Introduction
Animal diets based on corn and soybean meal are very deficient in available phosphorus (P) to the point that supplementation is necessary using sources of high P availability. Dicalcium phosphate is commonly used as a source of supplemental P. Commercial dicalcium phosphate is a mixture of varying amounts of monocalcium and dicalcium phosphates, phosphoric acid, calcium carbonate, and impurities (Lima et al., 1995). Some animal production technicians point out that rock phosphates is an alternative dietary P source in animal nutrition due to their very low price compared to feed grade phosphates. It is well known that rock phosphates are not intended to be used in animal diets for not being submitted to manufacturing procedures to guarantee the adequate purity degree for feeding food-producing animals, and to their very low P bioavailability. In addition, high levels of certain mineral elements can be toxic to the animal.
Digestibility methods are not adequate for feed availability determination. They only give apparent digestibility of the total P in the diet (Cromwell, 1992) and do not allow to assess the P digestibility in individual ingredients. Slope ratio has been chosen to assess the P availability by many researchers. In general, the P availability is calculated as a relative biological value compared with a standard phosphate source (purified grade) and is expressed simultaneously from several biological response criteria, such as animal performance and bone characteristics. Performance and bone characteristics have shown to be an easy and efficient method to study the available P in feedstuffs.
In an attempt to determine the P bioavailability in feed grade phosphates (FP) and rock phosphates (RP), and to verify if the use of such alternative P sources could be used in animal nutrition, an experiment with growing pigs fed with FP or RP as a supplemental P source was carried out. The aim of this study was to evaluate three FP and three RP, using a purified-grade calcium phosphate as the reference standard on performance, blood and bone characteristics of growing pigs.
Material and Methods
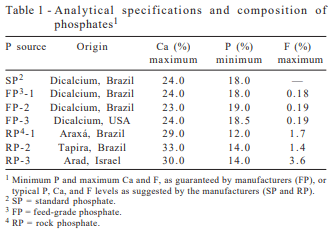
Three rock phosphates were tested along with three feed-grade phosphates. The reference standard phosphate (SP) was a purified-grade dibasic dehydrate calcium phosphate (CaHPO4.2H2O) produced in Brazil. Feed-grade dicalcium phosphates FP-1 and FP-2 were products from the Brazilian phosphate industry and FP-3 (monodicalcium phosphate) was from U.S. RP-1 and RP-2 were obtained from Brazilian mines (Araxá and Tapira), and RP-3 was from Arad mine site in Israel (Table l).
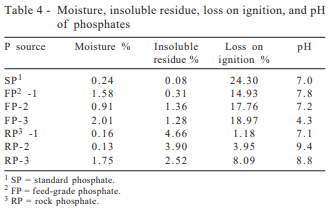
Standard analytical methods recommended by AOAC (1995) were used. All phosphates were analyzed for moisture, insoluble residue, loss on ignition, and pH. Mineral analysis included the essential Ca, P, Mg, Na, K (macroelements), Co, Cu, Fe, Mn, Mo, S, Se, Zn (microelements), and the potentially toxic elements Al, F, As, B, Ba, Bi, Cd, Cr, Hg, Ni, Pb, Sb, Sn, Ti, V, W, and the radioactive U.
Analysis were performed at the IMC-Agrico laboratory, which employed a segmented-flux analytical system for Ca and P determinations and an ion selective electrode for fluorine analysis. Atomic absorption (Perkin-Elmer model 5100, Perkin-Elmer, Norwalk, CT, 06859) was used for Al, Fe, and Mg. Trace-elements were analyzed by plasma spectrometry (Perkin-Elmer Soiax Elan 500) and moisture determinations were performed using a vacuum oven.
pH values were obtained from 5 g of phosphate samples in 250 mL solutions (water pH = 7.0) using a potentiometer. Moisture was determined at 80°C, basically to quantify the hygroscopicity of the products, since water of crystallization losses may occur with temperatures as low as 109°C. Loss on ignition was determined at 1,000°C in dry samples and represents losses in water of crystallization, carbon dioxide from carbonates and volatile mineral elements such as As, Hg and halogens. Insoluble residue in HCl followed by HNO3 basically quantifies the silica content.
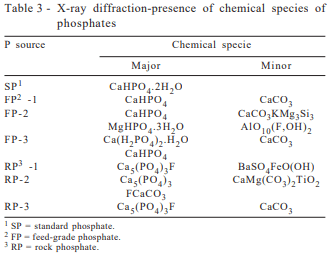
All X-ray diffraction assays were performed at the “Laboratório de Caracterização Tecnológica” (LCT) laboratory using methodology described by Lima et al., 1995, for identification of the chemical species present in the samples. Analytical procedures employed an X-ray diffractometer PHILLIPS-PW1880 (PHILLIPS, 7602 EA, Almelo, Holland) with a PW1710 controller and PC-APD (automated powder diffraction) software.
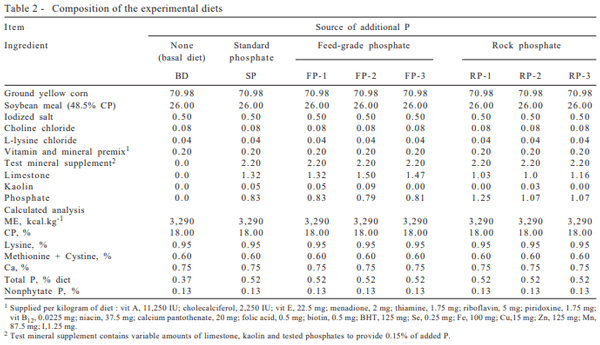
Ingredients from the diets were analyzed according to AOAC (1995) methods to determine dry matter, crude protein, ether extract, ash, Ca and P. A corn and soybean meal-based diet (Table 2) was formulated to contain adequate levels of all nutrients, except for P, in order to meet the swine requirements (NRC, 1998), and contained 0.37% of P. The supplemented diets were formulated by using appropriate levels of each one of the seven P sources to provide 0.15% of additional phosphorus (making 0.52% of total P). The calcium content of the diets was kept constant (0.75%) by varying the limestone and kaolin levels added. Calcium and phosphorus analyses in the diets were in agreement with the calculated values.
One hundred and ninety-two crossbred pigs with initial average weight of 14.3 kg, were distributed as a complete randomized block design into eight treatments [(4 P sources (without P, SP, FP[3], RP[3] x 2 genders [barrows e gilts)] with six replicates blocks (pens) of four pigs per pen (two barrows and two gilts) each, for 35 days (14.4 to 39.9 kg). Pigs were housed in forty-eight 2.25 x 0.73 m partially slotted heated-floor pens, in groups of 2 barrows and 2 gilts per pen. The appropriate experimental diet and water were provided ad libitum throughout the entire 35-d experimental period. Weight gain, feed intake, and feed gain for each pen were recorded at 7, 14, 21, 28 and 35 days.
At the end of the experiment, one barrow and one guilt from each pen were randomly selected for blood sampling by ear vein puncture, and one barrow and one guilt were also randomly selected from each pen and slaughtered (electrical stunning followed by exsanguination). Metacarpal, metatarsal and femur were excised, and were cleaned from muscles and connective tissues, and tested for bone strength. To measure the bone strength, each metacarpal and metatarsal was held by two supports spaced 2.5 cm apart (5 cm for femurs) and force was applied to the midpoint of the bone by a probe attached to a 50 kg load cell (500 kg for femurs), with a crosshead speed of 50 mm/min (Ottawa Texture Measuring Instrument)1. The breaking strength was recorded in kilograms by pen deflection through a recorder. The same metacarpal and metatarsal samples were subsequently extracted with diethyl ether for 8 h. Combined metacarpal and metatarsal bone samples from each replicate were ashed at 600°C overnight for ash content determination.
The plasma was separated by blood centrifugation for 10 min at 2,000 x g and saved for plasma calcium, phosphorus, and alkaline phosphatase (ALP) determinations. Plasma samples were processed by an autoanalyzer Technicon (model RA-100)2. Plasma characteristics were determined according to methods described by Connerty and Briggs (1966) for calcium, Gomorry (1942) and Raabe (1955) for phosphorus, and Bowers and McComb (1966) for ALP.
Statistical analysis was performed according to a complete randomized block design. The 35-d weight gain, feed intake, feed:gain ratio, plasma Ca, P and ALP, as well as bone ash percentage, and bone breaking strength data were analyzed using analysis of variance procedures of SAS software (SAS, 1990). The analysis of data adopted the statistic model represented by Yij = m + Bi + Sj + eij, where Yij is the observed value of the variable response of the cage; m is the estimated mean; Bj is the block effect; Sj is the source effect; eij is the experimental error. Orthogonal comparisons were used to test the effects of the various phosphate groups, by partitioning the 7 degrees of freedom for the treatment effect. The relative bioavailability of phosphorus (RBP) for each test phosphate was determined by the “three point assay” as described by Ammerman et al. (1995). Body weight gain, bone ash percentage, and bone breaking strength values were regressed on phosphorus added to basal diet within each phosphorus source and the slope of the regression line for each test source was divided by the slope of the standard phosphate (100% RBP). Values obtained for the control group were used as a common intercept in regression equations to estimate the P availability.
Results and Discussion
Analytical values obtained for the three FP sources were all in compliance with the manufacturer’s guarantee levels. The most predominant chemical substances present in all commercial FP were calcium phosphates and calcium carbonates (Table 3). For the reference standard, purified grade dibasic dehydrated calcium phosphate, dibasic dehydrated calcium phosphate seems to be the most predominant product found.
Among the feed-grade samples, monocalcium phosphate was found only in the U.S. di-monocalcium phosphate (FP-3). The anhydrous salt of dicalcium phosphate was identified as the most predominant substance present in FP-1, FP-2, and FP-3. Calcitic limestone was found in large proportions in all three commercial dicalcium phosphate products and may be the result of excess calcium carbonate added to neutralize the phosphoric acid during the industrial processing.
These results are in agreement with previous publication from Lima et al. (1995, 1999), which reported X-ray diffraction evaluations for two pure and ten commercial dicalcium phosphates and found that monocalcium phosphate was present in only some commercial products. According to these reports, the most predominant chemical species present in all commercial dicalcium phosphates were calcium carbonate and anhydrous dicalcium phosphate. These results are consistent with the pH values obtained (Table 4), showing the lowest value for FP-3 (pH=4.3), when compared to SP (pH=7.0), and FP-1 (pH=7.8), and FP-2 (pH=7.2).
Considering the rock phosphates, the most predominant chemical substance present was fluorohapatite, followed by smaller amounts of barium sulfate and iron oxide (RP-1), calcium carbonates (RP-2 and RP-3), and titanium oxide (RP-2).
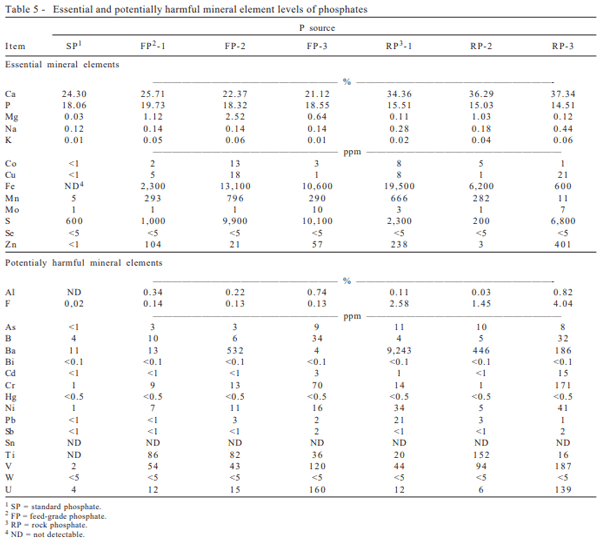
In routine feed formulation for swine and poultry, phosphate supplements are largely used at levels not to exceed the 2% limit in conventional diets. Considering the swine requirements for minerals according to the NRC (1998), Cu, Mg, Mn, Se and Zn levels present in feed phosphates are inconsequential.
The comparison between the maximum tolerable level of several mineral elements for domestic animals, as suggested by the NRC (2005) and values observed in this study (Table 5) indicates no toxicity risks for the use of any of the feed phosphates studied in normal diets for swine.
Previous publications from Lima et al. (1995, 1999) reported analytical values for 26 to 31 mineral elements in three pure dicalcium phosphates, and in fourteen commercial dicalcium phosphate samples. The comparison between the average results for three dicalcium phosphate samples obtained in this study and the levels reported by Lima et al. (1995, 1999) showed comparable values for most mineral elements analyzed.
Nutritionally essential element concentrations in common macromineral supplements have been reported by NRC (2005). Fe, Cu, Mg, Mn, and Zn levels are generally compared to those obtained in this study.
In contrast to the safety demonstrated for feed-grade phosphates in supplementing animal diets, this does not seem to be true for the rock phosphates tested. Rock phosphates may represent considerable risk of toxicity for use in animal diets, considering their F and Ba levels. According to recommendations from the AAFCO (1973), phosphate sources for use in animal feeds should not contain more than 1 part of F to 100 parts of P, and according to NRC (2005), the maximum tolerable level for swine in phosphate sources, used at a dietary level of 2%, is 1,000 ppm for Ba. Rock from Araxa (RP-1) was found to contain 1,510% excess F (2.58 vs 0.16%), and 1,330% excess Ba (14,270 vs 1,000 ppm). The F level of Tapira phosphate (RP-2) exceeded the tolerance limit by 870% (1.45 vs 0.15 %). The F level of the Arad sample (RP-3) was found to exceed by 2,790% the tolerance limit (4.04 vs 0.15%).
These results are in line with the findings of Lima et al. (1999), who found for three FP and seven RP samples, that the mineral concentrations were safe for all FP when compared to NRC or European standards. However, the RP levels exceeded the tolerance limits for F, Ba, As, Pb and Cd, and were particularly high when compared to FP, for the radioactive Th. Tapira rock was high in F and As, Araxá rock was high in F, Ba, As, Pb and Th, rock from Patos de Minas was high in F, two rock samples from Catalão, Copebras and Goiasfértil, were high in F, Ba, and As, Arad and North Carolina rocks were high in F, As and Cd.
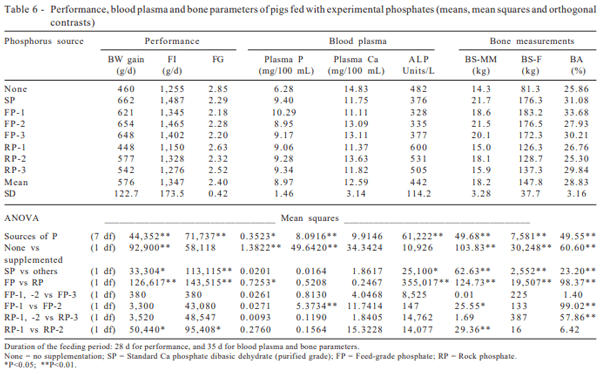
Higher level of dietary P improved the performance parameters, regardless of the P source (P<0.01), when compared to the control diet (Table 6). Considering an average value for all 7 supplemented diets, compared with the control diet fed pigs, the performance was improved (P<0.01) by 22.4% for BW gain (522 vs 460 g/d), 8.4% for FI (1,360 vs 1,255 g/d), and 18% for FG (2.34 vs 2.85). These findings are in agreement with Cromwell et al. (1970), which observed improvements in BW gain, FI and FG of pigs in response to increase dietary P. Similar results were recorded for broilers by Fernandes et al.(1999), who studied four feed-grade, and four agricultural-grade phosphates, as well as one standard purified grade P source, and reported that the performance was improved by 31% for BW, 34% for BW gain, 7% for FI, and 20% for FG, in response to increasing dietary P (0.08 or 0.16% P added to a 0.48% P basal diet).
The toxicity of feeding rock phosphates to pigs was clearly shown by the evaluation of the performance results.
The performance was strongly depressed (P<0.01) by supplementing diets with rock phosphates (RP-1, 2 or 3) when compared to feeding the feed-grade products. Calculating an average value for the 3 diets supplemented with RP-1, 2 or 3 when compared to the FP-1, 2 or 3, the performance was depressed (P<0.01) by 18.5% for BW gain (522.3 vs 641.0 g/d), 9% for FI (1,276 vs 1,402 g/d), and 13% for FG (2.49 vs 2.20). These results were in disagreement with Plumlee et al. (1958), who found similar performance results for swine fed either with dicalcium phosphate or with soft rock phosphate in a 90-day feeding trial. However, the results of this study are in agreement with Potter (1988), who observed a poor performance for turkeys fed with Curaçao rock phosphate, and with the report of Barbosa et al. (1992), who recorded a reduction of 11.6% in the BW gain, and a depression of 5% in FG for swine fed with diets supplemented with rock phosphate originated from the Brazilian mine of Patos de Minas.
Similar results were recorded for broilers by Fernandes et al. (1999), who observed a decrease in performance by 11% for BW, 12% for BW gain, and 14% for FI when diets were supplemented with agricultural-grade phosphates, when compared to feed-grade phosphates. The results of this study were also in agreement with Garzillo et al. (1997), who reported that the performance was depressed (P<0.01) by supplementing diets with RP (7 samples), when compared to FP (3 samples) supplementation. Body weight was decreased by 7% (679.9 vs 729.2 g), BW gain by 7% (638.0 vs 687.1 g) and FI by 7% (1,003 vs 1,074 g).
Higher levels of dietary phosphorus increased the plasma phosphorus concentration (P<0.01). The average value of all supplemented diets was 32.9% higher than the basal diet (9.36 vs 6.28 mg/dL, respectively). Previous experiments have demonstrated that higher dietary P levels increase the plasma phosphorus concentrations (Plumlee et al., 1958; Mahan, 1982). However, the plasma calcium levels decreased as the plasma phosphorus levels increased (Mahan, 1982), in response to the dietary phosphorus addition. Maxson & Mahan (1983) also reported similar response of plasma phosphorus and calcium levels, with an inverse relationship between both minerals. Despite this relationship, it was observed in the present study that both level and source of phosphorus did not influence the calcium levels. Based on these results, it could be concluded that the plasma calcium concentration was a non-sensitive approach to assess the relative phosphorus bioavailability.
Alkaline phosphatase measurements showed a decrease of about 10.5% in the plasma concentrations when basal diet had 0.15% of additional phosphorus (481.5 vs 435.9 IU/L); however, the contrast between basal diet and supplemented diets was not significant. The average value for dicalcium phosphates (347 IU/L) was 57.3% lower (P<0.0001) than that observed for rock phosphates (545 IU/L). While FP decreased the plasma levels (346 IU/L), RP promoted the highest levels in all classes of phosphorus source (545 IU/L), which raised the average of the supplemented diets. Thus, the difference between basal diet (482 IU/L) and average for all supplemented diets (436 IU/L) was not significant. This fact was confirmed by the difference (P<0.01) between dicalcium phosphate and rock phosphate. Reduction by 2.6% in plasma P was not significant when rock was compared to dicalcium phosphate. On the other hand, Boyd et al. (1983) observed ALP activity declining along increasing dietary P levels, until the diet reached levels close to NRC (1998) minimum requirements. Although the present results showed differences between feed grade and rock phosphates only for ALP, the influence of rock phosphate in the P concentrations was noted by Plumlee et al. (1958), who verified that supplementation by dicalcium phosphate increased (P<0.01) the P levels more than Curaçao rock phosphate.
Lower levels of dietary P (basal diet) caused lower metacarpal and metatarsal, and femur breaking strength (P<0.01), and BA (P<0.01). When swine received additional P, the average of BS-MM was 18.7 kg, BS-F was 157 kg, and BA was 29.26%. Swine fed with basal diet had 14.3 kg, 81 kg, and 25.86%, respectively. These results showed an improvement of 23.8% (metacarpal/metatarsal) and 48.3% (femur) in BS and 11.6% in the mineral content of bones, when diets had additional P. The increasing in BS-MM was according to Cromwell et al. (1972), who observed increasing from 125.9 to 141.1 kg of metacarpal breaking strength when dietary phosphorus increased in 0.15%. The average BS-F was reduced (P<0.01) almost to half (from 157 to 81 kg) when animals did not receive P supplementation. Also, the average BA was lower (P< 0.01) for non-supplemented pigs (25.86%) when compared to supplemented animals (29.26%).
Elaborated phosphorus sources improved (P>0.01) metacarpal and metatarsal, and BS-F. The average BS for swine supplemented with dicalcium sources was 18.6% higher than rock sources (20.1 vs 16.4 kg). The same behavior was observed by Plumlee et al. (1958) who reported higher BS-F (P<0.05) in response to feed-grade phosphate supplementation than that obtained for soft phosphates. Gomes et al. (1992) observed improved (P<0.05) bone ash with FP in comparison to rock phosphate. Also, higher metacarpal and femur breaking strength values were observed (P<0.01). Both BA and BS reduction were probably the result of less available P on diets supplemented with RP. For BS-F, the difference was 26.2% (177 vs 131 kg, respectively). Bone ash was improved in 10.8% (30.6 vs 27.3%, respectively). All bone measurements showed difference (P<0.01) between feed-grade and rock phosphates. The same behavior was reported by Fernandes et al. (1999), who observed that diets supplemented with agricultural phosphates caused a decrease (P<0.01) in bone ash and breaking strength of broilers, when compared to FP.
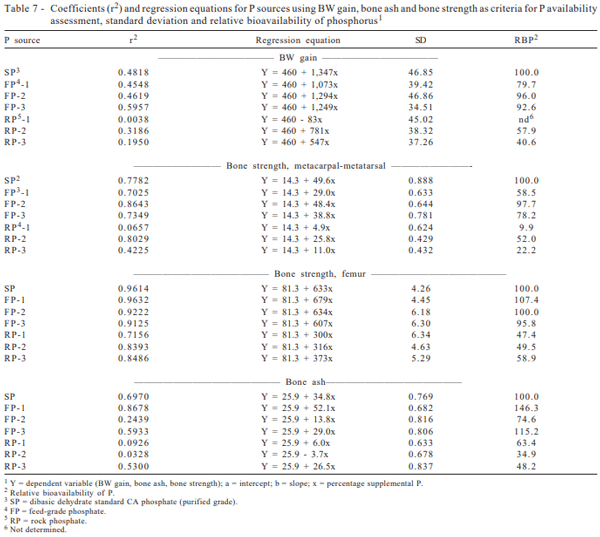
Coefficients of RBP determination were expressed as percentage of the dibasic dehydrate standard calcium phosphate (purified-grade), for which P was considered to be 100% available (Table 7). All FP samples were more available than RP samples, considering any of the studied parameters.
Average RBP for BW gain was 89.4% and 49.2% for FP and RP, respectively. Among the several sources, the highest value for bioavailability was observed for SP. The RP-1 source resulted in negative intercept, being not possible to calculate the availability. This fact must be due to the lack of quality and excess of impurities, resulting in very low animal performance, when compared to FP sources. Gomes et al. (1992) observed 98.6% of RBP for RP-2 in relation to dicalcium phosphate.
Mean RBP values calculated through BS-MM for FP and RP were 78.1 and 28.0%, respectively. Once again, the lowest availability value was for RP-3 (9.9% BRP).
The RBP values observed for FP and RP were 101.1% and 51.9%, respectively, when considering BS-F.
The relative bioavailability of phosphorus of FP and RP relative to BA were 112 and 48.8%, respectively.
Conclusions
The analytical values obtained for the three feed-grade phosphate sources studied indicate that no mineral elements was found at toxic levels for use in animal diets. Analytical evaluations of the three rock phosphates studied revealed that these products could be toxic to animals. All feed-grade phosphates used in this trial resulted in better performance when compared to the use of rock phosphates. Rock phosphates promoted lower bone strength and less bone ash when compared to feed-grade phosphates. The relative bioavailability of phosphorus from feed-grade phosphates was considered high, while rock phosphates showed poor phosphorus availability, which means lower animal production, higher levels of supplementation and more phosphorus releasing to the environment. Since most studies conducted to assess the bone characteristics revealed better breaking strength values and ash content for feedgrade phosphates, the results show that rock phosphate should not be used as source of supplementary phosphorus.
Acknowledgments
On behalf of his students, colleagues and many friends throughout Brazil and abroad, this paper is dedicated to the life, accomplishments and memories of their respected counselor, teacher, researcher and friend, the late Professor, Doctor Felix R. de Lima. Special acknowledgements are due to Dr Virgil W. Hays, former professor at the University of Kentucky and Dr Gary L. Cromwell, researcher at the University of Kentucky, for their cooperation with students of the Departamento de Nutrição e Produção Animal of FMVZ-USP.
The authors are grateful to IMC-Agrico, Mundelein, IL, 60060, Ralston Purina, Paulinia, SP, Brazil in laboratory analysis. The assistance of Mitsui S.A., São Paulo, SP, 01311-940, Brazil, and Fosfértil, Uberaba, MG, 38102-970, Brazil in obtaining some phosphate source samples is acknowledged.
This article was originally published in Revista Brasileira de Zootecnia., v.38, n.1, p.90-98, 2009. This is an Open Access article distributed under a Creative Commons Attribution License. 








.jpg&w=3840&q=75)