Introduction
It is necessary to maintain intestinal wall integrity in order to facilitate nutrient digestion and absorption processes. Intestinal morphology can be affected during inflammatory processes(Amador et al., 2007). Weaning is the most critical period in the life of the pig. During weaning, the digestive system of the piglet has to adapt rapidly from milk consumption to a solid diet. Successful adaptation requires morphological and enzymatic adjustments of the gastrointestinal tract, such as changes in morphology of intestinal crypts and villi, as well as changes in expression and activity of brush border membrane enzymes and gut microbial populations (Mosenthin, 1998; Marion et al., 2002).
During lactation the predominant bacteria in the stomach and small intestine of piglets are lactobacilli and streptococci. Early weaning leads to a brief period of fasting and disappearance of lactobacilli populations in the gastrointestinal tract. This favors the increase of enteropathogenic bacterial populations, especially Escherichia coli, during the first week post-weaning. When E. coli dies it releases proinflammatory substances, such as lipopolysaccharides (LPS) from its cell walls. Lipopolysaccharides cause sepsis and is recognized by the mammalian host as an important pathogenic agent (Amador et al., 2007).
It is known that LPS causes physical injury to the intestine, triggering a coordinated series of cellular signaling pathways that promote an inflammatory response, which in turn causes changes in the structure and functional capacity of this organ. This alteration manifests as diarrhea, which leads to a digestion process deficit and malfunction of nutrient absorption and metabolism (Pitman and Blumberg, 2000; GarcíaHerrera et al., 2003; Garcia-Herrera et al., 2008).
Lipopolysaccharides have been used to study acute infection processes, as it has highly reproducible effects while lacking the side effects associated with chronic infections(Albin et al., 2007). This study aims to evaluate the effects of LPS ingestion in weaned pigs on the molecular expression and enzymatic activity of intestinal brush border membrane for the following enzymes: sucrase-isomalatasa (SI, 3.2.1.48-10 EC), lactase-phlorizin hydrolase (LPH, EC 3.2.1.23-62), maltase-glucoamylase (MGA, EC 3.2.1.20), aminopepetidasa A (ApA, EC 3.4.11.7), aminopeptidase N (ApN , EC 3.4.11.2), and dipeptidyl peptidase IV (DpP-IV, EC 3.4.14.5).
Materials and methods
Ethical considerations
All experimental procedures were conducted according to guidelines suggested by “The International Guiding Principles for Biomedical Research Involving Animals” (CIOMS, 1985), and approved by the Ethics Committee on Animal Experimentation of Universidad Nacional de Colombia (Medellín; CEMED 001, from January 26, 2009).
Location
The field work was conducted at Universidad Nacional de Colombia, Centro San Pablo, with an altitude of 2100 meters, and a temperature range of 12 to 18 °C, corresponding to a tropical lower-montane wet forest zone (bmh-MB).
Animals
A total of 64 Landrace x Duroc male pigs were used. Animals were weaned at exactly 21 days of age, weighing 6.5 ± 0.5 kg. Pigs were group-penned in nursery crates with trough-type feeders, in a controlled temperature room (26 ± 3 °C). Water was provided ad libitum throughout the experiment. No solid feed was offered to the piglets during lactation.
Diets
Four experimental diets were evaluated; a basal diet without LPS (control) and different levels (0.3, 0.5, and 1.0 µg LPS/mg feed) of LPS from E. coli (serotype 0111: B4; Sigma-Aldrich, St. Louis, MO, USA) were used. The basal diet (BD) consisted of milk, milk byproducts, and was supplemented with vitamins, minerals, and HCL-lysine to meet the nutritional requirements for the weaning pig (NRC, 2012). Pigs were offered a minimum of 300 g feed/ day/crate—additional feed was provided when required by the animals. The diets were fed from days 1 to 10 post-weaning.
Small intestine sampling
Pigs were sequentially slaughtered as follows: a total of 16 pigs were slaughtered on weaning day 1 (representing four pigs per each LPS-inclusion level) to assess the overall health status and to evaluate the macroscopic appearance of the organs before pigs received LPS. Four pigs of each LPS-inclusion level (0, 0.3, 0.5, and 1.0 µg LPS/mg feed) were slaughtered on days 5, 7, and 10 after weaning. All animals were slaughtered 2.5 hours after their last meal and were sedated by carbon dioxide inhalation for three minutes pre-slaughter. Pigs were then slaughtered by exsanguination through section of the jugular vein. After slaughter, the small intestine was removed from the pyloric junction to the ileocecal valve (Segalés and Domingo, 2003). The intestine was lined up on a table, measured without any tension, divided into three equally sized sections (duodenum, jejunum, and ileum) and 20 cm samples from the center of each segment were taken. The digesta contained in the samples was removed by a cold saline infusion wash as previously described (Makkink et al., 1994; Reis de Souza and Mariscal, 2005). Then, 1 cm subsamples were cut from each segment and the intestinal mucosa was removed by gentle scraping with a microscope slide. Intestinal mucosa and samples for RNA analysis were immediately frozen in liquid nitrogen and stored at -70 °C until analysis.
Total RNA extraction from gut tissue and material quality check
Tissue samples (25 mg) were mashed in mortar using liquid nitrogen. The RNA was extracted using an ULTRACLEANTM Tissue & Cells RNA Isolation Kit (MO BIO Laboratories Inc., San Diego, CA, USA) and stored at -80 °C. Integrity of the extracted material was assessed by running the samples on agarose gel with 1% ethidium bromide (100V for 50 min) and observing the rRNA bands. When not enough material or bands were clearly observed, the extraction was repeated under identical conditions.
cDNA synthesis from total RNA
A QuantiTect® Reverse Transcription Kit (QIAGEN, Valencia, CA, USA) was used to synthesize cDNA from the total RNA extracted. RNA samples (1 μg) were used for all reverse transcription reactions. Genomic DNA debris was removed and enzyme plus triphosphate nucleotides were added to synthesize cDNA following the manufacturer’s recommendations. The obtained cDNA was stored at -20 °C until analysis (Kubista et al., 2006).
Reverse transcription coupled -PCR (RT-PCR) from cDNA for intestinal enzymes
The primers of the genes of interest and constitutive expression (cyclophilin) are shown in Table 1. These primers were taken from the literature, verifying that their design had been performed on the intron/ exon edges to avoid amplification of genomic DNA. Additionally, primers were evaluated through bioinformatic analysis to verify their specificity with the studied mRNA or rRNA fragment and the absence of secondary structures such as lenses or dimer formations with each other or with the other primer. Primers were chosen to amplify fragments with fewer than 150 base pairs, as recommended by some researchers (Bustin, 2000; Kubista et al., 2006).
To determine the expression level of each gene, a relative quantification method using a constitutively expressed gene (cyclophilin) was used, following recommendations from several reports about gene choice and overall normalization strategy (Muller et al., 2002; Vandesompele et al., 2002; Huggett et al., 2005). The amount of material from which RNA was extracted, the RNA amount in the reverse transcription reaction, and the amount of cDNA for PCR amplification were identical for all samples, quantified in Nanodrop 1000 (Thermo Fisher Scientific Inc., Waltham, MA, USA). The constitutively expressed gene chosen was validated in previous trials under similar experimental conditions (Paulin et al., 2007).
The results were analyzed using ImageJ software, version 1.43. Amplification of the mRNA of LPH, SI, MGA, ApN, ApA, and DDP-IV by RT-PCR was conducted as described above on individual samples of the proximal, middle and distal intestine segments (Petersen et al., 2001). The density of RTPCR products from each enzyme was expressed in units relative to the band density of the constitutive expression gene (cyclophilin gene).
Intestinal mucosa process for detection of enzyme activity
Intestinal mucosa samples obtained from small intestine segments were processed and analyzed at the Molecular Studies Interdisciplinary Group laboratory (GIEM) of Universidad de Antioquia (Colombia). Samples were freeze-dried (Labcondo Freeze Dry System/Freezone 4.5, Kansas City, MO, USA), ground and soluble proteins were then extracted using a buffer solution (50 mM, pH 6.5) containing NaCl (250 mM) for 10 min at 4 °C. The resulting solution was centrifuged at 12000 rpm for 15 min at 4 °C, and supernatant was stored in a separate eppendorf at -20 °C until analysis.
Enzyme activity determination (SI; EC 3.2.1.48-10)
The sucrase-isomaltase (SI), lactase-phlorizin hydrolase (LPH; EC 3.2.1.23-62), maltaseglucoamylase (MgA; EC 3.2.1.20), aminopeptidase N (ApN; EC 3.4.11.2), and dipeptidyl peptidase IV (DpP-IV; EC 3.4.14.5) enzymatic activity were determined according to the method by Sangild et al. (1995). To SI, the supernant of the extracted sample (733 μL) were mixed with 1470 μL of a citric acid buffer solution at pH 6.5 (50 mM) containing sucrose (29.21 mM) and NaCl (250 mM). The reaction was performed at 37 °C, with agitation before and after each measurement. Time periods were 0, 15, 30, 45, and 60 minutes. At each time period 200 μL of aliquots was mixed with 200 μL DNS (Solution A: 0.25 g of acid 3,5 dinitrosalicylic acid + 5 mL NaOH 2M + 12 mL of distilled water; Solution B: 7.5 g of potassium sodium tartrate in 25 mL of distilled water) in test tubes. The mixture was heated for 10 min at 100 °C, added with 600 μL distilled water, and absorbance was read at 540 nm. The analysis was performed using the method of initial velocities. Results are shown as the average glucose (µmol) released per minute per gram of sample, as the product of sucrose hydrolysis. Results took the calibration curve into account.
To determine LPH, MgA, ApN, DpP-IV enzymatic activity, the samples were diluted 1/10 in a buffer solution at pH 6.5 (50 mM) containing NaCl (250 mm) and substrate (specified concentration) for each of the enzymes in question. 50 μL samples were reacted with 100 µL Gly-Pro-p-nitroanilide HCl substrate for DPPIV activity, with 100 µL L-alanine 4 nitroanilide HCl substrate for ApN activity, with 100 µL 4-Nitrophenyl β-D-galactopyranoside for lactase activity, and with 100 µL 4-Nitrophenyl α-Dglucopyranoside for maltase activity. Concentration in all substrates used was 250 μM. Substrates were prepared from a stock solution (20 mM) at pH 6.5 (50 mM), which also contained in DMSO (10 mM) and NaCl (250 mm). The reaction was performed in ELISA wells. Absorbance (405 nm) was read every 2 minutes for 16 min. Analyses were conducted using the method of initial rates. Results are shown as the average (µmol) of p-nitroaniline (the hydrolysis product of each aminopeptidase), and the average (µmol) of p-nitrophenol (hydrolysis product of disaccharidases) released per minute per gram of sample (µmol/min/g). The results take the calibration curve into account.
Statistical design
The experiment was performed as a randomized block design (2 blocks, by weight of the animals) in a 4x4 factorial arrangement (four experimental diets and four periods after weaning; Steel and Torrie, 1980). Animals were blocked by initial weight. Each animal was assigned to one of 16 treatments and each treatment had 4 repetitions. The General Linear Model procedure (GLM) of SAS® software, version 9.1 (SAS Institute Inc., Cary, NC, USA) (2006) was used for the statistical analysis of the data. Duncan test (p<0.05) was used to separate treatment means.
Results
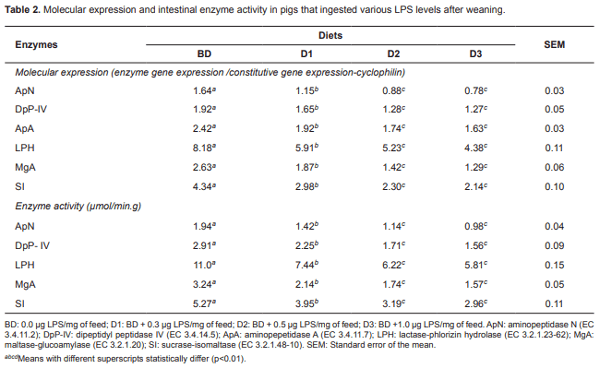
Pigs fed the basal diet did not show health problems, while pigs receiving LPS had rectal temperature increases above 38 °C throughout the experiment. Molecular expression of enzymes was obtained as the relationship between gene expression for each enzyme and the constitutive gene expression (cyclophilin). Average molecular expression and enzyme activity (µmol/min/g) of the enzymes studied for each diet, period of exposure and intestinal section are presented in Tables 2, 3, and 4. No statistical interaction was observed between LPS concentrations and post-weaning periods for any of the variables studied.
Table 2 shows that diets D2 and D3 had the lowest expression of aminopeptidases ApN, DpPIV, and ApA (p<0.01). Animals on D3 showed the lowest values of these enzymes (0.78, 1.27, and 1.63, respectively) compared to samples from BD pigs (1.64, 1.92, and 2.42, respectively). No statistical difference was observed between D2 and D3 (p>0.01). The LPH, MgA, and SI disaccharidases also showed differences between diets (p<0.01). Animals fed D3 had the lowest expression of these enzymes (4.38, 1.29, and 2.14, respectively) compared to BD (8.18, 2.63, and 4.34, respectively). Similarly, differences were statistically significant (p<0.01) between the experimental diets for LPH, MgA, and SI disaccharidases. The animals fed D3 showed the lowest molecular expression values for intestinal disaccharidases (4.38, 1.29, and 2.14, respectively) compared with animals fed BD (8.18, 2.63, and 4.34, respectively). ApN and DpP-IV differed (p<0.01) between diets. Animals on D3 had lower values for those two enzymes (0.98, and 1.56 µmol/min/g, respectively) with respect to BD (1.94 and 2.91 µmol/min/g, respectively), and did not show activity differences between D2 and D3. Similarly, LPH, MgA, and SI disaccharidases had differences (p<0.01) between diets. Animals fed D3 showed the lowest activity for those enzymes (Table 2; 5.81, 1.57, and 2.96 µmol/min/g, respectively) compared to BD (11.0, 3.24, and 5.27 µmol/min/g, respectively).
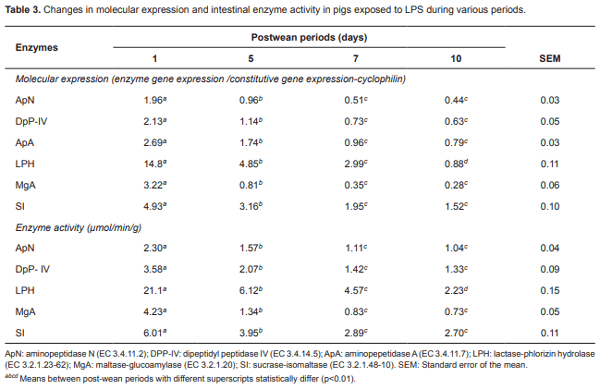
Table 3 presents molecular expression of aminopeptidases ApN, DpP-IV, and ApA at different LPS exposure periods for the average diet. A significant decrease (p<0.01) was found from day one after weaning, where animals weaned for 10 days showed the lowest values (0.44, 0.63, and 0.75, respectively). Similarly, the molecular expression of disaccharidases LPH, MgA, and SI decreased (p<0.01) from day one post-weaning, reaching its lowest level on day 10 postweaning (0.88, 0.28, and 1.52, respectively).
With regard to the enzymatic activity of ApN and DpP-IV aminopeptidases during LPS exposure, the overall evaluation of animals (BD, B1, D2, and D3; Table 3) shows a significant decrease (p<0.01) from day one post-weaning. Animals showed the lowest values at day 10 post-weaning (1.04 and 1.33 µmol/ min/g, respectively). Similarly, enzymatic activity of LPH, MgA, and SI decreased (p<0.01) from day one, reaching its highest level at day 10 post-weaning (2.23, 0.73, and 2.70 µmol/min/g, respectively).
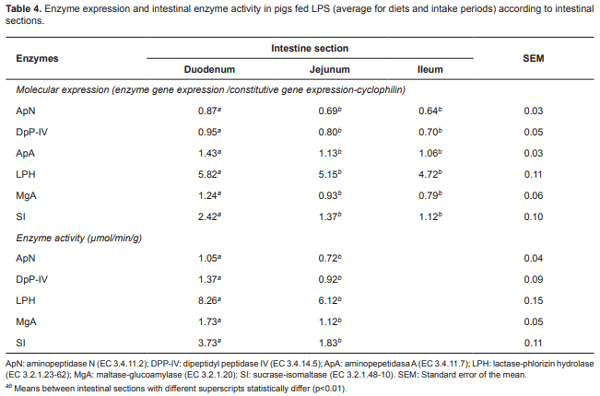
The enzyme expression differed among the intestinal segments (Table 4; p<0.01). The duodenum had the highest expression of ApN, DpP-IV, and ApA (0.87, 0.95, and 1.43, respectively). The ileum showed the lowest values for those enzymes (0.64, 0.70, and 1.06, respectively). There were no enzyme expression differences between jejunum and ileum (p> 0.01). Duodenum had the highest molecular expression values for disaccharidases LPH, MgA, and SI (5.82, 1.24, and 2.42, respectively). There were no differences between jejunum and ileum for aminopeptidases expression (p>0.01).
With regard to intestinal sections, there were significant changes (p<0.01) in the activity of the studied enzymes study. The duodenum had the highest enzymatic activity for ApN and DpPIV aminopeptidases (1.05 and 1.47 µmol/min/g, respectively) and LPH, MGA, and SI disaccharidases (8.26, 1.73, and 3.73 µmol/min/g, respectively). Ileum enzymatic study was not conducted because there were no differences in enzyme molecular expression between jejunum and ileum (Table 4).
Discussion
The study focused on the effects of LPS levels on protein expression and enzyme activity from intestinal segments after various periods of exposure. Few reports have been found on LPS effects using this animal model, which makes it difficult to compare the results.
Intestinal digestion and absorption processes can be drastically impaired by the presence of pathogenic bacteria (Fan, 2002). Some years ago, alterations of intestinal processes were considered to be a result of allergic reactions to food proteins (Dréau and Lallès, 1999; Lallès et al., 2004). However, enteric pathogens and their toxins are now recognized as the main causes of this disfunction (Nabuurs et al., 1996). Toxins produced by these pathogens reach their maximum stability and ability to bind the intestinal epithelium during the first four days after weaning (Lallès et al., 2004).
Bacteria-derived LPS compounds are capable of activating a wide variety of signaling pathways through uncontrolled synthesis of pro-inflammatory cytokines, mainly TNF-α (Johnson et al., 2005). According to the report by Parra et al. (2013), LPS increases the TNF-α expression at the intestinal level in weaned pigs. The TNF-α has mitotic effects on the intestinal mucosa (Zachrisson et al., 2001; Schaeffer et al., 2006), stimulating morphological changes, such as decrease in villus height, increased crypt depth, reduction in enzyme activity, and occurrence of diarrhea. These factors may contribute to high mortality rates of pigs in this stage (Muller et al., 2002).
The effects on intestine functionality observed in this study could be explained by LPS’s ability to activate a variety of mediators and intracellular signaling pathways (Pié et al., 2004; Amador et al., 2007) that affect normal cell growth and induce tissue inflammation (Yoo et al., 2000). Moreover, LPS causes significant changes in intestinal structure and functional capacity, reducing villi height and area, increasing villi width, as well as increasing depth and width of intestinal glands (GarciaHerrera et al., 2008). The LPS also increase paracellular transport of molecules favoring the incidence of diarrhea and altering digestion, absorption, and nutrient metabolism processes (Pitman and Blumberg, 2000; García-Herrera, 2003).
It has been reported that LPS potentiates TNF-α production (Delgado et al., 1999), which activates a variety of signaling pathways (Pié et al., 2004; Amador et al., 2007), in turn affecting cell growth by stimulation of apoptosis (Ruemmele et al., 1999). Studies in gnotobiotic piglets indicate that E. coli increases general cell turnover by stimulating apoptosis through the expression of TNF-α and increased cell proliferation. However, it has not yet been established whether a similar mechanism causes the decrease of intestinal cells and its atrophy in commercial pigs (Willing and van Kessel, 2007).
In conclusion, data from this study suggest that LPS from E. coli decreases intestinal enzyme expression and activity. This could be due to its effect on the early inflammatory response, which is known to increase the synthesis and production of TNF-α, affecting cell turnover and growth by stimulating intestinal mucosal apoptosis.
Acknowledgements
The authors wish to thank the Sustainability Project 2011-2012 (CODI, Universidad de Antioquia) its support to this study.
Conflicts of interest
The authors declare they have no conflicts of interest with regard to the work presented in this report.










.jpg&w=3840&q=75)