich, J.D. and M. Cardoso, 2012. Salomelose. In: Doencas dos Suinos, Sobestiansky, Y. and D. Barcellos (Eds.). Canone Editorial, Goiania, pp: 257-264.
Kim, J., M.R. Marshall and C.I. Wei, 1995. Antibacterial activity of some essential oil components against five foodborne pathogens. J. Agric. Food Chem., 43: 2839-2845.
Kim, G.B., Y.M. Seo, C.H. Kim and I.K. Paik, 2011. Effect of dietary prebiotic supplementation on the performance, intestinal microflora and immune response of broilers. Poult Sci., 90: 75-82.
Kosina, P., J. Gregorova, J. Gruz, J. Vacek and M. Kolar et al., 2010. Phytochemical and antimicrobial characterization of Macleaya cordata herb. Fitoterapia, 81: 1006-1012.
Kreuzer, S., P. Janczyk, J. Aβmus, M.F.G. Schmidt, G.A. Brockmann and K. Nockler, 2012. No beneficial effects evident for Enterococcus faecium NCIMB 10415 in weaned pigs infected with Salmonella enterica serovar typhimurium DT104. Applied Environ. Microbiol., 78: 4816-4825. Kuhn, K.G., G. Sorensen, M. Torpdahl, M.K. Kjeldsen and T. Jensen et al., 2013. A long-lasting outbreak of Salmonella Typhimurium U323 associated with several pork products, Denmark, 2010. Epidemiol. Infect., 141: 260-268.
Leonard, S.G., T. Sweeney, B. Bahar, B.P. Lynch and J.V. O'Doherty, 2010. Effect of maternal fish oil and seaweed extract supplementation on colostrum and milk composition, humoral immune response and performance of suckled piglets. J. Anim. Sci., 88: 2988-2997.
Letellier, A., S. Messier, J. Pare, J. Menard and S. Quessy, 1999a. Distribution of Salmonella in swine herds in Quebec. Vet. Microbiol., 67: 299-306.
Letellier, A., S. Messier, L. Lessard and S. Quessy, 1999b. Assessment of different treatments to reduce Salmonella in swine. Proceedings of the 3rd International Symposium on Epidemiology and Control of Salmonella in Pork, August 5-7, 1999, Washington, DC., USA., pp: 299-302.
Leyman, B., F. Boyen, E. Verbrugghe, A. van Parys, F. Haesebrouck and F. Pasmans, 2012. Vaccination of pigs reduces Salmonella Typhimurium numbers in a model mimicking pre-slaughter stress. Vet. J., 194: 250-252.
Lo Fo Wong, D.M.A., J. Dahl, H. Stege, P.J. van der Wolf, L. Leontides, A. von Altrock and B.M. Thorberg, 2004. Herd-level risk factors for subclinical Salmonella infection in European finishing-pig herds. Prev. Vet. Med., 62: 253-266.
Lynch, M.B., T. Sweeney, J.J. Callan, J.T. O'Sullivan and J.V. O'Doherty, 2010. The effect of dietary Laminaria-derived laminarin and fucoidan on nutrient digestibility, nitrogen utilisation, intestinal microflora and volatile fatty acid concentration in pigs. J. Sci. Food Agric., 90: 430-437.
Machado, A.R., F.C. Gouveia, L.C.A. Picinin, J.D. Kich, M.R.I. Cardoso and S.M. Ferraz, 2013. [Microbiological and physico-chemical evaluation of pork leg treated with organic acids and/or steam under pressure in the control of surface contamination by Salmonella Typhimurium]. Ciencia Animal Brasileira, 14: 345-351, (In Portuguese).
Madec, F., F. Humbert, G. Salvat and P. Maris, 1999. Measurement of the residual contamination of post-weaning facilities for pigs and related risk factors. J. Vet. Med. Ser. B, 46: 47-56.
Majowicz, S.E., J. Musto, E. Scallan, F.J. Angulo and M. Kirk et al., 2010. The global burden of nontyphoidal Salmonella gastroenteritis. Clin. Infect. Dis., 50: 882-889.
Mannion, C., F.C. Leonard, P.B. Lynch and J. Egan, 2007. Efficacy of cleaning and disinfection on pig farms in Ireland. Vet. Rec., 161: 371-375.
Mannion, C., J. Egan, B.P. Lynch, S. Fanning and N. Leonard, 2008. An investigation into the efficacy of washing trucks following the transportation of pigs-a Salmonella perspective. Foodborne Pathog. Dis., 5: 261-271.
Margas, E., N. Meneses, B. Conde-Petit, C.E.R. Dodd and J. Holah, 2014. Survival and death kinetics of Salmonella strains at low relative humidity, attached to stainless steel surfaces. Int. J. Food Microbiol., 187: 33-40.
Martin-Pelaez, S., G.R. Gibson, S.M. Martin-Orue, A. Klinder and R.A. Rastall et al., 2008. In vitro fermentation of carbohydrates by porcine faecal inocula and their influence on Salmonella Typhimurium growth in batch culture systems. FEMS Microbiol. Ecol., 66: 608-619.
Martin-Pelaez, S., B. Peralta, E. Creus, A. Dalmau and A. Velarde et al., 2009. Different feed withdrawal times before slaughter influence caecal fermentation and faecal Salmonella shedding in pigs. Vet. J., 182: 469-473.
Martin-Pelaez, S., A. Costabile, L. Hoyles, R.A. Rastall and G.R. Gibson et al., 2010. Evaluation of the inclusion of a mixture of organic acids or lactulose into the feed of pigs experimentally challenged with Salmonella Typhimurium. Vet. Microbiol., 142: 337-345.
Martins, F.S., G. Dalmasso, R.M.E. Arantes, A. Doye and E. Lemichez et al., 2010. Interaction of Saccharomyces boulardii with Salmonella enterica serovar Typhimurium protects mice and modifies T84 cell response to the infection. PLoS One, Vol. 5. 10.1371/journal.pone.0008925
Mastroeni, P., J.A. Chabalgoity, S.J. Dunstan, D.J. Maskell and G. Dougan, 2001. Salmonella: Immune responses and vaccines. Vet. J., 161: 132-164.
Maxwell, F.J., S.H. Duncan, G. Hold and C.S. Stewart, 2004. Isolation, growth on prebiotics and probiotic potential of novel bifidobacteria from pigs. Anaerobe, 10: 33-39.
McDonnell, P., S. Figat and J.V. O'Doherty, 2010. The effect of dietary laminarin and fucoidan in the diet of the weanling piglet on performance, selected faecal microbial populations and volatile fatty acid concentrations. Animal, 4: 579-585.
Meunier, J.P., J.M. Cardot, E.G. Manzanilla, M. Wysshaar and M. Alric, 2007. Use of spray- cooling technology for development of microencapsulated capsicum oleoresin for the growing pig as an alternative to in-feed antibiotics: A study of release using in vitro models. J. Anim. Sci., 85: 2699-2710.
Miller, G., X. Liu, P. McNamara and D.A. Barber, 2005. Influence of Salmonella in pigs preharvest and during pork processing on human health costs and risks from pork. J. Food Protect., 68: 1788-1798.
Mogelmose, V., B. Nielsen, L.L. Sorensen, J. Dahl and A. Wingstrand et al., 1999. Eradication of multi-resistant Salmonella Typhimurium DT104 infections in 15 Danish swine herds. Proceedings of the 3rd International Symposium on Epidemiology and Control of Salmonella in Pork, August 5-7, 1999, Washington, DC., USA., pp: 367-369.
Molla, B., A. Sterman, J. Mathews, V. Artuso-Ponte and M. Abley et al., 2010. Salmonella enterica in commercial swine feed and subsequent isolation of phenotypically and genotypically related strains from fecal samples. Applied Environ. Microbiol., 76: 7188-7193.
Morrow, W.M., M.T. See, J.H. Eisemann, P.R. Davies and K. Zering, 2002. Effect of withdrawing feed from swine on meat quality and prevalence of Salmonella colonization at slaughter. J. Am. Vet. Med. Assoc., 220: 497-502.
Muller, A.J., P. Kaiser, K.E.J. Dittmar, T.C. Weber and S. Haueter et al., 2012. Salmonella gut invasion involves TTSS-2-dependent epithelial traversal, basolateral exit and uptake by epithelium-sampling lamina propria phagocytes. Cell Host Microbe, 11: 19-32.
Naqid, I.A., J.P. Owen, B.C. Maddison, D.S. Gardner and N. Foster et al., 2015. Prebiotic and probiotic agents enhance antibody-based immune responses to Salmonella Typhimurium infection in pigs. Anim. Feed Sci. Technol., 201: 57-65.
Neubauer, A. and M. Roof, 2005. Enterisol® SC-54 crossprotection against a virulent S. typhimurium strain. Proceedings of the 42nd Annual Meeting of American Association of Swine Veterinarians, March 2005, Toronto, pp: 245-248.
Newton, S.M., C. Lau, S.S. Gurcha, G.S. Besra and C.W. Wright, 2002. The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis. J. Ethnopharmacol., 79: 57-67.
O'Bryan, C.A., P.G. Crandall, V.I. Chalova and S.C. Ricke, 2008. Orange essential oils antimicrobial activities against Salmonella spp. J. Food Sci., 73: M264-M267.
O'Doherty, J.V., S. Dillon, S. Figat, J.J. Callan and T. Sweeney, 2010. The effects of lactose inclusion and seaweed extract derived from Laminaria spp. on performance, digestibility of diet components and microbial populations in newly weaned pigs. Anim. Feed Sci. Technol., 157: 173-180.
O'Reilly, K., A. Miller, E. Snary and A. Cook, 2007. Zoonoses action plan for Salmonella in slaughterage pigs: How will changes in sampling methods influence estimates of Salmonella? Proceedings of the 7th International Symposium on the Epidemiology and Control of Foodborne Pathogens in Pork, May 9-11, 2007, Verona, Italy, pp: 305-308.
Oyofo, B.A., J.R. DeLoach, D.E. Corrier, J.O. Norman, R.L. Ziprin and H.H. Mollenhauer, 1989. Prevention of Salmonella typhimurium colonization of broilers with D-Mannose. Poult. Sci., 68: 1357-1360.
Papatsiros, V.G., P.D. Katsoulos, K.C. Koutoulis, M. Karatzia, A. Dedousi and G. Christodoulopoulos, 2013. Alternatives to antibiotics for farm animals. CAB Rev.: Perspect. Agric. Vet. Sci. Nutr. Nat. Resour., 8: 1-15.
Partanen, K.H. and Z. Mroz, 1999. Organic acids for performance enhancement in pig diets. Nutr. Res. Rev., 12: 117-145.
Peisker, M., 2006. Feed processing-impacts on nutritive value and hygienic status in broiler feeds. Proceedings of the 18th Australian Poultry Science Symposium, February 20-22, 2006, Sydney, New South Wales, pp: 7-16.
Pellikaan, W.F., N. Andres-Elias, A. Durand, L.J.G.M. Bongers, S. van Laar-van Schuppen and
D. Torrallardona, 2010. Effect of carob bean gum, spray dried porcine plasma and sanguinarine on fermentation activity in the gut of weanling pigs. Livestock Sci., 133: 164-168.
Pickler, L., B.C.B. Beirao, R.M. Hayashi, J.F. Durau, M.C. Lourenco, L.F. Caron and E. Santin, 2013. Effect of sanguinarine in drinking water on Salmonella control and the expression of immune cells in peripheral blood and intestinal mucosa of broilers. J. Applied Poult. Res., 22: 430-438.
Pie, S., A. Awati, S. Vida, I. Falluel, B.A. Williams and I.P. Oswald, 2007. Effects of added fermentable carbohydrates in the diet on intestinal proinflammatory cytokine-specific mRNA content in weaning piglets. J. Anim. Sci., 85: 673-683.
Pieper, R., J. Bindelle, B. Rossnagel, A. van Kessel and P. Leterme, 2009. Effect of carbohydrate composition in barley and oat cultivars on microbial ecophysiology and proliferation of Salmonella enterica in an in vitro model of the porcine gastrointestinal tract. Applied Environ. Microbiol., 75: 7006-7016.
Piva, A., V. Pizzamiglio, M. Morlacchini, M. Tedeschi and G. Piva, 2007. Lipid microencapsulation allows slow release of organic acids and natural identical flavors along the swine intestine. J. Anim. Sci., 85: 486-493.
Poljak, Z., C.E. Dewey, R.M. Friendship, S.W. Martin and J. Christensen, 2008. Multilevel analysis of risk factors for Salmonella shedding in Ontario finishing pigs. Epidemiol. Infect., 136: 1388-1400.
Proescholdt, T.J., P. Turkson, J. McKean, P. Davies, J. Funk, S. Hurd and G. Beran, 1999. Salmonella in commercial swine from weaning through slaughter. Proceedings of the 3rd International Symposium on the Epidemiology and Control of Salmonella in Pork, August 5-7, 1999, Washington, pp: 161-164.
Quessy, S., E. Guevremont, G. Beauchamp, S. D'Allaire and S. Fournaise et al., 2005. Risk factors associated with presence of Salmonella in pigs in Canada. Proceedings of the 6th International Symposium on the Epidemiology and Control of Foodborne Pathogens in Pork, September 6-9, 2005, California, pp: 16-18.
Rajic, A., B.P. O'Connor, A.E. Deckert, J. Keenliside and M.E. McFall et al., 2007. Farm-level risk factors for the presence of Salmonella in 89 Alberta swine-finishing barns. Can. J. Vet. Res., 71: 264-270.
Rajkowski, K.T., S. Eblen and C. Laubauch, 1998. Efficacy of washing and sanitizing trailers used for swine transport in reduction of Salmonella and Escherichia coli. J. Food Protect., 61: 31-35.
Rattanachaikunsopon, P. and P. Phumkhachorn, 2010. Antimicrobial activity of basil (Ocimum basilicum) oil against Salmonella enteritidis in vitro and in food. Biosci. Biotechnol. Biochem., 74: 1200-1204.
Rawling, M.D., D.L. Merrifield and S.J. Davies, 2009. Preliminary assessment of dietary supplementation of Sangrovit® on red tilapia (Oreochromis niloticus) growth performance and health. Aquaculture, 294: 118-122.
Ricke, S.C., 2005. Ensuring the Safety of Poultry Feed. In: Food Safety Control in the Poultry Industry, Mead, G.C. (Ed.). Chapter 7, Woodhead Publishing Ltd., Cambridge, UK., ISBN-13: 978-0849334283, pp: 174-194.
Roberfroid, M., G.R. Gibson, L. Hoyles, A.L. McCartney and R. Rastall et al., 2010. Prebiotic effects: Metabolic and health benefits. Br. J. Nutr., 104: S1-S63.
Roof, M.B. and D.D. Doitchinoff, 1995. Safety, efficacy and duration of immunity induced in swine by use of an avirulent live Salmonella choleraesuis-containing vaccine. Am. J. Vet. Res., 56: 39-44.
Rosler, U., M. Stief, M. Leffler, U. Truyen and J. Lehmann et al., 2010. [Persistence, excretion and efficacy of an attenuated Salmonella vaccine in suckling piglets]. Der Praktische Tierarzt, 91: 59-65, (In German).
Rossel, R., L. Jouffe and P.A. Beloeil, 2009. [Analysis of factors associated with Salmonella isolation on pork carcasses using bayesian networks]. Journees Recherche Porcine, 41: 43-48, (In French).
Rostagno, M.H., H.S. Hurd, J.D. McKean, C.J. Ziemer, J.K. Gailey and R.C. Leite, 2003. Preslaughter holding environment in pork plants is highly contaminated with Salmonella enterica. Applied Environ. Microbiol., 69: 4489-4494.
Rostagno, M.H., H.S. Hurd and J.D. McKean, 2009. Split marketing as a risk factor for Salmonella enterica infection in swine. Foodborne Pathogens Dis., 6: 865-869.
Rostagno, M.H., S.D. Eicher and D.C. Lay Jr., 2011. Immunological, physiological and behavioral effects of Salmonella enterica carriage and shedding in experimentally infected finishing pigs. Foodborne Pathogens Dis., 8: 623-630.
Rostagno, M.H. and T.R. Callaway, 2012. Pre-harvest risk factors for Salmonella enterica in pork production. Food Res. Int., 45: 634-640.
Saif, Y.M., A.M. Fadly, J.R. Glisson, L.R. McDougald, L.K. Nolan and D.E. Swayne, 2008. Diseases of Poultry. 12th Edn., Blackwell Publishing, New York, ISBN: 9780813807188, Pages: 1409.
Sanchez-Vargas, F.M., M.A. Abu-El-Haija and O.G. Gomez-Duarte, 2011. Salmonella infections: An update on epidemiology, management and prevention. Travel Med. Infect. Dis., 9: 263-277. Schultz, A., 2008. Escherichia Coli. In: Therapeutic Microbiology: Probiotics and Related Strategies, Versalovic, J. and M. Wilson (Eds.)., ASM Press, Washington, D.C., pp: 83-96.
Schwartz, K.J., 1999. Salmonellosis. In: Diseases of Swine, Straw, B.E., S. D'Allaire, W.L. Mengeling and D.J. Taylor (Eds.)., Wiley, Oxford, UK., pp: 535-551.
Searle, L.E., W.A. Cooley, G. Jones, A. Nunez and B. Crudgington et al., 2010. Purified galactooligosaccharide, derived from a mixture produced by the enzymic activity of Bifidobacterium bifidum, reduces Salmonella enterica serovar Typhimurium adhesion and invasion in vitro and in vivo. J. Med. Microbiol., 59: 1428-1439.
Sedo, A., R. Malik, J. Vicar, V. Simanek and J. Ulrichova, 2003. Quaternary benzo[c]phenanthridine alkaloids as inhibitors of dipeptidyl peptidase IV-like activity bearing enzymes in human blood plasma and glioma cell lines. Physiol Res., 52: 367-372.
Shoaf, K., G.L. Mulvey, G.D. Armstrong and R.W. Hutkins, 2006. Prebiotic galactooligosaccharides reduce adherence of enteropathogenic Escherichia coli to tissue culture cells. Infect. Immunity, 74: 6920-6928.
Si, W., J. Gong, C. Chanas, S. Cui, H. Yu, C. Caballero and R.M. Friendship, 2006. In vitro assessment of antimicrobial activity of carvacrol, thymol and cinnamaldehyde towards Salmonella serotype Typhimurium DT104: Effects of pig diets and emulsification in hydrocolloids. J. Applied Microbiol., 101: 1282-1291.
Siekkinen, K.M., L. Nuotio, J. Ranta, R. Laukkanen, S. Hellstrom, H. Korkeala and R. Maijala, 2006. Assessing hygiene proficiency on organic and conventional pig farms regarding pork safety: A pilot study in Finland. Livestock Sci., 104: 193-202.
Skandamis, P., E. Tsigarida and G.J.E. Nychas, 2000. Ecophysiological attributes of Salmonella typhimurium in liquid culture and within a gelatin gel with or without the addition of oregano essential oil. World J. Microbiol. Biotechnol., 16: 31-35.
Smid, J.H., L. Heres, A.H. Havelaar and A. Pielaat, 2012. A biotracing model of Salmonella in the pork production chain. J. Food Protect., 75: 270-280.
Splichal, I., I. Trebichavsky, A. Splichalova and P.A. Barrow, 2005. Protection of gnotobiotic pigs against Salmonella enterica serotype Typhimurium by rough mutant of the same serotype is accompanied by the change of local and systemic cytokine response. Vet. Immunol. Immunopathol., 103: 155-161.
Splichalova, A., I. Trebichavsky, V. Rada, E. Vlkova, U. Sonnenborn and I. Splichal, 2011. Interference of Bifidobacterium choerinum or Escherichia coli Nissle 1917 with Salmonella Typhimurium in gnotobiotic piglets correlates with cytokine patterns in blood and intestine. Clin. Exp. Immunol., 163: 242-249.
Spring, P., C. Wenk, K.A. Dawson and K.E. Newman, 2000. The effects of dietary mannaoligosaccharides on cecal parameters and the concentrations of enteric bacteria in the ceca of salmonella-challenged broiler chicks. Poult. Sci., 79: 205-211.
Suryanarayana, M.V.A.N., J. Suresh and M.V. Rajasekhar, 2012. Organic acids in swine feeding-a review. Agric. Sci. Res. J., 2: 523-533.
Swanenburg, M., H.A. Urlings, D.A. Keuzenkamp and J.M.A. Snijders, 2001. Salmonella in the lairage of pig slaughterhouses. J. Food Protect., 64: 12-16.
Sweeney, T., S. Dillon, J. Fanning, J. Ega and C.J. O'Shea et al., 2011. Evaluation of seaweed- derived polysaccharides on indices of gastrointestinal fermentation and selected populations of microbiota in newly weaned pigs challenged with Salmonella Typhimurium. Anim. Feed Sci. Technol., 165: 85-94.
Szabo, I., L.H. Wieler, K. Tedin, L. Scharek-Tedin and D. Taras et al., 2009. Influence of a probiotic strain of Enterococcus faecium on Salmonella enterica serovar Typhimurium DT104 infection in a porcine animal infection model. Applied Environ. Microbiol., 75: 2621-2628.
Tanaka, T., K. Metori, S. Mineo, M. Hirotani, T. Furuya and S. Kobayashi, 1993. Inhibitory effects of berberine-type alkaloids on elastase. Planta Medica, 59: 200-202.
Tassou, C.C., E.H. Drosinos and G.J.E. Nychas, 1995. Effects of essential oil from mint (Mentha piperita) on Salmonella enteritidis and Listeria monocytogenes in model food systems at 4° and 10°C. J. Applied Bacteriol., 78: 593-600.
Ten Bruggencate, S.J.M., I.M.J. Bovee-Oudenhoven, M.L.G. Lettink-Wissink, M.B. Katan and R. van der Meer, 2004. Dietary fructo-oligosaccharides and inulin decrease resistance of rats to salmonella: Protective role of calcium. Gut, 53: 530-535.
Trebichavsky, I., V. Rada, A. Splichalova and I. Splichal, 2009. Cross-talk of human gut with bifidobacteria. Nutr. Rev., 67: 77-82.
Tschirner, K., 2004. Untersuchungen zur wirksamkeit und zum nachweis des pflanzlichen alkaloids sanguinarin beim schwein. Christian Albrechts Universitat Kiel, Kiel.
Turgis, M., J. Han, S. Caillet and M. Lacroix, 2009. Antimicrobial activity of mustard essential oil against Escherichia coli O157:H7 and Salmonella typhi. Food Control, 20: 1073-1079.
Van Immerseel, F., F. Boyen, I. Gantois, L. Timbermont and L. Bohez et al., 2005. Supplementation of coated butyric acid in the feed reduces colonization and shedding of Salmonella in poultry. Poult. Sci., 84: 1851-1856.
Van Immerseel, F., J.B. Russell, M.D. Flythe, I. Gantois and L. Timbermont et al., 2006. The use of organic acids to combat Salmonella in poultry: A mechanistic explanation of the efficacy. Avian Pathol., 35: 182-188.
Van der Heijden, M., H. van Dam, D. Niewerth and K. Frankena, 2005. Effectiveness of Salmonella control strategies in fattening pigs. Proceedings of the 6th International symposium on the epidemiology and control of foodborne pathogens in pork, September 6-9, 2005, Iowa State University, California, pp: 145-148.
Van der Wolf, P.J., F.W. van Schie, A.R.W. Elbers, B. Engel, H.M.J.F. van der Heijden, W.A. Hunneman and M.J.M. Hunneman, 2001a. Epidemiology: Administration of acidified drinking water to finishing pigs in order to prevent Salmonella infections. Vet. Quart., 23: 121-125.
Van der Wolf, P.J., W.B. Wolbers, A.R.W. Elbers, H.M.J.F. van der Heijden and J.M.C.C. Koppen et al., 2001b. Herd level husbandry factors associated with the serological Salmonella prevalence in finishing pig herds in The Netherlands. Vet. Microbiol., 78: 205-219. Varga, C., A. Rajic, M.E. McFall, R.J. Reid-Smith and S.A. McEwen, 2009. Associations among antimicrobial use and antimicrobial resistance of Salmonella spp. isolates from 60 Alberta finishing swine farms. Foodborne Pathog. Dis., 6: 23-31.
Vieira-Pinto, M., R. Tenreiro and C. Martins, 2006. Unveiling contamination sources and dissemination routes of Salmonella sp. in pigs at a Portuguese slaughterhouse through macrorestriction profiling by pulsed-field gel electrophoresis. Int. J. Food Microbiol., 110: 77-84.
Wales, A.D., V.M. Allen and R.H. Davies, 2010. Chemical treatment of animal feed and water for the control of Salmonella. Foodborne Pathog. Dis., 7: 3-15.
Walsh, M.C., M.H. Rostagno, G.E. Gardiner, A.L. Sutton, B.T. Richert and J.S. Radcliffe, 2012. Controlling infection in weanling pigs through water delivery of direct-fed microbials or organic acids. Part I: Effects on growth performance, microbial populations and immune status. J. Anim. Sci., 90: 261-271.
Wegener, H.C., 2010. Danish initiatives to improve the safety of meat products. Meat Sci., 84: 276-283.
Wheatley, P., E.S. Giotis and A.I. McKevitt, 2014. Effects of slaughtering operations on carcass contamination in an Irish pork production plant. Irish Vet. J., Vol. 67.
Wierup, M., 1997. Principles for integrated surveillance and control of Salmonella in swine production. Proceedings of the 2nd International Symposium of Epidemiology and Control of Salmonella in Pork, August 20-22, 1997, Copenhagen, pp: 42-49.
Wierup, M. and P. Haggblom, 2010. An assessment of soybeans and other vegetable proteins as source of Salmonella contamination in pig production. Acta Vet. Scand., Vol. 52.
Wilkins, W., A. Rajic, C. Waldner, M. McFall, E. Chow, A. Muckle and L. Rosengren, 2010. Distribution of Salmonella serovars in breeding, nursery and grow-to-finish pigs and risk factors for shedding in ten farrow-to-finish swine farms in Alberta and Saskatchewan. Can. J. Vet. Res., 74: 81-90.
Williams, Jr. L.P. and K.W. Newell, 1968. Sources of Salmonellas in market swine. J. Hygiene, 66: 281-293.
Wray, C., 2001. Review of research into Salmonella infection in pigs: A report. Meat and Livestock Commission, UK.
Yin, F., A. Farzan, Q. Wang, H. Yu and Y. Yin et al., 2014. Reduction of Salmonella enterica serovar Typhimurium DT104 infection in experimentally challenged weaned pigs fed a lactobacillus- fermented feed. Foodborne Pathogens Dis., 11: 628-634.
Yin, Y.L., Z.R. Tang, Z.H. Sun, Z.Q. Liu and T.J. Li et al., 2008. Effect of galacto-mannan- oligosaccharides or chitosan supplementation on cytoimmunity and humoral immunity in early- weaned piglets. Asian-Australasian J. Anim. Sci., 21: 723-731.
Zdarilova, A., E. Vrublova, J. Vostalova, B. Klejdus and D. Stejskal et al., 2008. Natural feed additive of Macleaya cordata: Safety assessment in rats a 90-day feeding experiment. Food Chem. Toxicol., 46: 3721-3726.
Zheng, D.M., M. Bonde and J.T. Sorensen, 2007. Associations between the proportion of Salmonella seropositive slaughter pigs and the presence of herd level risk factors for introduction and transmission of Salmonella in 34 Danish organic, outdoor (non-organic) and indoor finishing-pig farms. Livestock Sci., 106: 189-199.







