Introduction
The association of all the stressors related to early weaning of piglets results in reduction in daily feed intake, especially during the first week. The interruption of feed intake in this phase results in intestinal villous atrophy and increased crypt depth (Pierce et al, 2005).
According to Pinheiro & Machado (2007), the performance of piglets in the first week after weaning is strongly correlated with the weight at the end of the nursery phase, and the number of days required to reach slaughter weight.
Some nutritional strategies for piglets at this stage have been taken to mitigate the harmful effects of weaning, including the use of lactose, whose benefit is mainly linked to the increase and maintenance of feed consumption due to the improved palatability of the feed (Bertol et al., 2000). However, diets containing lactose have their cost increased due to the high price of this ingredient, which is also used in human food. Thus, it is necessary to look for ingredients that, like lactose or by different mechanisms, can minimize the detrimental effects of early weaning.
The amino acid glutamine constitutes an ingredient with the potential to improve the performance of piglets through its action in the maintenance of the small intestine mucosal structure (Yi et al., 2005), in addition to its role as precursor of proteins, nucleotides and polyamines (Sources et al., 2007). The action of this amino acid, termed conditionally essential, is related to the multiplication of cells of intense development, such as enterocytes of the intestinal mucosa and immune cells (Abreu & Donzele, 2008; Wu et al., 1996).
Thus, there is a need to evaluate the effect of adding L-glutamine and L-glutamate in diets containing different levels of lactose on the performance and intestinal mucosa structure of piglets weaned at 21 days.
Material and Methods
The experiment was conducted in the Setor de Suinocultura do Departamento de Zootecnia da Universidade Federal de Viçosa. One hundred and eight hybrid piglets weaned at 21±1 day of age and weighing 6.12±0.70 kg were allotted in a completely randomized block design with six treatments, six replicates, and three piglets per experimental unit. Weaning weight and piglets’ kinship were considered as criteria in the blocks formation.
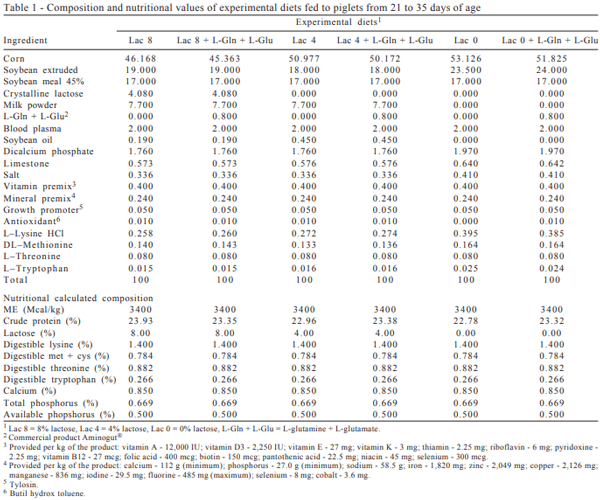
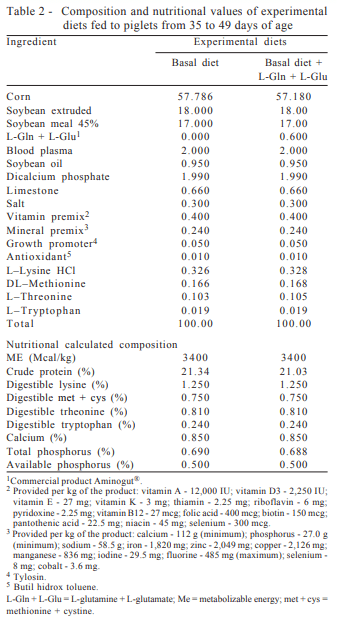
Treatments consisted of diets containing 0.0, 4.0, and 8.0% lactose, supplemented with 0.8% L-glutamine + L-glutamate (L-L-Glu + Gln), from 21 to 35 days. From 36 to 49 days, all piglets were fed the same diet free of lactose, and those that were fed 0.80% L-glutamine + L-glutamate from 21 to 35 days received 0.60% L-glutamine + L-glutamate. The experimental diets from 21 to 35 days (Table 1) and from 35 to 49 days (Table 2) were formulated to meet or exceed the requirements of pigs at the pre-starter phase (7 to 15 kg) for all nutrients (Rostagno et al., 2005).
Piglets were housed in suspended metal cages with slatted floors, semi-automatic feeders and nipple drinkers, located in a facility with concrete floor, wood ceiling and covered with ceramic tile. The room temperature was kept at 28 °C, using hoods and/or heaters when needed.
For evaluation of performance, piglets were weighed at the beginning (35 days) and end of the experiment (49 days), and feed leftovers were collected and weighed daily when the animals reached 35 and 49 days, i.e., at the end of each period, for calculation of average daily feed intake (ADFI), average daily gain (ADG) and feed conversion (FC).
At 49 days of age, one piglet from each experimental unit, with body weight closest to the average weight of the cage was slaughtered. Samples of 2 cm in length were taken from the regions corresponding to 4, 50 and 96% of the small intestine, corresponding respectively to the duodenum, jejunum and ileum. The collected material was sent to the Laboratório de Histologia do Departamento de Medicina Veterinária da Universidade Federal de Viçosa. Histological cuts were washed in physiologic solution, fixed in bouin for a 24-h period, dehydrated in ethyl alcohol, diaphanized in xylol, and included in paraffin. Each slide contained two 5 μm thick cuts. Slides were put back in xylol solution for removal of paraffin excess and then rehydrated. The pigments used were hematoxylin and eosin. After staining, the slides were dehydrated again. For the morphological readings a OLYMPUS BX50 optical microscope (40 × magnification) coupled to the image analyzer “Image-Pro Plus 1.3.2” (1994) was used. Thirty villi and their respective 30 crypts were selected, well targeted, longitudinally sectioned, and had their heights measured. The villous:crypt ratio was also computed.
On the day of slaughter, samples of intestinal content of pigs in the colon proximal were collected in sterile containers and frozen immediately. Samples were subsequently sent to the Laboratório de Microbiologia Industrial do Departamento de Microbiologia da Universidade Federal de Viçosa. The total DNA extraction of the digesta was done by following the protocol described by Lu et al. (2003) with some modifications made in the Laboratório de Microbiologia Industrial do Departamento de Microbiologia da Universidade Federal de Viçosa. To amplify the rDNA 16S gene, corresponding to total Eubacteria, universal primers of Eubacterias with the following sequences of nucleotides were used: primer 2: 5’ATTACCGCGGCTGCTGG3’ and p r i m e r : 5 ’ C G C C C G C C G C G C G C G G C G G G CGGGGCGGGGGCACGGGGGGCCTACGGGAGGCAGCAG3’ (Muyzer et al., 1993). To amplify the rDNA 16S region corresponding to Lactobacillus spp. specific primers with the sequences were used as follows: CGCCCGCCGCGCCCCGCGCCCGTCCCGCCGCCCCGC C C C T T T A Y G C G A A C A Y Y T R G G K G T CCAAACATCACVCCRACTT. The intestinal microbiota genetic profile was assessed by the electrophoresis technique in gel with gradient denaturant – DGGE (Muyzer et al., 1993). The data obtained by using the program Bionumerics, version 5.10, were used to calculate the richness, measured in operational taxonomic units (OUT), the Shannon-Weaver diversity (H’) and equitably (E’) indexes, and with similarity corresponding to the diets containing different lactose levels.
The variable R was generated using a binary matrix in which the absence of DNA band corresponding to each OUT was coded as zero (0) and the presence as one (1). For each level of lactose, the frequency of occurrence of each OUT was obtained by counting the number of presences in the treatments. The index H ‘was calculated based on the abundance, expressed as optical density of the band in the lane of the gel, using the formula:
where N = optical density in the lane, obtained by the sum of the bands optical densities in the lane; ni = optical density of each band in the lane.
E’ was calculated as:
The performance and morphological intestinal mucosa data obtained, corresponding to the pigs fed diets containing lactose supplemented or not with L-glutamine + L-glutamate, were subjected to analysis of variance and F test at 5% probability, using the program SAEG (Statistical Analysis System and Genetics, version 8.1), developed at the Universidade Federal de Viçosa. In the absence of interaction between the factors studied, the study proceeded to the analysis of the factors alone.
The evaluation of the composition of the intestinal microbiota of piglets was carried out using the program Bionumerics, version 5.10.
Results and Discussion
There was no interaction (P>0.05) between the factors studied on any parameters evaluated.
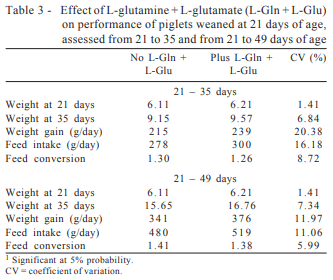
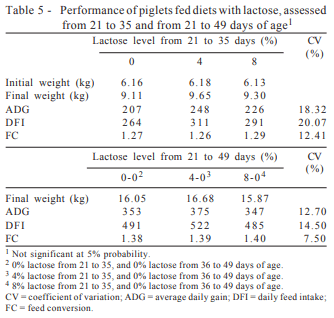
Adding L-glutamine + L-glutamate in the diets did not affect (P>0.05) the performance of pigs from 21 to 35 days of age (Table 3). However, in absolute values, the inclusion of L-glutamine + L-glutamate resulted in an increase of 4.6, 11.1, and 7.9% in the body weight at 35 days, average daily gain (ADG), and average daily feed intake (ADFI) of piglets, respectively, followed by a reduction of 3.2% in feed conversion (FC), also in absolute values.
From 21 to 49 days of age (Table 3), there was effect (P>0.05) of L-glutamine + L-glutamate on body weight at 49 days and on the body weight gain of piglets which increased 7.1 and 10.2%, respectively, compared with those fed diets without L-glutamine + L-glutamate. The ADFI and FC in this phase were not influenced by adding of L-glutamine + L-glutamate in the diets.
Beneficial effect of adding L-glutamine + L-glutamate in the diet on piglet performance was observed by Wu et al. (1996), Kitt et al. (2003) and Zou et al. (2006). Assessing four levels of glutamine supplementation (0, 0.2, 0.6, and 1.0%) on the performance of weaned piglets analyzed at weaning and in the first and second subsequent weeks, Wu et al. (1996) found a significant improvement in the order of 25% in the feed efficiency of piglets fed glutamine compared with those fed the control diet from 7 to 14 days after weaning.
In diets for weaned piglets challenged with LPS (E. coli lipopolysaccharide), Kitt et al. (2003) found that the inclusion of 5% glutamine maintained satisfactory the performance of the animals, which was not seen in those challenged and non-supplemented. In studies with piglets weaned at 21 days, Zou et al. (2006) found improvement in the FC within 10 days after weaning and for ADG within 20 days after weaning in response to the inclusion of 1% glutamine in the diet, followed by reduced rate of diarrhea and increased serum levels of growth hormone.
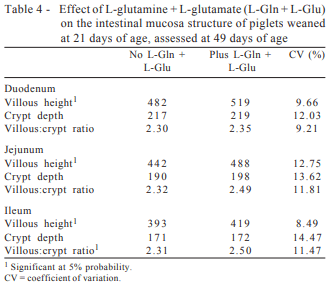
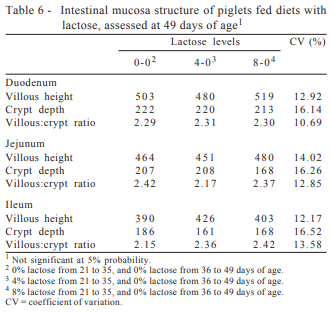
The inclusion of L-glutamine + L-glutamate resulted in an increase (P>0.05) in the villi height of the three segments of the small intestine evaluated, which were 7.7, 10.4 and 6.6% higher, respectively, in the duodenum, jejunum and ileum than those presented by piglets that were not supplemented with L-glutamine + L-glutamate (Table 4). However, the inclusion of L-glutamine + L-glutamate did not affect (P>0.05) crypt depth in any of the intestinal segments, and the values found in both groups of animals were very close to each other (Table 4).
The villous:crypt ratio, which is the result of the division between the measurement of villus height and the depth of its respective crypt, in the ileum was positively affected (P>0.05) by inclusion of L-Glu + L-Gln in the diet, evidently by the increasing in villi height (Table 4). Piglets fed diets containing L-glutamine + L-glutamate in their composition showed yet, in absolute values, villous:crypt ratio in the duodenum and jejunum 2.1 and 7.3% higher compared with those whose diets did not contain L-glutamine + L-glutamate. These results confirmed those found by Wu et al. (1996) and Yi et al. (2005).
Studying the effect of glutamine on intestinal villus height of piglets after weaning, Wu et al. (1996) reported that in addition to substantial improvement in performance, the inclusion of 1.0% glutamine in the diets of the piglets was able to prevent villus height atrophy of the jejunum in both first and the second week after weaning.
Comparing the effect of adding glutamine or plasma in diets for piglets weaned at 17 days of age and challenged with Escherichia coli, Yi et al. (2005) found that the inclusion of 2% glutamine or 7% plasma significantly increased villus height and crypt depth of piglets with regard to the non-supplemented challenged treatment, although the inclusion of glutamine provided better performance. The authors attributed the beneficial effect of glutamine, mainly to the maintenance of the intestinal mucosa structure, electrolyte absorption, and intestinal homeostasis preventing diarrhea.
Comparing the effect of adding glutamine or plasma in diets for piglets weaned at 17 days of age and challenged with Escherichia coli, Yi et al. (2005) found that the inclusion of 2% glutamine or 7% plasma significantly increased villus height and crypt depth of piglets concerning the non-supplemented challenged treatment, although the inclusion of glutamine provided better performance. The authors attributed the beneficial effect of glutamine mainly to the maintenance of the intestinal mucosa structure, electrolyte absorption, and intestinal homeostasis preventing diarrhea.
The nutrition of piglets is closely linked to the immune system. It is known that early weaning is related to immunosuppression, reduced feed intake, and susceptibility to intestinal disorders. According to Newsholme (2001), the increased demand for glutamine in conditions of stress increases the release of glutamine by the muscle, resulting in decreased concentration of this amino acid in the muscle, and elevated rates of protein degradation, which is not desirable in growing pigs.
In a study with piglets infected or not by Escherichia coli, Yoo et al. (1997) found that supplementation with glutamine was able to maintain the concentration of glutamine in the muscle, and the concentrations of lymphocytes and leukocytes. However, the authors reported no effect of supplementation with glutamine on the nonchallenged piglets, suggesting that supplementation with glutamine is important, among other factors, for the maintenance of its extracellular concentration, preventing the mobilization of muscle glutamine to meet the needs of the immune system.
According to Karinch et al. (2001), moderate or severe infections change the flow of glutamine in the body. The complexity of glutamine metabolism in different sanitary conditions of piglet production may explain why, in this research, even no effect (P> 0.05) of lactose was detected on the performance (Table 5) nor on the intestinal mucosa structure (Table 6); the inclusion of glutamine in the diet provided significant improvement in the ADG from 21 to 49 days of age and in the villi height. In general, the animals have good health, indicating low level of health challenge. Under these conditions, the small intestine is the organ of highest uptake of glutamine in the body, and it is provided primarily by the muscle (Karinch et al., 2001), which would explain the increase in villus height and, consequently, the increase in the ADG.
Under conditions of systemic inflammatory response, such as induction of immune challenge observed in the study of Kitt et al. (2003), Yi et al. (2005), and Yoo et al. (1997) the liver becomes the most important organ of glutamine uptake and exportation of this amino acid for the synthesis of immune cells due to the increase in the production of these specific cells (Karinch et al., 2001).
The effect of L-glutamine + L-glutamate in this study was self-evident although no inflammatory response was induced in the piglets. In addition to the low health challenge reflected by the low number of piglets suffering from diarrhea during the experiment and the overall health of the piglets, the environmental condition of sanitary challenge typically found on the first weeks after early weaning was attenuated by the good sanitary conditions of the rooms and by adding blood plasma and antibiotics in the experimental diets. The results obtained by Abreu et al. (2010), in a study evaluating the combination of glutamine, nucleotides and plasma in diets for weanling pigs showed that the response of piglets to plasma, the association of plasma + nucleotides or plasma + glutamine supplanted the response in performance afforded by glutamine alone, suggesting, furthermore, that the inclusion of plasma may attenuate the inflammatory response of piglets at weaning.
The levels of lactose did not influence (P>0.05) performance (Table 5) or intestinal mucosa structure of piglets (Table 6).
These results do not confirm the findings of Hauptili et al. (2005), who observed improved performance of piglets weaned at 21 days of age during the nursery phase by increasing the level of inclusion of whey powder in the diets, which was attributed to the lactose present in the food.
The lactose level in diets for piglets weaned at 21 days recommended by Bertol (1999) is 12 to 14% on the first week. In addition, Ludke et al. (1998) point out that in feeding programs for young piglets the need of milk powder inclusion in the diet rises as early as weaning is carried out, which is confirmed by Mahan et al. (2004), who worked with piglets weaned at different ages.
However, the effect of the addition of lactose in the diet of piglets may vary depending on the environment in which they are housed, as we can see in the study of Cromwell et al. (2008), who found, in three different experimental stations, different responses in piglet performance when the level of dietary lactose increased.
The inclusion of crystalline lactose, milk powder or whey powder in diets for early weaned piglets has been adopted mainly due to its effect on feed intake, especially in the first week after weaning. The maintenance of feed intake prevents villi atrophy of the intestinal mucosa (Wu et al., 1997), which is necessary for digestion and absorption of nutrients, and therefore, performance.
However, in this study, there was no effect of adding lactose in the piglets’ diet on the intestinal mucosa structure, These results corroborate those achieved by Pierce et al. (2006), who, although did not find any significant effect of lactose on villous height, crypt depth or villous:crypt ratio of piglets, observed increase in ration intake in the same period.
In a study to evaluate the replacement of dietary lactose by maltodextrin in diets for piglets weaned at 21 days of age, emphasizing its role as an energy source for piglets, Hauptili (2009) verified the viability of full replacement of lactose by maltodextrin without compromising performance.
However, part of dietary lactose may escape enzymatic digestion in the stomach and serve as a substrate for intestinal microbial fermentation. Delcenserie et al. (2008) accurately described the variety of physiological effects brought about by some genera of microorganisms, particularly regarding the production of inflammatory cytokines. Therefore, the existence of a relationship between the level of lactose inclusion in the diet of piglets and the intensity of sanitary challenge are feasible to be verified, given the importance of the study in the intestinal microbial diversity.
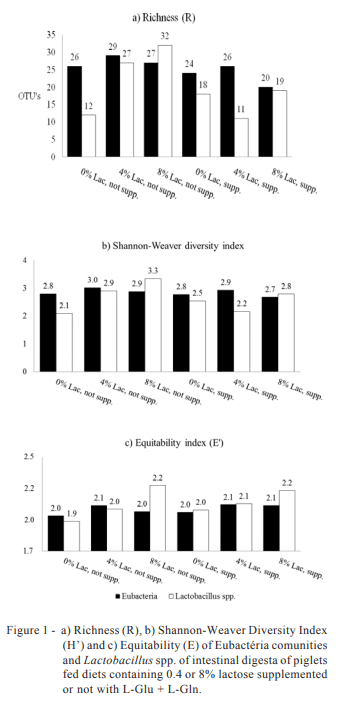
Operational Taxonomic Units (OTUs) quantitatively represent the microbial population of a given environment (Figure 1).
It was observed that the level of 4% inclusion of lactose in the diet provided the highest counts of UTOs, both in non-supplemented (29 UTOs) and supplemented diets (27 UTOs) with L-glutamine + L-glutamate. Regarding the community of Lactobacillus spp. of piglets fed diets with no addition of L-glutamine + L-glutamate the increase in the lactose level from 0 to 8% propitiated a crescent increase in the UTOs count.
However, the pattern of response to the inclusion of these amino acids in the diet for the amount of Lactobacillus spp. was not maintained as the diet with 4% lactose and L-glutamine + L-glutamate resulted in the lowest value of UTO’s (11).
Similarly, the Shannon-Weaver Diversity Index (H’) (Figure 1) followed the results of Richness (R). Considering only the community of Eubacteria, the level of 4% lactose resulted in higher values of UTOs being verified by H’ equal to 3.01 and 2.92, respectively, for piglets fed both supplemented and non-supplemented diets with L-glutamine + L-glutamate.
The Equitability (E’) is indicative of uniformity in the distribution of groups of microorganisms found in an environment, demonstrating a possible existence of dominance (Figure 1). The higher the E’, the higher the balance between populations. The results concerning Eubacteria consistently indicated that 0% lactose inclusion resulted in the lowest values of E’ in both non-supplemented (1.98) and supplemented (2.01) diets with L-glutamine + L-glutamate, which allows to infer about a likely dominance exerted by a particular microorganism in relation to others. Regarding the groups of Lactobacillus spp., there was an increase in E’ when the level of lactose was increased in the piglets fed both non-supplemented and supplemented diets, which was 2.22 and 2.18, respectively.
With regard to the Richness and intestinal microbiota diversity of piglets, although the association of L-glutamine + L-glutamate with 4 and 8% lactose did not follow the same pattern of response for Lactobacillus spp. of piglets fed non-supplemented diets, it is not possible to say that the inclusion of L-glutamine + L-glutamate influenced the composition of the microflora.
Industrial amino acids, in general, have high digestibility, being fully used until the ileum. Stool et al. (1998) postulate that glutamine is degraded along the entire small intestine, which would not allow its fermentation by bacteria in the large intestine, where samples of digesta were collected. Collectively, the results suggest that the inclusion of lactose had a beneficial effect on the microbiota and the supplementation with L-Glu + L-Gln had no influence on the microbiota. The electrophoresis in denaturing gradient gel (DGGE) shows the profiles of similarity among treatments contributing to the consolidation of the results of H’, E’ and R (Figure 2).
The occurrence of a high similarity in the intestinal microbiota of piglets that were fed diets containing the same levels of lactose (T1 and T2 = 8%, T3 and T4 and T5 = 4%, and T6 = 0% lactose) was verified for the community of Eubacteria, regardless of supplementation with L-glutamine + L-glutamate.
The similarity observed between treatments with the same level of lactose was 68.0, 76.4, and 63.8, respectively, for the levels 0.4 and 8% lactose. However, it was observed that the treatments with 0 and 4% lactose, with or without L-glutamine + L-glutamate inclusion presented average similarity, lower than that seen between treatments with the same level of lactose. The similarity is indicative of the resemblance between microbial communities in microecosystems, in which case the digesta of animals is strongly influenced by the diet (Williams et al. 2001).
The analysis of intestinal microbiota consistently indicated that the level of lactose exerted influence on the total bacterial community in the colon of piglets, regardless of the presence of L-glutamine + L-glutamate in the diets.
Conclusions
The inclusion of L-glutamine + L-glutamate in diets for piglets weaned at 21 days of age improves performance and morphology of the intestinal mucosa, regardless of the addition of lactose. The level of 8% lactose improves the composition of the intestinal microbiota of piglets.
This article was originally published in Revista Brasileira de Zootecnia. vol.41 no.1 Viçosa Jan. 2012. http://dx.doi.org/10.1590/S1516-35982012000100015. This is an Open Access article licensed under a Creative Commons Attribution License. 











.jpg&w=3840&q=75)